About the Report
Base Year 2024Middle East Syphilis Testing Market Overview
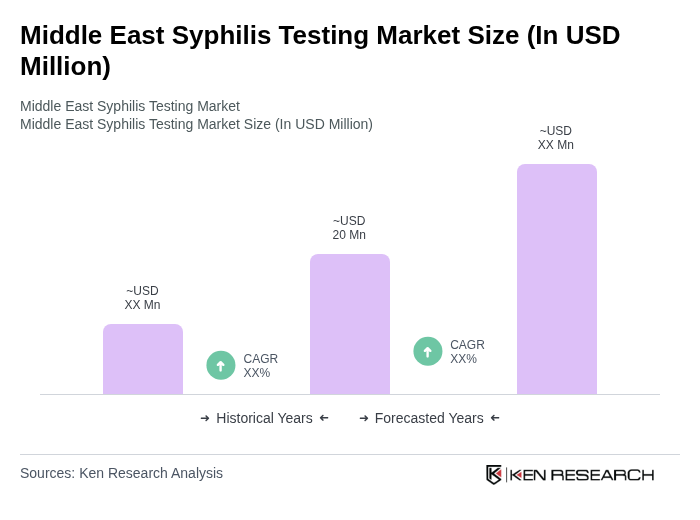
- The Middle East Syphilis Testing Market is valued at USD 20 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of sexually transmitted infections (STIs), expansion of government-led screening programs, and rapid advancements in diagnostic technologies. The rising prevalence of syphilis, particularly among high-risk populations and in urban centers, continues to drive demand for effective testing solutions .
- Key countries in this market include Saudi Arabia, the United Arab Emirates, and Egypt. These nations lead the region due to robust healthcare infrastructure, government support for STI testing initiatives, and a relatively high prevalence of syphilis. Urbanization and rising healthcare expenditure further strengthen their market position, while public health campaigns and integration of syphilis screening into maternal and child health programs are notable drivers .
- The Saudi Arabian government has implemented the "National Policy and Strategy for Prevention and Control of Sexually Transmitted Infections, 2023" issued by the Ministry of Health. This binding instrument mandates routine syphilis screening for pregnant women, increased funding for public health campaigns, and the expansion of diagnostic facilities, significantly enhancing early detection and management of syphilis cases in the country .
Middle East Syphilis Testing Market Segmentation
By Type:The market is segmented by testing methods, including Serological Tests, Nucleic Acid Amplification Tests, Rapid Point-of-Care Tests, and Immunoassays & Others. Each method addresses specific clinical needs, with serological and rapid tests widely used for screening and confirmation, while nucleic acid amplification is utilized for high-sensitivity detection in specialized settings .
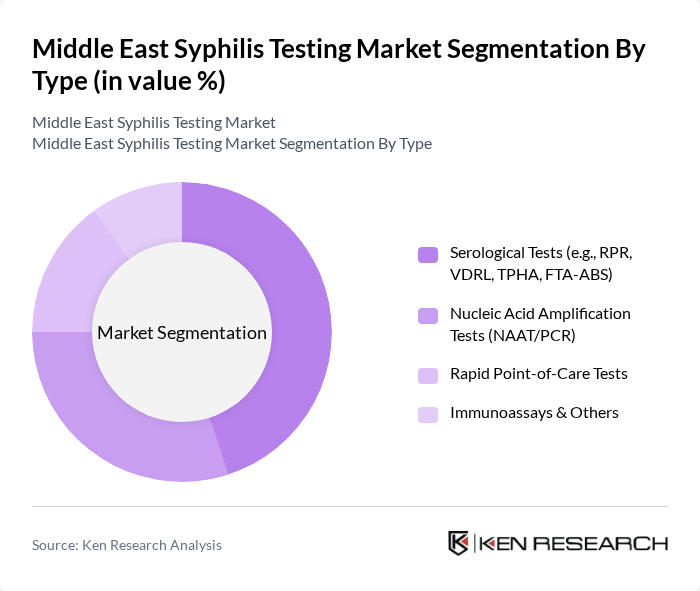
The Serological Tests segment dominates the market due to their widespread adoption in clinical and public health settings for syphilis diagnosis. Tests such as RPR and VDRL are preferred for their cost-effectiveness, reliability, and suitability for large-scale screening. The growing prevalence of syphilis and the need for rapid, accurate testing have led to higher uptake of these methods. Accessibility of serological tests in both urban and rural healthcare facilities further reinforces their leading market position .
