Region:Middle East
Author(s):Shubham
Product Code:KRAA8602
Pages:87
Published On:November 2025
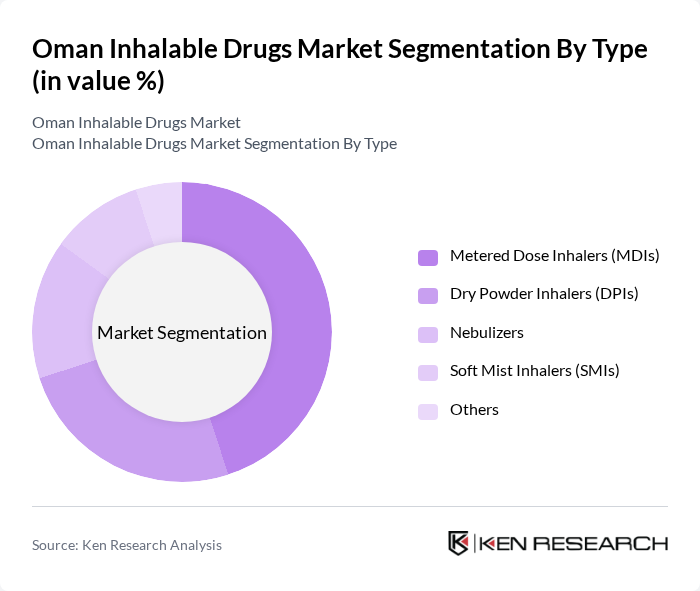
By Type:The inhalable drugs market is segmented into Metered Dose Inhalers (MDIs), Dry Powder Inhalers (DPIs), Nebulizers, Soft Mist Inhalers (SMIs), and Others. Metered Dose Inhalers (MDIs) remain the most widely used type due to their convenience, portability, and effectiveness in delivering medication directly to the lungs. The increasing incidence of asthma and COPD, coupled with patient preference for easy-to-use devices, continues to drive demand for MDIs, making them the dominant segment in the market.
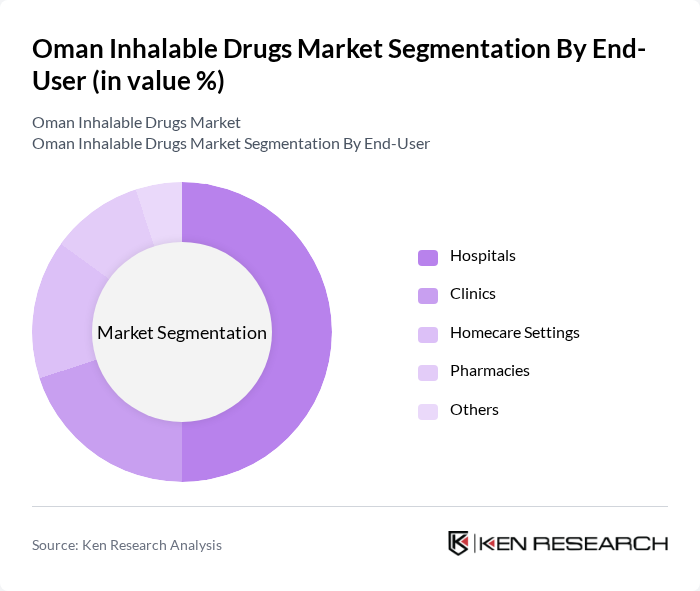
By End-User:The end-user segmentation includes Hospitals, Clinics, Homecare Settings, Pharmacies, and Others. Hospitals are the leading end-user segment, attributed to their comprehensive respiratory care facilities and the high volume of patients requiring inhalable therapies for acute and chronic respiratory conditions. The increasing number of hospital admissions for respiratory diseases and the availability of specialized respiratory care units have significantly contributed to the demand for inhalable drugs in this sector.
The Oman Inhalable Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as AstraZeneca, GlaxoSmithKline (GSK), Novartis, Boehringer Ingelheim, Teva Pharmaceutical Industries, Merck & Co., Sanofi, Viatris (formerly Mylan N.V.), Chiesi Farmaceutici, Hikma Pharmaceuticals, Cipla, Sandoz (a Novartis division), Sun Pharmaceutical Industries, Aurobindo Pharma, Alvogen, Oman Pharmaceutical Products Co. LLC, Gulf Pharmaceutical Industries (Julphar), Pharmax Pharmaceuticals contribute to innovation, geographic expansion, and service delivery in this space.
The future of the inhalable drugs market in Oman appears promising, driven by increasing healthcare investments and a growing focus on patient-centric solutions. As the government continues to enhance healthcare infrastructure, the accessibility of inhalable therapies is expected to improve significantly. Furthermore, the integration of digital health technologies will likely facilitate better patient monitoring and adherence, paving the way for innovative treatment approaches. Overall, these trends indicate a robust growth trajectory for the inhalable drugs market in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Metered Dose Inhalers (MDIs) Dry Powder Inhalers (DPIs) Nebulizers Soft Mist Inhalers (SMIs) Others |
| By End-User | Hospitals Clinics Homecare Settings Pharmacies Others |
| By Therapeutic Area | Asthma Chronic Obstructive Pulmonary Disease (COPD) Cystic Fibrosis Pulmonary Arterial Hypertension Acute Respiratory Distress Syndrome Others |
| By Distribution Channel | Retail Pharmacies Online Pharmacies Hospital Pharmacies Others |
| By Region | Muscat Dhofar Al Batinah Al Sharqiyah Others |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Policy Support | Subsidies for inhalable drug development Tax incentives for local manufacturers Grants for research initiatives Fast-track regulatory approvals Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Professionals | 100 | Pulmonologists, General Practitioners, Pharmacists |
| Pharmaceutical Distributors | 60 | Sales Managers, Distribution Heads |
| Patients Using Inhalable Drugs | 80 | Chronic Asthma Patients, COPD Patients |
| Regulatory Authorities | 40 | Health Policy Makers, Regulatory Affairs Officers |
| Market Analysts | 50 | Healthcare Market Researchers, Industry Analysts |
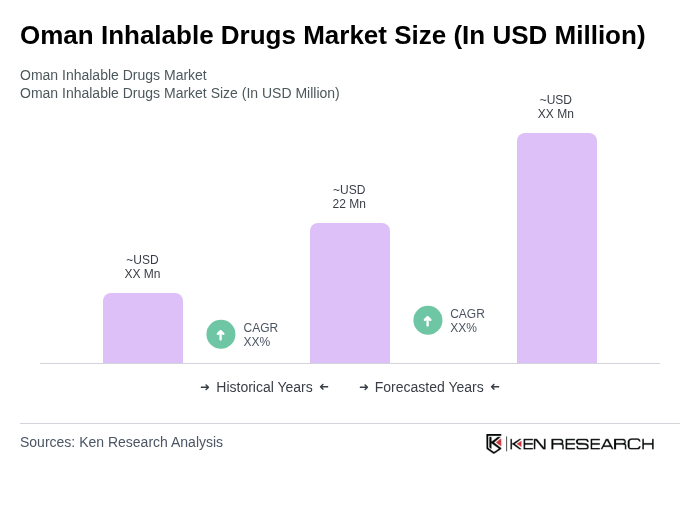
The Oman Inhalable Drugs Market is valued at approximately USD 22 million, reflecting a significant growth driven by the increasing prevalence of respiratory diseases, rising healthcare expenditure, and advancements in inhalation technology.