Oman In Vitro Diagnostics (IVD) Quality Control Market Overview
- The Oman In Vitro Diagnostics (IVD) Quality Control Market is valued at USD 150 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases, the rising demand for accurate diagnostic testing, and advancements in IVD technologies. The market is also supported by the growing healthcare infrastructure and government initiatives aimed at improving healthcare services. For instance, the Middle East region, including Oman, is witnessing significant investments in healthcare infrastructure, which is boosting the demand for quality control measures in diagnostics.
- Muscat, the capital city, is a dominant player in the market due to its advanced healthcare facilities and concentration of diagnostic laboratories. Other regions like Dhofar and Al Batinah also contribute significantly, driven by their expanding healthcare services and increasing awareness of quality control in diagnostics. The overall growth in healthcare expenditure in these areas further enhances their market presence.
- In Oman, there is a strong emphasis on adhering to international quality standards for IVD testing. While specific regulations are not detailed here, the general trend in the region is towards ensuring compliance with global standards to enhance patient safety and care. Compliance with these standards is crucial for laboratories to maintain their operational licenses and gain accreditation.
 Quality Control Market.png)
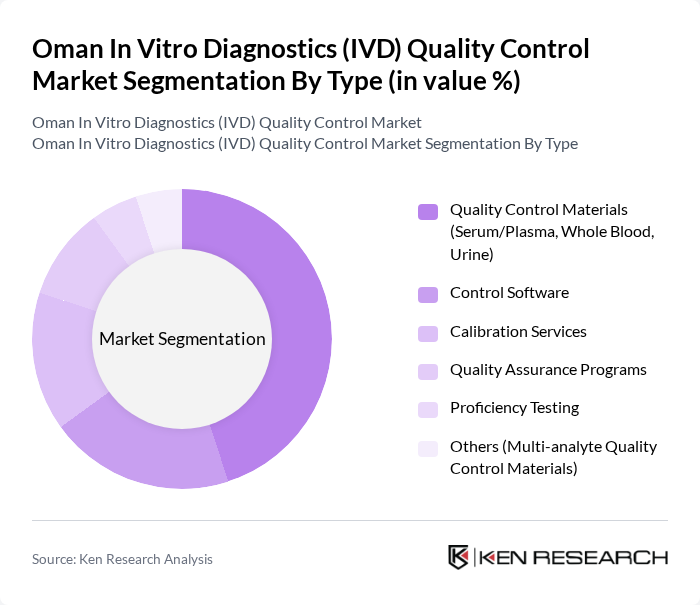
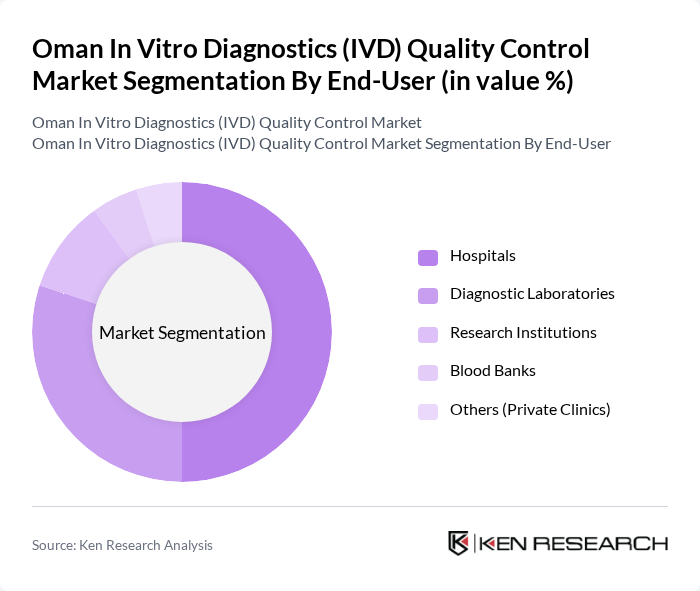
Oman In Vitro Diagnostics (IVD) Quality Control Market Segmentation
By Type:The market is segmented into various types, including Quality Control Materials, Control Software, Calibration Services, Quality Assurance Programs, Proficiency Testing, and Others. Among these, Quality Control Materials, which include Serum/Plasma, Whole Blood, and Urine, dominate the market due to their essential role in ensuring the accuracy of diagnostic tests. The increasing focus on patient safety and the need for reliable test results drive the demand for these materials.