By End-User:The market is also segmented by end-users, including Hospitals, Diagnostic Laboratories, Public Health Agencies & Screening Programs, Clinics & Physician Offices, and Others. Each category plays a vital role in the distribution and utilization of syphilis testing services, with hospitals and laboratories accounting for the majority of test volumes .
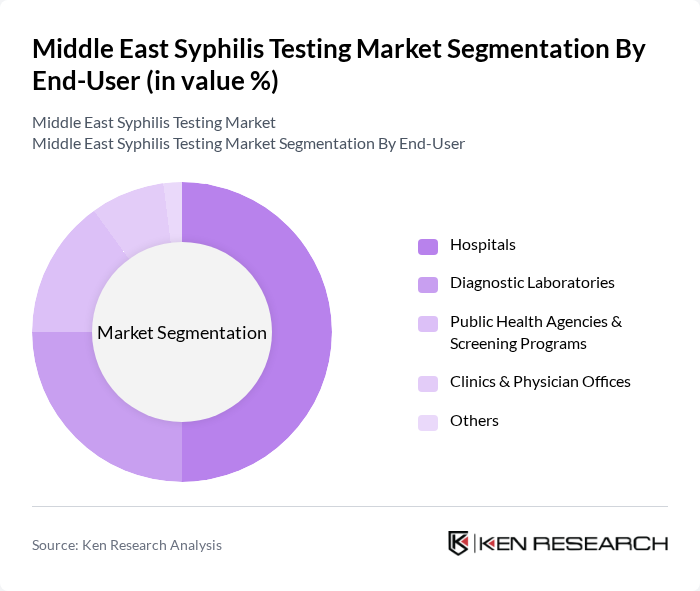
Hospitals are the primary end-users in the syphilis testing market, attributed to their comprehensive healthcare services and capacity for immediate treatment following diagnosis. The integration of syphilis testing into hospital-based care pathways ensures efficient patient management and follow-up. Hospitals also possess the infrastructure and resources to perform a broad range of diagnostic tests, making them the preferred setting for syphilis screening and confirmation .
Middle East Syphilis Testing Market Competitive Landscape
The Middle East Syphilis Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, Siemens Healthineers, Bio-Rad Laboratories, Cepheid, Hologic, Inc., Becton, Dickinson and Company, Thermo Fisher Scientific, Ortho Clinical Diagnostics, QuidelOrtho Corporation, Alere Inc. (now part of Abbott), GenMark Diagnostics, Grifols S.A., MedMira Inc., Chembio Diagnostic Systems, Inc., BioMérieux S.A., Trinity Biotech plc, Transasia Bio-Medicals Ltd., Shenzhen Bioeasy Biotechnology Co., Ltd., Beckman Coulter, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
Middle East Syphilis Testing Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Syphilis:The Middle East has witnessed a significant rise in syphilis cases, with reported infections increasing by 30% from 2018 to 2022, according to the World Health Organization. In future, approximately 16,000 new cases were documented across the region. This alarming trend necessitates enhanced testing capabilities, driving demand for syphilis testing services. The growing incidence highlights the urgent need for effective screening and treatment strategies to combat this public health challenge.
