About the Report
Base Year 2024Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Overview
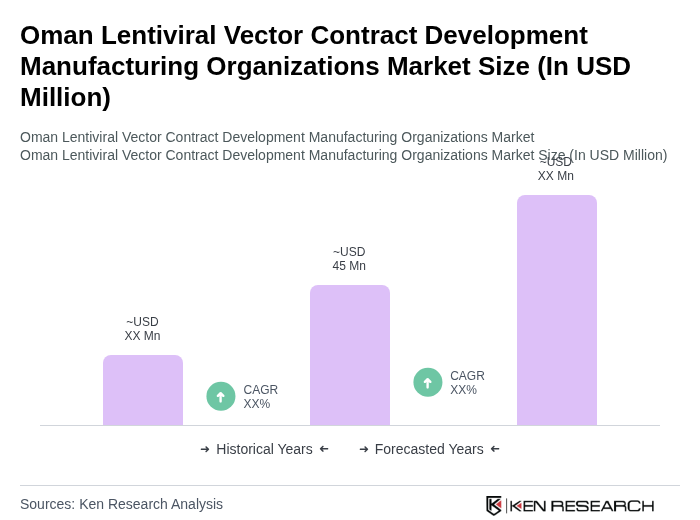
- The Oman Lentiviral Vector Contract Development Manufacturing Organizations Market is valued at USD 45 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for advanced gene therapies, rising prevalence of genetic disorders, expanding applications in CAR-T cell therapies, and increasing investment in personalized medicine, which necessitate the use of lentiviral vectors for effective treatment solutions.
- Key players in this market are concentrated in major cities such as Muscat and Salalah, where there is a robust healthcare infrastructure and a growing focus on biotechnology research. The presence of research institutions and pharmaceutical companies in these regions further enhances the market's growth potential.
- The Pharmaceutical and Biotechnology Regulatory Authority Rules, 2022 issued by the Ministry of Health requires licensing for biopharmaceutical manufacturing facilities, including compliance with Good Manufacturing Practices for gene therapy products, facility standards for viral vector production, and quality control thresholds for therapeutic vectors. This initiative aims to position Oman as a regional hub for advanced therapeutic solutions, fostering innovation and attracting foreign investment.
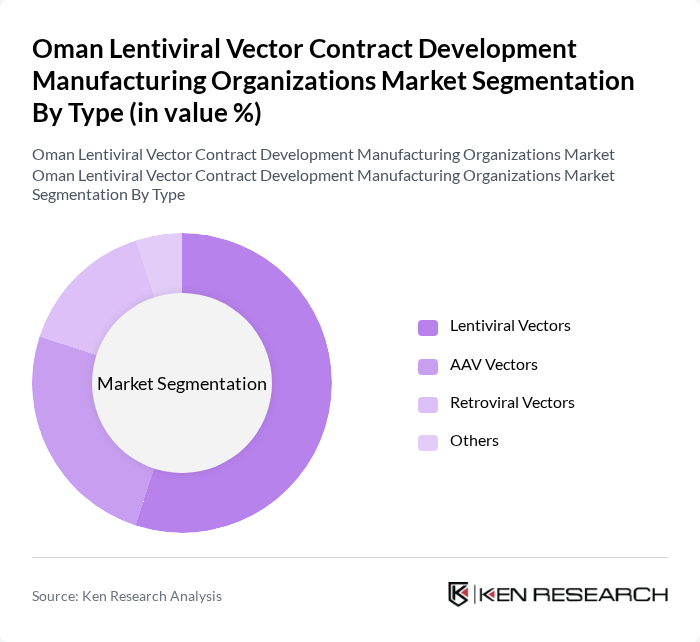
Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Segmentation
By Type:The market can be segmented into various types of vectors, including Lentiviral Vectors, AAV Vectors, Retroviral Vectors, and Others. Among these, Lentiviral Vectors are leading the market due to their high efficiency in gene delivery and ability to integrate into the host genome, making them suitable for long-term expression of therapeutic genes. AAV Vectors are also gaining traction, particularly in gene therapy applications, due to their safety profile and low immunogenicity. The demand for Retroviral Vectors remains steady, primarily in research settings.
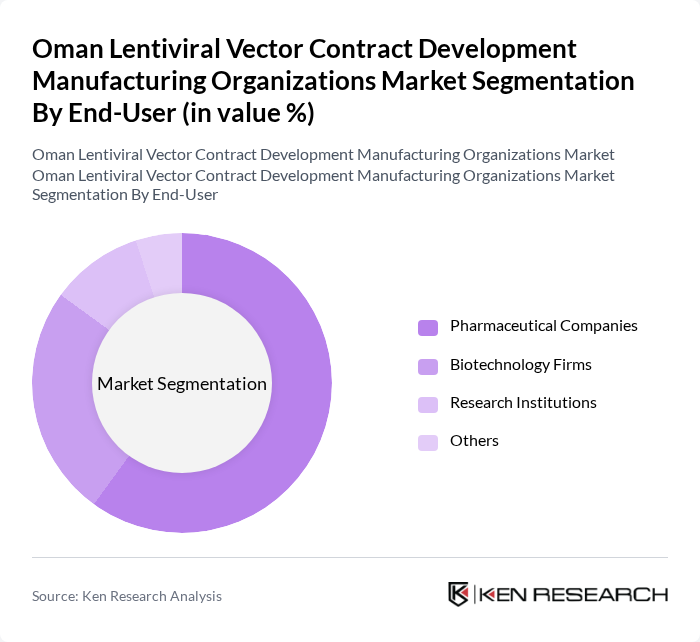
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Research Institutions, and Others. Pharmaceutical Companies dominate the market as they are increasingly investing in gene therapies and advanced biopharmaceuticals. Biotechnology Firms are also significant players, focusing on innovative solutions for genetic disorders. Research Institutions contribute to the market by conducting essential studies and trials, while other end-users include academic institutions and government laboratories.
Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Competitive Landscape
The Oman Lentiviral Vector Contract Development Manufacturing Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as Oxford Biomedica, Novartis, GSK, Merck KGaA, Catalent, AAVLife, Virovek, Genethon, Bluebird Bio, Cellectis, Sangamo Therapeutics, Amgen, Kite Pharma, Voyager Therapeutics, Synlogic contribute to innovation, geographic expansion, and service delivery in this space.
Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Industry Analysis
Growth Drivers
- Increasing Demand for Gene Therapies:The global gene therapy market is projected to reach $13.3 billion in future, driven by a surge in genetic disorders and chronic diseases. In Oman, the prevalence of genetic disorders is notably high, with approximately 20% of the population affected. This rising demand for innovative treatments is propelling the need for lentiviral vector manufacturing, as these vectors are crucial for delivering therapeutic genes effectively, thus fostering market growth.
- Advancements in Vector Technology:Technological innovations in vector design and production are enhancing the efficiency and safety of gene therapies. For instance, the development of self-inactivating lentiviral vectors has improved therapeutic outcomes while minimizing risks. In Oman, investments in biotechnology research have increased by 15% annually, indicating a robust environment for adopting these advancements, which is expected to significantly boost the local lentiviral vector market.
- Rising Investment in Biopharmaceuticals:The biopharmaceutical sector in Oman is experiencing substantial growth, with investments reaching $1.2 billion in future. This influx of capital is directed towards research and development of novel therapies, including gene therapies that utilize lentiviral vectors. The government's commitment to enhancing healthcare infrastructure and fostering innovation is further driving this investment, creating a favorable landscape for contract development manufacturing organizations in the region.
Market Challenges
- High Production Costs:The production of lentiviral vectors involves complex processes and high-quality materials, leading to significant operational costs. In Oman, the average cost of producing a single batch of lentiviral vectors can exceed $500,000. This financial burden poses a challenge for local manufacturers, limiting their ability to compete with established global players who benefit from economies of scale and advanced technologies.
- Regulatory Hurdles:Navigating the regulatory landscape for gene therapies can be daunting, with stringent requirements for clinical trials and product approvals. In Oman, the regulatory framework is still evolving, which can lead to delays in bringing new therapies to market. The average time for regulatory approval can extend beyond 18 months, creating uncertainty for manufacturers and potentially stifling innovation in the lentiviral vector sector.
Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Future Outlook
The future of the lentiviral vector market in Oman appears promising, driven by increasing collaborations between research institutions and biopharmaceutical companies. As the demand for personalized medicine grows, the focus on developing tailored gene therapies will likely intensify. Additionally, advancements in vector technology and manufacturing processes are expected to enhance production efficiency, making it more accessible for local organizations to participate in this burgeoning market, ultimately benefiting patient care and treatment outcomes.
Market Opportunities
- Expansion of Research Collaborations:Collaborative efforts between universities and biotech firms in Oman are on the rise, with funding for joint research projects increasing by 25% in future. This trend presents opportunities for contract development organizations to engage in innovative projects, enhancing their capabilities and market presence.