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Blood Banks, and Others. Hospitals are the leading end-users, driven by the increasing number of diagnostic tests performed and the need for high-quality control measures to ensure patient safety. The growing trend of point-of-care testing in hospitals further enhances their demand for quality control solutions.
Oman In Vitro Diagnostics (IVD) Quality Control Market Competitive Landscape
The Oman In Vitro Diagnostics (IVD) Quality Control Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, Thermo Fisher Scientific, Ortho Clinical Diagnostics, Sysmex Corporation, Beckman Coulter, Becton Dickinson, Agilent Technologies, QIAGEN, Hologic, Mindray, PerkinElmer, Grifols contribute to innovation, geographic expansion, and service delivery in this space.
Oman In Vitro Diagnostics (IVD) Quality Control Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Diseases:The rise in chronic diseases such as diabetes and cardiovascular conditions is a significant growth driver for the IVD quality control market in Oman. According to the World Health Organization, approximately 25% of the Omani population is affected by chronic diseases, leading to an increased demand for reliable diagnostic tests. This trend is expected to escalate healthcare spending, projected to reach OMR 2 billion in future, thereby boosting the IVD market.
- Rising Demand for Accurate Diagnostic Testing:The need for precise diagnostic testing is growing, driven by the increasing awareness of disease prevention and management. In Oman, the healthcare sector is projected to invest OMR 250 million in advanced diagnostic technologies in future. This investment reflects a commitment to enhancing diagnostic accuracy, which is crucial for effective treatment plans and patient outcomes, thereby propelling the IVD quality control market forward.
- Technological Advancements in IVD:Innovations in IVD technologies, including automation and molecular diagnostics, are transforming the landscape of healthcare in Oman. The Ministry of Health has allocated OMR 60 million for the adoption of cutting-edge diagnostic equipment in future. These advancements not only improve testing efficiency but also enhance the reliability of results, driving the demand for quality control solutions in the IVD sector.
Market Challenges
- High Costs of Advanced Diagnostic Equipment:The financial burden associated with acquiring advanced diagnostic equipment poses a significant challenge for healthcare providers in Oman. The average cost of high-end IVD systems can exceed OMR 120,000, which limits accessibility for smaller healthcare facilities. This financial barrier can hinder the widespread adoption of quality control measures, impacting overall diagnostic accuracy and patient care.
- Regulatory Hurdles in Product Approval:Navigating the regulatory landscape for IVD products in Oman can be complex and time-consuming. The approval process often requires extensive documentation and compliance with international standards, which can delay market entry for new technologies. This challenge is exacerbated by the limited regulatory resources available, potentially stifling innovation and slowing the growth of the IVD quality control market.
Oman In Vitro Diagnostics (IVD) Quality Control Market Future Outlook
The future of the IVD quality control market in Oman appears promising, driven by ongoing investments in healthcare infrastructure and technological advancements. As the government continues to prioritize healthcare improvements, the integration of artificial intelligence and point-of-care testing is expected to enhance diagnostic capabilities. Furthermore, the growing emphasis on personalized medicine will likely create new avenues for innovation, ensuring that the market remains dynamic and responsive to evolving healthcare needs.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Omani government is investing OMR 350 million in expanding healthcare facilities in future. This expansion will create opportunities for IVD quality control providers to supply advanced diagnostic solutions, enhancing overall healthcare delivery and patient outcomes.
- Growth in Personalized Medicine:The increasing focus on personalized medicine in Oman presents a significant opportunity for the IVD market. With an estimated OMR 120 million allocated for research and development in this area in future, companies can leverage this trend to develop tailored diagnostic solutions that meet specific patient needs.
 Quality Control Market.png)