- Rising Awareness and Education:Public health campaigns have significantly improved awareness regarding sexually transmitted infections (STIs) in the Middle East. In future, over 65% of the population reported increased knowledge about syphilis and its implications, as per regional health surveys. Educational initiatives led by health ministries and NGOs have contributed to this rise, fostering a culture of proactive health management. This heightened awareness is expected to drive more individuals to seek testing and treatment, further propelling market growth.
- Government Initiatives for Health Screening:Governments in the Middle East are increasingly prioritizing health screening initiatives, with budgets for public health programs rising by 25% in future. Countries like Saudi Arabia and the UAE have launched national campaigns aimed at increasing STI testing rates. These initiatives include free testing services and mobile clinics, which are expected to enhance accessibility and encourage more individuals to undergo syphilis testing, thereby expanding the market significantly.
Market Challenges
- Stigma Associated with STIs:The stigma surrounding sexually transmitted infections remains a significant barrier to testing in the Middle East. A future study indicated that nearly 45% of individuals avoid seeking testing due to fear of social repercussions. This cultural challenge hampers public health efforts and discourages individuals from accessing necessary healthcare services, ultimately limiting the growth potential of the syphilis testing market in the region.
- Limited Access to Healthcare Facilities:Access to healthcare services, particularly in rural areas, poses a significant challenge for syphilis testing. According to the World Bank, approximately 30% of the population in rural regions lacks access to basic healthcare facilities. This limited access results in lower testing rates and delayed diagnoses, which can exacerbate the spread of syphilis. Addressing these disparities is crucial for improving testing rates and overall public health outcomes.
Middle East Syphilis Testing Market Future Outlook
The future of the Middle East syphilis testing market appears promising, driven by technological advancements and increased public health funding. The integration of digital health solutions is expected to enhance testing accessibility and efficiency. Additionally, the growing emphasis on preventive healthcare measures will likely lead to more proactive testing initiatives. As governments and organizations continue to address stigma and improve healthcare access, the market is poised for significant growth, ultimately contributing to better health outcomes in the region.
Market Opportunities
- Expansion of Telehealth Services:The rise of telehealth services presents a unique opportunity for syphilis testing in the Middle East. With a projected increase in telehealth consultations by 45% in future, remote testing options can significantly enhance accessibility for individuals in underserved areas. This shift can facilitate timely diagnosis and treatment, ultimately improving public health outcomes.
- Development of Rapid Testing Kits:The demand for rapid testing kits is on the rise, with an estimated market growth of 30% anticipated in future. These kits offer quick results and ease of use, making them ideal for widespread distribution. Their development can significantly enhance testing rates, particularly in remote areas, thereby addressing the urgent need for effective syphilis screening solutions.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Serological Tests (e.g., RPR, VDRL, TPHA, FTA-ABS) Nucleic Acid Amplification Tests (NAAT/PCR) Rapid Point-of-Care Tests Immunoassays & Others |
| By End-User | Hospitals Diagnostic Laboratories Public Health Agencies & Screening Programs Clinics & Physician Offices Others |
| By Distribution Channel | Direct Sales Online Sales Distributors/Wholesalers Pharmacies Others |
| By Geography | GCC Countries (Saudi Arabia, UAE, Kuwait, Oman, Qatar, Bahrain) Levant Region (Jordan, Lebanon, Syria, Palestine) North Africa (Egypt, Libya, Sudan, etc.) Others (Yemen, Iraq, etc.) |
| By Testing Method | Laboratory-Based Testing Point-of-Care Testing Home/At-Home Testing Others |
| By Age Group | Adolescents (10-19 years) Adults (20-64 years) Elderly (65+ years) Others |
| By Risk Group | High-Risk Populations (e.g., MSM, sex workers) General Population Pregnant Women Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Public Health Authority)
Manufacturers and Producers of Diagnostic Kits
Distributors and Retailers of Medical Supplies
Healthcare Providers and Clinics
Non-Governmental Organizations (NGOs) focused on Sexual Health
Pharmaceutical Companies involved in STI Treatments
Health Insurance Companies
Players Mentioned in the Report:
Abbott Laboratories
Roche Diagnostics
Siemens Healthineers
Bio-Rad Laboratories
Cepheid
Hologic, Inc.