- Growth in Personalized Medicine:The personalized medicine market is expected to reach $2.5 billion in Oman in future. This growth is driven by advancements in genomics and patient-specific therapies, creating a demand for lentiviral vectors tailored to individual genetic profiles, thus opening new avenues for development and manufacturing organizations.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Lentiviral Vectors AAV Vectors Retroviral Vectors Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Research Institutions Others |
| By Application | Gene Therapy Vaccine Development Cell Therapy Others |
| By Delivery Method | In Vivo Ex Vivo Others |
| By Payload Capacity | Low Payload Medium Payload High Payload Others |
| By Region | Muscat Salalah Sohar Others |
| By Research Phase | Preclinical Clinical Trials Commercialization Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman)
Pharmaceutical and Biotechnology Companies
Contract Research Organizations (CROs)
Healthcare Providers and Hospitals
Biomanufacturing Facilities
Industry Associations (e.g., Oman Pharmaceutical Association)
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Oxford Biomedica
Novartis
GSK
Merck KGaA
Catalent
AAVLife
Virovek
Genethon
Bluebird Bio
Cellectis
Sangamo Therapeutics
Amgen
Kite Pharma
Voyager Therapeutics
Synlogic
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for gene therapies
3.1.2 Advancements in vector technology
3.1.3 Rising investment in biopharmaceuticals
3.1.4 Supportive government policies
3.2 Market Challenges
3.2.1 High production costs
3.2.2 Regulatory hurdles
3.2.3 Limited skilled workforce
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Expansion of research collaborations
3.3.2 Growth in personalized medicine
3.3.3 Increasing prevalence of genetic disorders
3.3.4 Development of novel delivery systems
3.4 Market Trends
3.4.1 Shift towards outsourcing manufacturing
3.4.2 Integration of AI in vector development
3.4.3 Focus on sustainable manufacturing practices
3.4.4 Rise in strategic partnerships and alliances
3.5 Government Regulation
3.5.1 Compliance with international standards
3.5.2 Approval processes for clinical trials
3.5.3 Intellectual property protections
3.5.4 Funding and grants for research initiatives
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Segmentation
8.1 By Type
8.1.1 Lentiviral Vectors
8.1.2 AAV Vectors
8.1.3 Retroviral Vectors
8.1.4 Others
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Research Institutions
8.2.4 Others
8.3 By Application
8.3.1 Gene Therapy
8.3.2 Vaccine Development
8.3.3 Cell Therapy
8.3.4 Others
8.4 By Delivery Method
8.4.1 In Vivo
8.4.2 Ex Vivo
8.4.3 Others
8.5 By Payload Capacity
8.5.1 Low Payload
8.5.2 Medium Payload
8.5.3 High Payload
8.5.4 Others
8.6 By Region
8.6.1 Muscat
8.6.2 Salalah
8.6.3 Sohar
8.6.4 Others
8.7 By Research Phase
8.7.1 Preclinical
8.7.2 Clinical Trials
8.7.3 Commercialization
8.7.4 Others
9. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Production Efficiency
9.2.8 R&D Investment Ratio
9.2.9 Regulatory Compliance Rate
9.2.10 Customer Satisfaction Index
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Oxford Biomedica
9.5.2 Novartis
9.5.3 GSK
9.5.4 Merck KGaA
9.5.5 Catalent
9.5.6 AAVLife
9.5.7 Virovek
9.5.8 Genethon
9.5.9 Bluebird Bio
9.5.10 Cellectis
9.5.11 Sangamo Therapeutics
9.5.12 Amgen
9.5.13 Kite Pharma
9.5.14 Voyager Therapeutics
9.5.15 Synlogic
10. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Funding Sources
10.2.3 Project Management Approaches
10.2.4 Cost-Benefit Analysis
10.3 Pain Point Analysis by End-User Category
10.3.1 Operational Inefficiencies
10.3.2 Compliance Challenges
10.3.3 Technology Gaps
10.3.4 Budget Constraints
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Infrastructure Readiness
10.4.3 Change Management Strategies
10.4.4 Support Systems
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Scalability Potential
10.5.3 User Feedback Mechanisms
10.5.4 Future Investment Plans
11. Oman Lentiviral Vector Contract Development Manufacturing Organizations Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Competitive Advantage Assessment
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from biotechnology and pharmaceutical associations in Oman
- Review of scientific publications and patents related to lentiviral vector technologies
- Examination of market trends and forecasts from global health organizations and regulatory bodies
Primary Research
- Interviews with executives from contract development manufacturing organizations (CDMOs) specializing in lentiviral vectors
- Surveys targeting researchers and developers in gene therapy and regenerative medicine sectors
- Field interviews with regulatory affairs specialists to understand compliance challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of quantitative data with qualitative insights from industry experts
- Sanity checks conducted through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall biopharmaceutical market size in Oman as a basis for lentiviral vector segment
- Segmentation of the market by application areas such as oncology, genetic disorders, and infectious diseases
- Incorporation of government funding and initiatives supporting gene therapy research
Bottom-up Modeling
- Collection of data on production capacities and service offerings from leading CDMOs in Oman
- Operational cost analysis based on pricing models of lentiviral vector production
- Volume estimates derived from clinical trial demands and commercial production needs
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as healthcare expenditure and technological advancements
- Scenario modeling based on potential regulatory changes and market entry of new therapies
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Research Institutions | 120 | Research Scientists, Lab Directors |
| Contract Development Manufacturing Organizations | 100 | Operations Managers, Business Development Executives |
| Regulatory Bodies and Compliance Experts | 50 | Regulatory Affairs Managers, Compliance Officers |
| Healthcare Providers and Clinical Trial Coordinators | 80 | Clinical Researchers, Medical Directors |
| Investors and Venture Capitalists in Biotech | 60 | Investment Analysts, Portfolio Managers |
Frequently Asked Questions
What is the current value of the Oman Lentiviral Vector Contract Development Manufacturing Organizations Market?
The Oman Lentiviral Vector Contract Development Manufacturing Organizations Market is valued at approximately USD 45 million, reflecting a growing demand for advanced gene therapies and personalized medicine solutions in the region.