About the Report
Base Year 2024Oman Vaccine Contract Manufacturing Market Overview
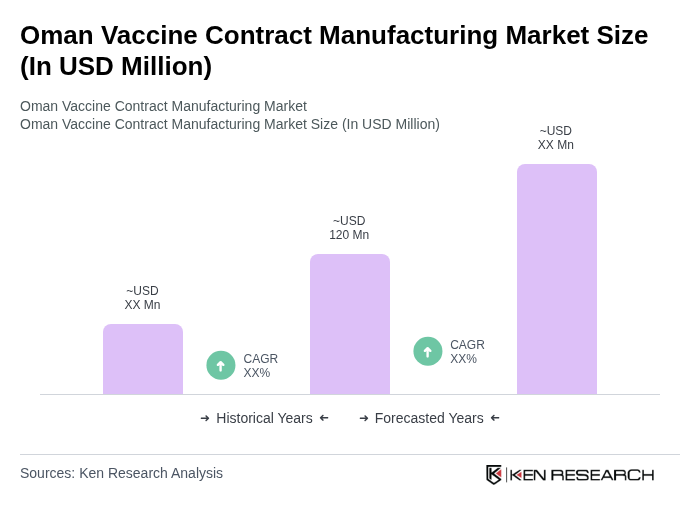
- The Oman Vaccine Contract Manufacturing Market is valued at USD 120 million, based on a five-year historical analysis. This market size reflects the segment's share within the broader Oman pharmaceutical contract manufacturing sector, which is experiencing steady growth due to increased public health initiatives, rising demand for vaccines amid infectious disease outbreaks, and the government's strategic efforts to enhance local manufacturing capabilities for vaccine security and self-sufficiency. These drivers align with global trends, where governments and healthcare organizations are prioritizing immunization programs and investing in scalable manufacturing infrastructure to ensure reliable vaccine supply .
- Key players in this market are concentrated in Muscat and Dhofar, where the infrastructure for pharmaceutical manufacturing is more advanced. These regions benefit from superior logistics networks, a skilled workforce, and robust government support, making them attractive hubs for both domestic and international vaccine manufacturers. The clustering of manufacturing operations in these areas facilitates efficient supply chain management and accelerates technology transfer, supporting rapid response to public health needs .
- In 2023, the Omani government enacted the “Regulation on Vaccine Manufacturing and Quality Standards, 2023” issued by the Ministry of Health. This regulation mandates that all vaccines produced within Oman must comply with international quality standards, including WHO prequalification. The regulation specifies operational requirements for manufacturing facilities, product testing protocols, and documentation standards, thereby enhancing the safety and efficacy of vaccines available in the local market and strengthening public confidence in immunization programs .
Oman Vaccine Contract Manufacturing Market Segmentation
By Type:The market is segmented into Live Attenuated Vaccines, Inactivated Vaccines, Subunit Vaccines, mRNA Vaccines, Viral Vector Vaccines, Protein-based Vaccines, and Others. Each type addresses distinct immunization needs and involves unique production technologies. Live attenuated vaccines utilize weakened pathogens to stimulate robust immune responses, while inactivated vaccines use killed organisms for safer profiles. Subunit and protein-based vaccines focus on specific antigens, mRNA and viral vector vaccines leverage genetic engineering for rapid development, and the “Others” category includes emerging platforms such as DNA and conjugate vaccines .
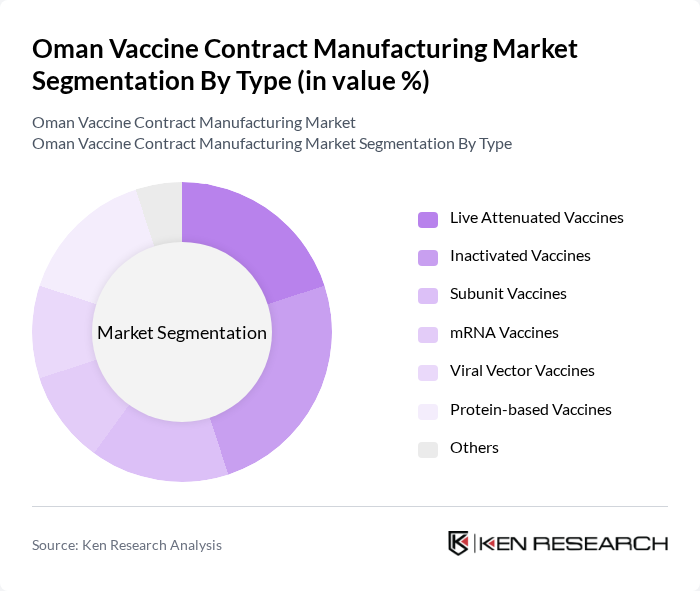
The Live Attenuated Vaccines segment maintains a leading position due to its proven effectiveness in generating long-term immunity and its established role in routine vaccination programs. The persistent prevalence of infectious diseases and the necessity for rapid outbreak response have further increased demand for these vaccines. Additionally, ongoing advancements in bioprocessing and cell culture technologies have enabled large-scale, cost-effective production, reinforcing the segment’s market leadership .