Becton, Dickinson and Company
Thermo Fisher Scientific
Ortho Clinical Diagnostics
QuidelOrtho Corporation
Alere Inc. (now part of Abbott)
GenMark Diagnostics
Grifols S.A.
MedMira Inc.
Chembio Diagnostic Systems, Inc.
BioMerieux S.A.
Trinity Biotech plc
Transasia Bio-Medicals Ltd.
Shenzhen Bioeasy Biotechnology Co., Ltd.
Beckman Coulter, Inc.
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Middle East Syphilis Testing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Middle East Syphilis Testing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Middle East Syphilis Testing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of syphilis in the region
3.1.2 Rising awareness and education about sexually transmitted infections
3.1.3 Government initiatives for health screening and testing
3.1.4 Advancements in testing technologies
3.2 Market Challenges
3.2.1 Stigma associated with sexually transmitted infections
3.2.2 Limited access to healthcare facilities in rural areas
3.2.3 High costs of testing and treatment
3.2.4 Regulatory hurdles in the approval of new testing methods
3.3 Market Opportunities
3.3.1 Expansion of telehealth services for remote testing
3.3.2 Partnerships with NGOs for outreach programs
3.3.3 Development of rapid testing kits
3.3.4 Increased funding for public health initiatives
3.4 Market Trends
3.4.1 Shift towards point-of-care testing
3.4.2 Integration of digital health solutions
3.4.3 Focus on preventive healthcare measures
3.4.4 Growing demand for home testing kits
3.5 Government Regulation
3.5.1 Mandatory reporting of syphilis cases
3.5.2 Guidelines for testing and treatment protocols
3.5.3 Funding for public health campaigns
3.5.4 Regulations on the approval of diagnostic tests
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Middle East Syphilis Testing Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Middle East Syphilis Testing Market Segmentation
8.1 By Type
8.1.1 Serological Tests (e.g., RPR, VDRL, TPHA, FTA-ABS)
8.1.2 Nucleic Acid Amplification Tests (NAAT/PCR)
8.1.3 Rapid Point-of-Care Tests
8.1.4 Immunoassays & Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Diagnostic Laboratories
8.2.3 Public Health Agencies & Screening Programs
8.2.4 Clinics & Physician Offices
8.2.5 Others
8.3 By Distribution Channel
8.3.1 Direct Sales
8.3.2 Online Sales
8.3.3 Distributors/Wholesalers
8.3.4 Pharmacies
8.3.5 Others
8.4 By Geography
8.4.1 GCC Countries (Saudi Arabia, UAE, Kuwait, Oman, Qatar, Bahrain)
8.4.2 Levant Region (Jordan, Lebanon, Syria, Palestine)
8.4.3 North Africa (Egypt, Libya, Sudan, etc.)
8.4.4 Others (Yemen, Iraq, etc.)
8.5 By Testing Method
8.5.1 Laboratory-Based Testing
8.5.2 Point-of-Care Testing
8.5.3 Home/At-Home Testing
8.5.4 Others
8.6 By Age Group
8.6.1 Adolescents (10-19 years)
8.6.2 Adults (20-64 years)
8.6.3 Elderly (65+ years)
8.6.4 Others
8.7 By Risk Group
8.7.1 High-Risk Populations (e.g., MSM, sex workers)
8.7.2 General Population
8.7.3 Pregnant Women
8.7.4 Others
9. Middle East Syphilis Testing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, Small)
9.2.3 Regional Market Share (%)
9.2.4 Revenue from Syphilis Testing (USD Million)
9.2.5 Revenue Growth Rate (CAGR %)
9.2.6 Number of Syphilis Test Kits Sold (Units)
9.2.7 Product Portfolio Breadth (Number of Syphilis Test Types)
9.2.8 Regulatory Approvals (Number of Middle East Country Approvals)
9.2.9 Distribution Network Coverage (Number of Countries/Distributors)
9.2.10 R&D Investment in Infectious Disease Diagnostics (USD Million)
9.2.11 Time-to-Result (Average Minutes for Rapid Tests)
9.2.12 Customer Satisfaction/Net Promoter Score (NPS)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Abbott Laboratories
