About the Report
Base Year 2024Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Overview
- The Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) market is valued at USD 135 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of PNH, rapid modernization of healthcare infrastructure, and the introduction of advanced biologic therapies. The adoption of novel complement inhibitors and the expansion of tertiary care centers have significantly improved patient outcomes, further propelling market growth. Rising awareness among healthcare professionals and patients, as well as the integration of rare disease management into national health priorities, are additional growth drivers.
- Key players in this market include Muscat, Salalah, and Sohar, which dominate due to their advanced healthcare infrastructure and specialized medical facilities. These cities have established centers for hematology and rare diseases, attracting patients from across the region and facilitating access to cutting-edge treatments and clinical trials. Recent expansions in hospital capacity and the establishment of new specialty centers have further consolidated their leadership in rare disease care.
- In 2023, the Omani government implemented the National Health Strategy for Rare Diseases, issued by the Ministry of Health, Oman. This initiative includes dedicated funding for rare disease research, mandates for early diagnosis protocols, and the expansion of specialized treatment centers. The strategy also supports awareness campaigns and ensures streamlined access to approved therapies for PNH and other rare hematological disorders.
 Market.png)
Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Segmentation
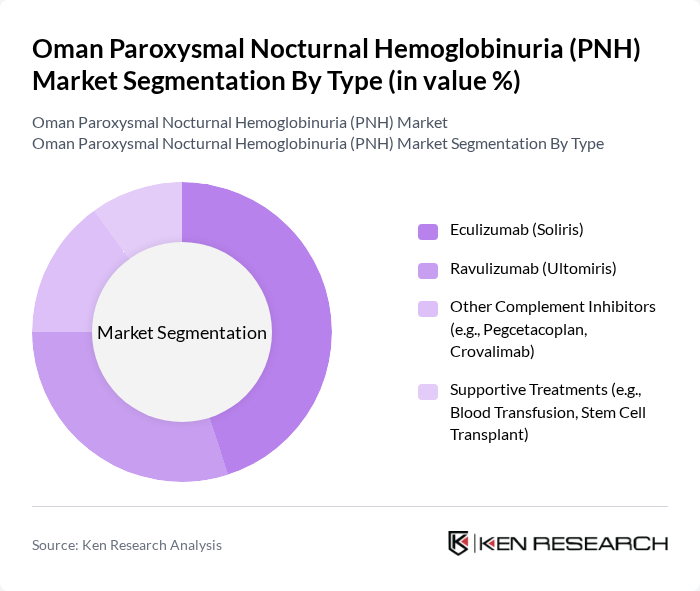
By Type:The market is segmented into various types of treatments available for PNH, including Eculizumab (Soliris), Ravulizumab (Ultomiris), other complement inhibitors, and supportive treatments. Among these, Eculizumab (Soliris) remains the leading treatment option due to its established efficacy and safety profile, making it the preferred choice for healthcare providers. The introduction of Ravulizumab (Ultomiris) and other next-generation complement inhibitors has expanded therapeutic options, especially for patients requiring longer dosing intervals or with refractory disease. The increasing number of patients diagnosed with PNH and the availability of advanced biologics have further solidified the market position of complement inhibitors.
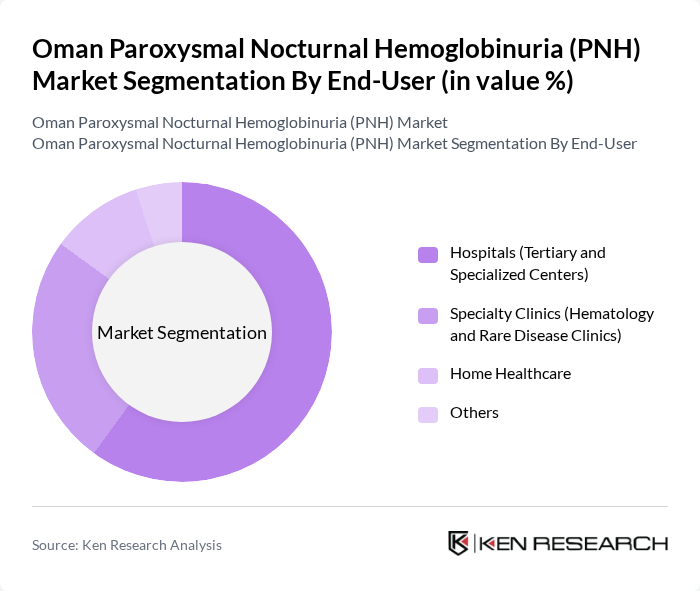
By End-User:The end-user segmentation includes hospitals, specialty clinics, home healthcare, and others. Hospitals, particularly tertiary and specialized centers, dominate the market due to their capacity to provide comprehensive care and access to advanced treatment options. The increasing number of patients seeking treatment in these facilities has led to a higher market share for hospitals compared to other end-users. Specialty clinics focused on hematology and rare diseases are also expanding, supported by government and private investment in rare disease infrastructure.
Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Competitive Landscape
The Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market is characterized by a dynamic mix of regional and international players. Leading participants such as Alexion Pharmaceuticals (AstraZeneca Rare Disease), Novartis AG, Roche Holding AG, Sanofi S.A., Apellis Pharmaceuticals, Amgen Inc., Pfizer Inc., GlaxoSmithKline plc (GSK), Takeda Pharmaceutical Company Limited, Sobi (Swedish Orphan Biovitrum AB), Regeneron Pharmaceuticals, Inc., AbbVie Inc., Johnson & Johnson (Janssen Pharmaceuticals), CSL Behring, Chugai Pharmaceutical Co., Ltd. (a member of the Roche Group) contribute to innovation, geographic expansion, and service delivery in this space.
Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Industry Analysis
Growth Drivers
- Increasing Prevalence of PNH:The prevalence of Paroxysmal Nocturnal Hemoglobinuria (PNH) in Oman is estimated to be around 1.5 cases per 100,000 individuals, translating to approximately 50 cases annually based on a population of 4.5 million. This rising incidence is driving demand for effective treatments and healthcare services, as more patients seek diagnosis and management options. The growing patient population is expected to significantly influence market dynamics, leading to increased investment in healthcare infrastructure and treatment modalities.
- Advancements in Treatment Options:Recent advancements in PNH treatment, including the introduction of complement inhibitors like eculizumab, have transformed patient outcomes. In Oman, the healthcare sector has seen a 30% increase in the availability of these therapies since the last few years. This progress not only enhances patient survival rates but also encourages healthcare providers to adopt innovative treatment protocols, thereby expanding the market for PNH therapies and improving overall patient care.
- Rising Awareness and Diagnosis Rates:Increased awareness of PNH among healthcare professionals and the general public has led to a significant rise in diagnosis rates. In Oman, diagnostic testing for PNH has increased by 40% over the past few years, with more patients being accurately identified and treated. This trend is supported by educational initiatives and patient advocacy groups, which are crucial in promoting early detection and timely intervention, ultimately driving market growth.
Market Challenges
- High Cost of Treatment:The cost of PNH treatments, particularly novel therapies, can exceed OMR 30,000 annually per patient. This high financial burden poses a significant challenge for many patients in Oman, where healthcare expenditure per capita is approximately OMR 1,500. Limited insurance coverage for rare diseases further exacerbates the issue, leading to treatment delays and reduced access to necessary therapies, which can hinder market growth.
- Limited Healthcare Infrastructure:Oman faces challenges in healthcare infrastructure, particularly in rural areas where specialized care for PNH is scarce. With only 1.5 hematologists per 100,000 people, access to expert care is limited. This shortage affects timely diagnosis and treatment, contributing to poorer health outcomes for PNH patients. The need for enhanced healthcare facilities and trained professionals is critical to overcoming this challenge and improving patient care.
Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Future Outlook
The future of the PNH market in Oman appears promising, driven by ongoing advancements in treatment options and increased awareness among healthcare providers. As the healthcare system evolves, the integration of telemedicine and digital health technologies is expected to enhance patient access to specialized care. Furthermore, the government’s commitment to improving healthcare infrastructure will likely facilitate better diagnosis and treatment, ultimately benefiting PNH patients and fostering market growth in the future.