By End-User:The market is segmented by end-users, including Government Health Programs, Private Healthcare Providers, Non-Governmental Organizations (NGOs), Research Institutions, Pharmaceutical Companies, and Others. Government Health Programs are the primary purchasers and distributors, leveraging national immunization campaigns and international partnerships to drive vaccine uptake. Private Healthcare Providers supplement public efforts by offering immunization services, while NGOs focus on outreach and coverage in underserved communities. Research Institutions and pharmaceutical companies contribute to innovation and clinical trials, and the “Others” segment encompasses specialty clinics and occupational health providers .
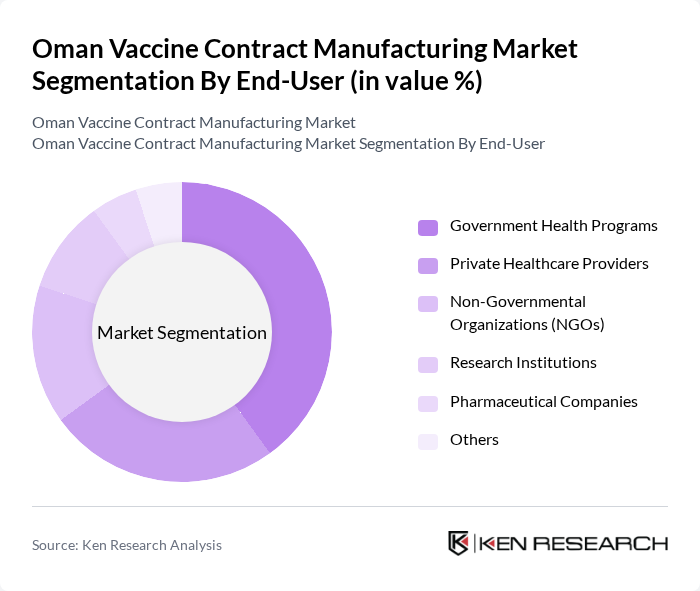
Government Health Programs dominate the end-user landscape, accounting for the largest market share. This leadership is driven by comprehensive national vaccination initiatives, substantial budget allocations for immunization, and strategic collaborations with global health organizations. These programs ensure broad population coverage and sustained demand for vaccine contract manufacturing services .
Oman Vaccine Contract Manufacturing Market Competitive Landscape
The Oman Vaccine Contract Manufacturing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Opal Bio Pharma (OBP), Philex Pharmaceuticals, Menagen Pharma, Dhofar Pharma, Izz Pharma, Health Supplies Factory, Technology Medical and Educational Supplies Company, Lonza Group Ltd., Catalent, Inc., Thermo Fisher Scientific Inc., Samsung Biologics Co., Ltd., FUJIFILM Diosynth Biotechnologies U.S.A., Inc., WuXi Biologics (Cayman) Inc., AGC Biologics, Inc., Recipharm AB contribute to innovation, geographic expansion, and service delivery in this space.
Oman Vaccine Contract Manufacturing Market Industry Analysis
Growth Drivers
- Increasing Demand for Vaccines:The demand for vaccines in Oman is significantly driven by public health initiatives, with the Ministry of Health allocating approximately OMR 50 million in future for vaccination programs. This investment aims to enhance immunization coverage, targeting a 90% vaccination rate among children. The rising awareness of vaccine-preventable diseases has led to increased public participation in vaccination campaigns, further propelling the need for local vaccine manufacturing capabilities.
- Government Investments in Healthcare Infrastructure:The Omani government has committed to investing OMR 1.2 billion in healthcare infrastructure in future, focusing on enhancing local manufacturing capabilities. This investment includes the establishment of new facilities and upgrading existing ones to meet international standards. Such initiatives are expected to create a conducive environment for vaccine contract manufacturing, ultimately reducing dependency on imports and improving local production efficiency.
