Region:Middle East
Author(s):Shubham
Product Code:KRAC8992
Pages:93
Published On:November 2025
By Type:
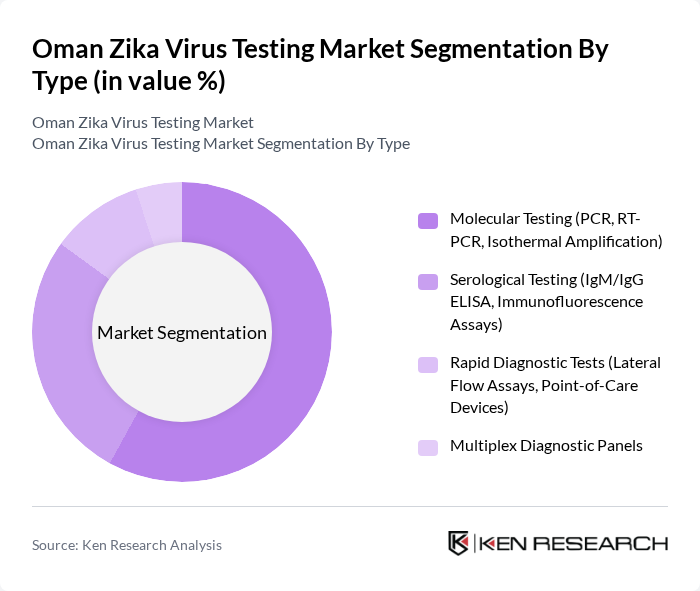
Themolecular testing segment, particularly PCR and RT-PCR methods, is dominating the market due to their high sensitivity and specificity in detecting Zika virus RNA. These methods are preferred in clinical settings for their rapid turnaround times and ability to confirm active infections. The increasing prevalence of Zika virus cases globally and the need for accurate differentiation from other arboviral infections have led to a surge in demand for molecular testing, as healthcare providers seek reliable diagnostic tools to manage outbreaks effectively .
By End-User:
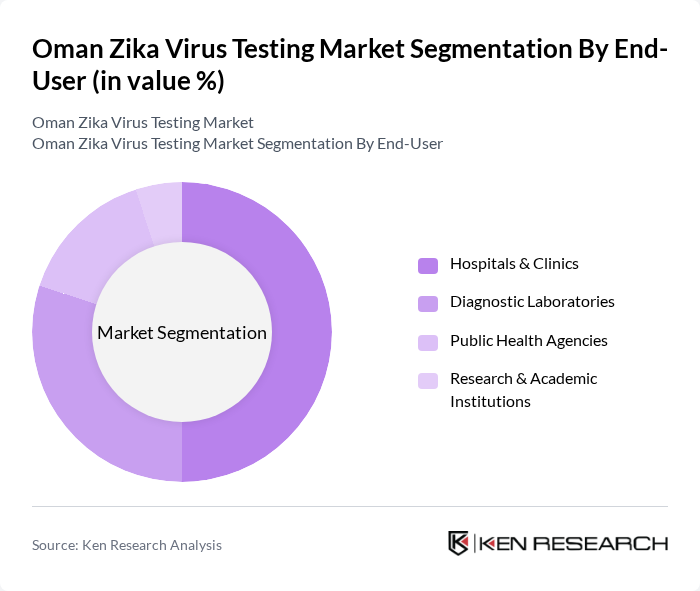
Hospitals and clinicsare the leading end-users in the Zika virus testing market, accounting for the largest share due to their direct involvement in patient care and disease management. These facilities are equipped with advanced diagnostic technologies and have the capacity to conduct a high volume of tests, making them essential in the early detection and treatment of Zika virus infections. The collaboration between hospitals and public health agencies further enhances the testing capabilities and response strategies in the region .
The Oman Zika Virus Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Bio-Rad Laboratories, Cepheid, Thermo Fisher Scientific, Hologic, Inc., QuidelOrtho Corporation, GenMark Diagnostics (a Roche company), Becton, Dickinson and Company (BD), PerkinElmer, Inc. (now Revvity, Inc.), Grifols S.A., DiaSorin S.p.A. (including Luminex Corporation), Quest Diagnostics, bioMérieux S.A. contribute to innovation, geographic expansion, and service delivery in this space.
The future outlook for the Oman Zika virus testing market appears promising, driven by increasing public health awareness and technological advancements. As the government continues to invest in healthcare infrastructure, the accessibility of testing facilities is expected to improve. Additionally, the integration of telemedicine services will facilitate remote consultations and testing, enhancing patient engagement. With ongoing collaborations with international health organizations, Oman is poised to strengthen its response to Zika virus outbreaks and improve overall public health outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Molecular Testing (PCR, RT-PCR, Isothermal Amplification) Serological Testing (IgM/IgG ELISA, Immunofluorescence Assays) Rapid Diagnostic Tests (Lateral Flow Assays, Point-of-Care Devices) Multiplex Diagnostic Panels |
| By End-User | Hospitals & Clinics Diagnostic Laboratories Public Health Agencies Research & Academic Institutions |
| By Age Group | Neonates & Infants Children Adults Pregnant Women |
| By Geographic Distribution | Muscat Governorate Dhofar Governorate Al Batinah Region Other Regions |
| By Testing Frequency | Routine Surveillance Testing Outbreak/Emergency Testing Prenatal/Screening Programs Travel-Related Testing |
| By Sample Type | Blood (Serum/Plasma) Urine Saliva Other Bodily Fluids |
| By Testing Methodology | Laboratory-Based Testing Point-of-Care Testing Home/At-Home Testing Kits Mobile Testing Units |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Public Health Officials | 50 | Epidemiologists, Health Program Managers |
| Laboratory Managers | 40 | Clinical Lab Directors, Quality Control Managers |
| Healthcare Providers | 40 | General Practitioners, Infectious Disease Specialists |
| Policy Makers | 40 | Health Policy Analysts, Government Advisors |
| Research Scientists | 40 | Virologists, Public Health Researchers |
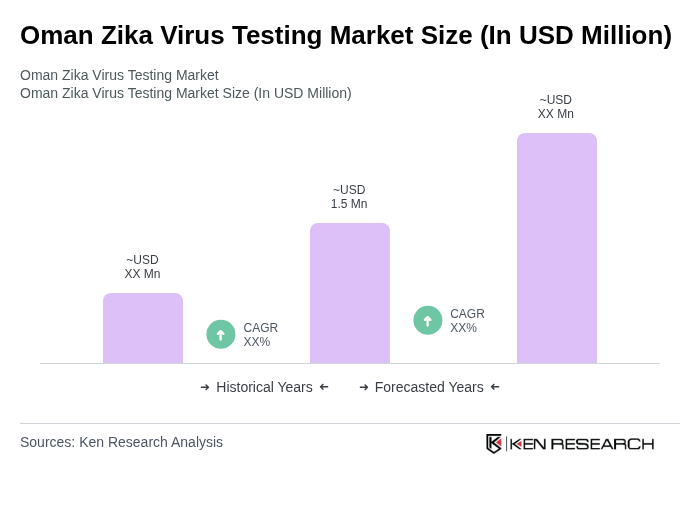
The Oman Zika Virus Testing Market is valued at approximately USD 1.5 million. This valuation is based on a five-year historical analysis and reflects the proportional allocation from the global Zika virus testing market size.