9.5.2 Roche Diagnostics
9.5.3 Siemens Healthineers
9.5.4 Bio-Rad Laboratories
9.5.5 Cepheid
9.5.6 Hologic, Inc.
9.5.7 Becton, Dickinson and Company
9.5.8 Thermo Fisher Scientific
9.5.9 Ortho Clinical Diagnostics
9.5.10 QuidelOrtho Corporation
9.5.11 Alere Inc. (now part of Abbott)
9.5.12 GenMark Diagnostics
9.5.13 Grifols S.A.
9.5.14 MedMira Inc.
9.5.15 Chembio Diagnostic Systems, Inc.
9.5.16 BioMérieux S.A.
9.5.17 Trinity Biotech plc
9.5.18 Transasia Bio-Medicals Ltd.
9.5.19 Shenzhen Bioeasy Biotechnology Co., Ltd.
9.5.20 Beckman Coulter, Inc.
10. Middle East Syphilis Testing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Health Initiatives
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Evaluation Criteria
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Health Infrastructure
10.2.2 Funding for Testing Programs
10.2.3 Partnerships with Private Sector
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Testing Facilities
10.3.2 Affordability of Tests
10.3.3 Awareness and Education Gaps
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Technology Acceptance
10.4.3 Integration with Existing Systems
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Testing Outcomes
10.5.2 Cost-Benefit Analysis
10.5.3 Expansion into New Markets
10.5.4 Others
11. Middle East Syphilis Testing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of health ministry reports and public health data from Middle Eastern countries
- Review of academic journals and publications focusing on syphilis prevalence and testing methodologies
- Examination of market reports from health organizations and NGOs involved in sexual health initiatives
Primary Research
- Interviews with healthcare professionals specializing in infectious diseases and sexual health
- Surveys conducted with laboratory managers and technicians involved in syphilis testing
- Focus groups with patients and advocacy groups to understand testing perceptions and barriers
Validation & Triangulation
- Cross-validation of findings with epidemiological data and testing rates from regional health authorities
- Triangulation of qualitative insights from interviews with quantitative data from surveys
- Sanity checks through expert panel reviews comprising public health officials and researchers
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total market size based on national health expenditure and syphilis testing budgets
- Segmentation of the market by country, testing type, and demographic factors
- Incorporation of regional health initiatives and funding allocations for syphilis testing programs
Bottom-up Modeling
- Collection of data on the number of testing facilities and their testing capacities across the region
- Estimation of average costs per test and volume of tests conducted annually
- Analysis of patient demographics to project testing demand based on risk factors
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating trends in sexual health awareness and testing accessibility
- Scenario modeling based on potential changes in public health policy and funding for sexual health
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Officials | 60 | Health Ministry Representatives, Epidemiologists |
| Laboratory Managers | 50 | Lab Directors, Quality Control Managers |
| Healthcare Providers | 70 | Doctors, Nurses, Sexual Health Counselors |
| Patient Advocacy Groups | 40 | Advocacy Leaders, Community Health Workers |
| Testing Facility Administrators | 40 | Facility Managers, Operations Directors |
Frequently Asked Questions
What is the current value of the Middle East Syphilis Testing Market?
The Middle East Syphilis Testing Market is valued at approximately USD 20 million, reflecting a five-year historical analysis. This growth is attributed to increased awareness of sexually transmitted infections, government-led screening programs, and advancements in diagnostic technologies.