- Rising Prevalence of Infectious Diseases:Oman has witnessed a notable increase in infectious diseases, with reported cases of measles rising by 30% in future. This trend has heightened the urgency for effective vaccination strategies. The World Health Organization (WHO) has identified Oman as a priority area for vaccine development, prompting local manufacturers to scale up production to address these public health challenges effectively and timely.
Market Challenges
- Limited Local Manufacturing Capabilities:Despite the growing demand for vaccines, Oman faces challenges due to limited local manufacturing capabilities. Currently, only 20% of vaccines administered are produced domestically, with the remainder imported. This reliance on foreign suppliers poses risks related to supply chain disruptions and delays, particularly during global health crises, highlighting the need for enhanced local production facilities and expertise.
- Stringent Regulatory Requirements:The vaccine manufacturing sector in Oman is subject to stringent regulatory requirements, which can hinder the speed of market entry for new products. Compliance with WHO standards and local regulations necessitates extensive documentation and testing, often extending timelines by 12-18 months. This regulatory landscape can deter potential investors and slow down the growth of local manufacturing capabilities, impacting overall market dynamics.
Oman Vaccine Contract Manufacturing Market Future Outlook
The future of the Oman vaccine contract manufacturing market appears promising, driven by increasing government support and a growing emphasis on local production. As the healthcare sector evolves, advancements in biopharmaceutical technologies and a focus on personalized medicine are expected to reshape the landscape. Additionally, strategic partnerships with global pharmaceutical companies will likely enhance local capabilities, fostering innovation and improving vaccine accessibility for the Omani population.
Market Opportunities
- Expansion of Vaccine Production Facilities:There is a significant opportunity for expanding vaccine production facilities in Oman, with the government planning to establish three new manufacturing plants in future. This expansion is projected to create over 500 jobs and enhance local production capacity, reducing reliance on imports and ensuring timely vaccine availability during health emergencies.
- Development of New Vaccine Technologies:The ongoing research and development in vaccine technologies present a lucrative opportunity for local manufacturers. With an estimated investment of OMR 30 million in R&D in future, companies can innovate and develop vaccines tailored to regional health needs, potentially leading to breakthroughs in combating endemic diseases prevalent in Oman.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Live Attenuated Vaccines Inactivated Vaccines Subunit Vaccines mRNA Vaccines Viral Vector Vaccines Protein-based Vaccines Others |
| By End-User | Government Health Programs Private Healthcare Providers Non-Governmental Organizations (NGOs) Research Institutions Pharmaceutical Companies Others |
| By Distribution Channel | Direct Sales Distributors Online Platforms Hospital Pharmacies Others |
| By Manufacturing Process | Cell Culture Egg-based Production Recombinant DNA Technology mRNA Synthesis Others |
| By Target Disease | Infectious Diseases Autoimmune Diseases Cancer Vaccines Zoonotic Diseases Others |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Al Sharqiyah Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Local Procurement Mandates Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Oman Pharmaceutical Association)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Manufacturing Organizations (CMOs)
Healthcare Providers and Hospitals
Supply Chain and Logistics Companies
Pharmaceutical Distributors
Players Mentioned in the Report:
Opal Bio Pharma (OBP)
Philex Pharmaceuticals
Menagen Pharma
Dhofar Pharma
Izz Pharma
Health Supplies Factory
Technology Medical and Educational Supplies Company
Lonza Group Ltd.
Catalent, Inc.
Thermo Fisher Scientific Inc.
Samsung Biologics Co., Ltd.
FUJIFILM Diosynth Biotechnologies U.S.A., Inc.
WuXi Biologics (Cayman) Inc.
AGC Biologics, Inc.