Market Opportunities
- Potential for New Drug Development:The ongoing research into novel therapies for PNH presents significant opportunities for pharmaceutical companies. With an estimated 20% of patients remaining untreated, the demand for innovative solutions is high. This gap in treatment options encourages investment in drug development, which could lead to breakthroughs that improve patient outcomes and expand market share.
- Collaborations with International Pharmaceutical Companies:Partnerships with global pharmaceutical firms can enhance local capabilities in drug manufacturing and distribution. Such collaborations can facilitate knowledge transfer and access to advanced therapies, addressing the treatment gap in Oman. By leveraging international expertise, local companies can improve their product offerings and contribute to the overall growth of the PNH market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Eculizumab (Soliris) Ravulizumab (Ultomiris) Other Complement Inhibitors (e.g., Pegcetacoplan, Crovalimab) Supportive Treatments (e.g., Blood Transfusion, Stem Cell Transplant) |
| By End-User | Hospitals (Tertiary and Specialized Centers) Specialty Clinics (Hematology and Rare Disease Clinics) Home Healthcare Others |
| By Patient Demographics | Age Group (Children, Adults, Elderly) Gender Socioeconomic Status Others |
| By Distribution Channel | Hospital Pharmacies Retail Pharmacies Online Pharmacies Others |
| By Treatment Setting | Inpatient Outpatient Home Care Others |
| By Geographic Distribution | Muscat Salalah Sohar Others |
| By Research and Development Focus | Clinical Trials Post-Marketing Studies Drug Repurposing Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman)
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Hospitals
Clinical Research Organizations
Health Insurance Companies
Patient Advocacy Groups
Players Mentioned in the Report:
Alexion Pharmaceuticals (AstraZeneca Rare Disease)
Novartis AG
Roche Holding AG
Sanofi S.A.
Apellis Pharmaceuticals
Amgen Inc.
Pfizer Inc.
GlaxoSmithKline plc (GSK)
Takeda Pharmaceutical Company Limited
Sobi (Swedish Orphan Biovitrum AB)
Regeneron Pharmaceuticals, Inc.
AbbVie Inc.
Johnson & Johnson (Janssen Pharmaceuticals)
CSL Behring
Chugai Pharmaceutical Co., Ltd. (a member of the Roche Group)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of PNH in Oman
3.1.2 Advancements in treatment options
3.1.3 Rising awareness and diagnosis rates
3.1.4 Supportive government policies for rare diseases
3.2 Market Challenges
3.2.1 High cost of treatment
3.2.2 Limited healthcare infrastructure
3.2.3 Lack of specialized healthcare professionals
3.2.4 Regulatory hurdles in drug approval
3.3 Market Opportunities
3.3.1 Potential for new drug development
3.3.2 Collaborations with international pharmaceutical companies
3.3.3 Expansion of healthcare facilities
3.3.4 Increased funding for rare disease research
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Growth in telemedicine services
3.4.3 Rising patient advocacy and support groups
3.4.4 Integration of digital health technologies
3.5 Government Regulation
3.5.1 Regulatory frameworks for rare diseases
3.5.2 Pricing regulations for pharmaceuticals
3.5.3 Approval processes for new treatments
3.5.4 Incentives for local drug manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Segmentation
8.1 By Type
8.1.1 Eculizumab (Soliris)
8.1.2 Ravulizumab (Ultomiris)
8.1.3 Other Complement Inhibitors (e.g., Pegcetacoplan, Crovalimab)
8.1.4 Supportive Treatments (e.g., Blood Transfusion, Stem Cell Transplant)
8.2 By End-User
8.2.1 Hospitals (Tertiary and Specialized Centers)
8.2.2 Specialty Clinics (Hematology and Rare Disease Clinics)
8.2.3 Home Healthcare
8.2.4 Others
8.3 By Patient Demographics
8.3.1 Age Group (Children, Adults, Elderly)
8.3.2 Gender
8.3.3 Socioeconomic Status
8.3.4 Others
8.4 By Distribution Channel
8.4.1 Hospital Pharmacies
8.4.2 Retail Pharmacies
8.4.3 Online Pharmacies
8.4.4 Others
8.5 By Treatment Setting
8.5.1 Inpatient
8.5.2 Outpatient
8.5.3 Home Care
8.5.4 Others
8.6 By Geographic Distribution
8.6.1 Muscat
8.6.2 Salalah
8.6.3 Sohar
8.6.4 Others
8.7 By Research and Development Focus
8.7.1 Clinical Trials
8.7.2 Post-Marketing Studies
8.7.3 Drug Repurposing
8.7.4 Others
9. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 PNH Portfolio Breadth (Number of Approved and Pipeline Products)
9.2.4 Revenue from PNH Products (USD, Oman and Global)
9.2.5 Market Penetration Rate in Oman (Share of Treated PNH Patients)
9.2.6 Patient Access Programs (Availability and Scope)
9.2.7 Clinical Trial Activity in Oman and GCC
9.2.8 Regulatory Approval Timelines (Oman and GCC)
9.2.9 Distribution Network Strength (Hospital and Pharmacy Coverage)
9.2.10 Local Partnerships and Licensing Agreements
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Alexion Pharmaceuticals (AstraZeneca Rare Disease)