Recipharm AB
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Oman Vaccine Contract Manufacturing Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Oman Vaccine Contract Manufacturing Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Oman Vaccine Contract Manufacturing Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for vaccines due to public health initiatives
3.1.2 Government investments in healthcare infrastructure
3.1.3 Rising prevalence of infectious diseases
3.1.4 Strategic partnerships with global pharmaceutical companies
3.2 Market Challenges
3.2.1 Limited local manufacturing capabilities
3.2.2 Stringent regulatory requirements
3.2.3 High initial capital investment
3.2.4 Competition from established international manufacturers
3.3 Market Opportunities
3.3.1 Expansion of vaccine production facilities
3.3.2 Development of new vaccine technologies
3.3.3 Increased focus on biopharmaceuticals
3.3.4 Growing demand for personalized medicine
3.4 Market Trends
3.4.1 Shift towards contract manufacturing partnerships
3.4.2 Adoption of advanced manufacturing technologies
3.4.3 Emphasis on sustainability in production processes
3.4.4 Rising consumer awareness about vaccine safety
3.5 Government Regulation
3.5.1 Compliance with WHO standards
3.5.2 Local content requirements for manufacturing
3.5.3 Licensing and approval processes for new vaccines
3.5.4 Incentives for local manufacturers
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Oman Vaccine Contract Manufacturing Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Oman Vaccine Contract Manufacturing Market Segmentation
8.1 By Type
8.1.1 Live Attenuated Vaccines
8.1.2 Inactivated Vaccines
8.1.3 Subunit Vaccines
8.1.4 mRNA Vaccines
8.1.5 Viral Vector Vaccines
8.1.6 Protein-based Vaccines
8.1.7 Others
8.2 By End-User
8.2.1 Government Health Programs
8.2.2 Private Healthcare Providers
8.2.3 Non-Governmental Organizations (NGOs)
8.2.4 Research Institutions
8.2.5 Pharmaceutical Companies
8.2.6 Others
8.3 By Distribution Channel
8.3.1 Direct Sales
8.3.2 Distributors
8.3.3 Online Platforms
8.3.4 Hospital Pharmacies
8.3.5 Others
8.4 By Manufacturing Process
8.4.1 Cell Culture
8.4.2 Egg-based Production
8.4.3 Recombinant DNA Technology
8.4.4 mRNA Synthesis
8.4.5 Others
8.5 By Target Disease
8.5.1 Infectious Diseases
8.5.2 Autoimmune Diseases
8.5.3 Cancer Vaccines
8.5.4 Zoonotic Diseases
8.5.5 Others
8.6 By Region
8.6.1 Muscat
8.6.2 Dhofar
8.6.3 Al Batinah
8.6.4 Al Dakhiliyah
8.6.5 Al Sharqiyah
8.6.6 Others
8.7 By Policy Support
8.7.1 Government Subsidies
8.7.2 Tax Incentives
8.7.3 Research Grants
8.7.4 Local Procurement Mandates
8.7.5 Others
9. Oman Vaccine Contract Manufacturing Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Annual Vaccine Production Capacity (doses/year)
9.2.4 Number of Vaccine Types Manufactured
9.2.5 Market Penetration Rate in Oman (%)
9.2.6 Regulatory Compliance (e.g., WHO prequalification, Omani MoH approvals)
9.2.7 R&D Investment as % of Revenue
9.2.8 Time-to-Market for New Vaccines (months)
9.2.9 Supply Chain Reliability (On-Time Delivery %)
9.2.10 Export Share of Production (%)
9.2.11 Product Diversification (number of therapeutic areas)
9.2.12 Local Employment Contribution (number of jobs created)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Opal Bio Pharma (OBP)
9.5.2 Philex Pharmaceuticals
9.5.3 Menagen Pharma
9.5.4 Dhofar Pharma
9.5.5 Izz Pharma
9.5.6 Health Supplies Factory
9.5.7 Technology Medical and Educational Supplies Company
9.5.8 Lonza Group Ltd.
9.5.9 Catalent, Inc.
9.5.10 Thermo Fisher Scientific Inc.
9.5.11 Samsung Biologics Co., Ltd.
9.5.12 FUJIFILM Diosynth Biotechnologies U.S.A., Inc.
9.5.13 WuXi Biologics (Cayman) Inc.
9.5.14 AGC Biologics, Inc.
9.5.15 Recipharm AB
10. Oman Vaccine Contract Manufacturing Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Vaccine procurement processes
10.1.2 Budget allocation for vaccine programs
10.1.3 Collaboration with international organizations
10.1.4 Evaluation criteria for suppliers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in manufacturing facilities
10.2.2 Funding for research and development
10.2.3 Expenditure on technology upgrades
10.2.4 Partnerships with local businesses
10.3 Pain Point Analysis by End-User Category
10.3.1 Supply chain disruptions
10.3.2 Quality assurance challenges
10.3.3 Regulatory compliance issues
10.3.4 Cost management concerns
10.4 User Readiness for Adoption
10.4.1 Training and education needs
10.4.2 Infrastructure readiness
10.4.3 Stakeholder engagement
10.4.4 Technology adoption barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of vaccine effectiveness
10.5.2 Expansion into new markets
10.5.3 Long-term sustainability assessments
10.5.4 Feedback mechanisms for continuous improvement
11. Oman Vaccine Contract Manufacturing Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market gaps identification
1.2 Value proposition development
1.3 Revenue stream analysis
1.4 Cost structure evaluation
1.5 Key partnerships and resources
1.6 Customer segments targeting
1.7 Channels for delivery
2. Marketing and Positioning Recommendations
2.1 Branding strategies
2.2 Product USPs
2.3 Target audience engagement
2.4 Digital marketing initiatives
2.5 Public relations strategies
2.6 Market penetration tactics
2.7 Customer feedback integration
3. Distribution Plan
3.1 Urban retail vs rural NGO tie-ups
3.2 Logistics and supply chain management
3.3 Distribution channel optimization
3.4 Partnerships with local distributors
3.5 Inventory management strategies
3.6 Customer service enhancements
3.7 Performance metrics for distribution
4. Channel & Pricing Gaps
4.1 Underserved routes
4.2 Pricing bands
4.3 Competitive pricing analysis
4.4 Value-based pricing strategies
4.5 Discounts and promotions
4.6 Customer segmentation for pricing
4.7 Price elasticity considerations
5. Unmet Demand & Latent Needs
5.1 Category gaps
5.2 Consumer segments
5.3 Emerging trends analysis
5.4 Future demand forecasting
5.5 Product development opportunities
5.6 Market entry barriers
5.7 Stakeholder needs assessment
6. Customer Relationship
6.1 Loyalty programs
6.2 After-sales service
6.3 Customer engagement strategies
6.4 Feedback collection mechanisms
6.5 Relationship management tools
6.6 Community building initiatives
6.7 Customer satisfaction metrics
7. Value Proposition
7.1 Sustainability
7.2 Integrated supply chains
7.3 Unique selling points
7.4 Competitive advantages
7.5 Customer-centric approaches
7.6 Value delivery mechanisms
7.7 Long-term benefits for stakeholders
8. Key Activities
8.1 Regulatory compliance
8.2 Branding
8.3 Distribution setup
8.4 Market research and analysis
8.5 Training and development
8.6 Performance monitoring
8.7 Stakeholder engagement
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product mix
9.1.2 Pricing band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target countries
9.2.2 Compliance roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven analysis
13.2 Long-term sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone tracking
15.2.2 Activity scheduling
15.2.3 Resource allocation
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of government health reports and vaccination statistics from the Ministry of Health, Oman
- Review of industry publications and market reports on vaccine manufacturing trends in the Middle East
- Examination of regulatory frameworks and guidelines from the World Health Organization (WHO) and local health authorities
Primary Research
- Interviews with key stakeholders in the vaccine supply chain, including manufacturers and distributors
- Surveys targeting healthcare professionals and public health officials regarding vaccine needs and preferences
- Field interviews with executives from local pharmaceutical companies involved in vaccine production
Validation & Triangulation
- Cross-validation of data through multiple sources, including government reports and industry insights
- Triangulation of findings from primary interviews with secondary data to ensure consistency
- Sanity checks conducted through expert panel discussions with industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total vaccine market size based on national healthcare expenditure and vaccination coverage rates
- Segmentation of the market by vaccine type, including pediatric, adult, and travel vaccines
- Incorporation of government vaccination initiatives and public health campaigns into market projections
Bottom-up Modeling
- Collection of production capacity data from local vaccine manufacturers
- Operational cost analysis based on manufacturing processes and supply chain logistics
- Volume estimates derived from historical sales data and projected demand growth
Forecasting & Scenario Analysis
- Multi-variable forecasting models incorporating population growth, disease prevalence, and vaccination rates
- Scenario analysis based on potential changes in government policy and public health priorities
- Development of baseline, optimistic, and pessimistic market growth scenarios through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Officials | 60 | Health Ministry Representatives, Epidemiologists |
| Vaccine Manufacturers | 40 | Production Managers, Quality Assurance Officers |
| Healthcare Providers | 80 | Doctors, Pharmacists, Clinic Administrators |
| Supply Chain Experts | 50 | Logistics Managers, Procurement Specialists |
| Regulatory Bodies | 40 | Compliance Officers, Regulatory Affairs Managers |
Frequently Asked Questions
What is the current value of the Oman Vaccine Contract Manufacturing Market?
The Oman Vaccine Contract Manufacturing Market is valued at approximately USD 120 million, reflecting its share within the broader pharmaceutical contract manufacturing sector in Oman, which is experiencing steady growth due to increased public health initiatives and rising vaccine demand.