9.5.2 Novartis AG
9.5.3 Roche Holding AG
9.5.4 Sanofi S.A.
9.5.5 Apellis Pharmaceuticals
9.5.6 Amgen Inc.
9.5.7 Pfizer Inc.
9.5.8 GlaxoSmithKline plc (GSK)
9.5.9 Takeda Pharmaceutical Company Limited
9.5.10 Sobi (Swedish Orphan Biovitrum AB)
9.5.11 Regeneron Pharmaceuticals, Inc.
9.5.12 AbbVie Inc.
9.5.13 Johnson & Johnson (Janssen Pharmaceuticals)
9.5.14 CSL Behring
9.5.15 Chugai Pharmaceutical Co., Ltd. (a member of the Roche Group)
10. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Finance
10.1.3 Ministry of Social Development
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Healthcare Infrastructure Investments
10.2.2 Research Funding
10.2.3 Treatment Accessibility Initiatives
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Patients
10.3.2 Healthcare Providers
10.3.3 Insurance Companies
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Accessibility of Treatments
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Treatment Efficacy
10.5.2 Patient Satisfaction
10.5.3 Long-term Health Outcomes
10.5.4 Others
11. Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published reports from health organizations and medical journals on PNH prevalence in Oman
- Review of government health statistics and epidemiological studies related to blood disorders
- Examination of pharmaceutical market reports focusing on hematology and rare diseases
Primary Research
- Interviews with hematologists and specialists treating PNH patients in Oman
- Surveys conducted with healthcare providers regarding treatment protocols and patient demographics
- Focus groups with PNH patient advocacy groups to understand patient experiences and treatment access
Validation & Triangulation
- Cross-validation of findings with multiple healthcare professionals and institutions
- Triangulation of data from clinical studies, patient registries, and market analysis
- Sanity checks through expert panel discussions to ensure data reliability
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on the prevalence of PNH in Oman
- Segmentation of the market by treatment type, including supportive care and novel therapies
- Incorporation of healthcare expenditure trends and government health initiatives
Bottom-up Modeling
- Collection of data on treatment costs from local hospitals and clinics
- Estimation of patient population growth based on demographic trends and disease incidence
- Volume x cost analysis for each treatment modality available in the market
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering factors such as healthcare access and treatment advancements
- Scenario modeling based on potential changes in healthcare policy and drug approvals
- Baseline, optimistic, and pessimistic forecasts for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hematology Clinics | 100 | Hematologists, Clinic Administrators |
| Patient Advocacy Groups | 50 | Patient Representatives, Support Group Leaders |
| Pharmaceutical Distributors | 40 | Distribution Managers, Sales Representatives |
| Healthcare Policy Makers | 40 | Health Ministry Officials, Policy Analysts |
| Research Institutions | 40 | Clinical Researchers, Epidemiologists |
Frequently Asked Questions
What is the current value of the Oman Paroxysmal Nocturnal Hemoglobinuria (PNH) market?
The Oman PNH market is valued at approximately USD 135 million, reflecting a five-year historical analysis. This growth is attributed to the increasing prevalence of PNH and advancements in healthcare infrastructure and treatment options.