Region:Asia
Author(s):Shubham
Product Code:KRAD0918
Pages:96
Published On:November 2025
By Product Type:
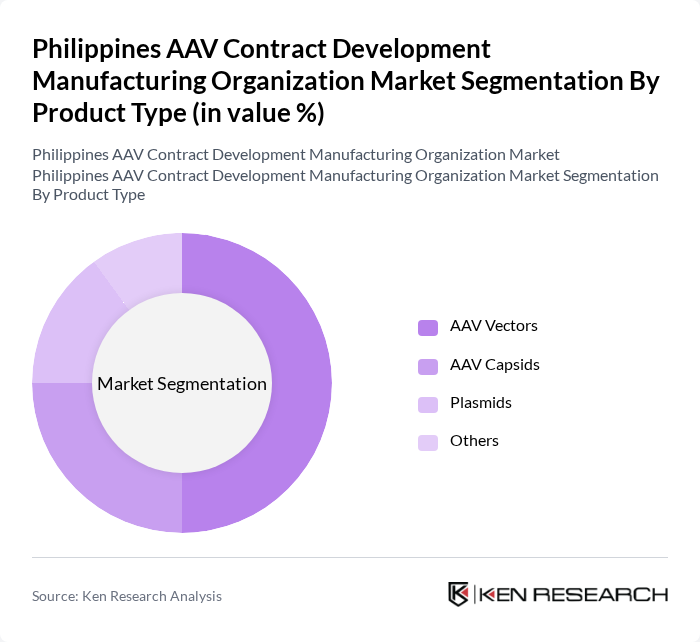
The product type segmentation includes AAV Vectors, AAV Capsids, Plasmids, and Others. Among these, AAV Vectors are the leading subsegment, driven by their critical role in gene therapy applications. The increasing focus on personalized medicine and advancements in gene editing technologies have further propelled the demand for AAV Vectors. AAV Capsids and Plasmids also contribute significantly, but the unique advantages of AAV Vectors in delivering therapeutic genes make them the preferred choice for many biopharmaceutical companies.
By Service Type:
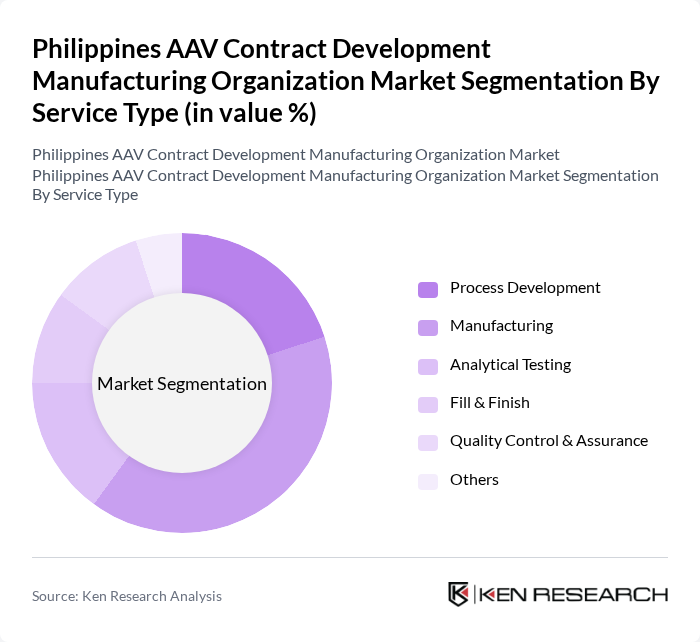
The service type segmentation encompasses Process Development, Manufacturing, Analytical Testing, Fill & Finish, Quality Control & Assurance, and Others. Manufacturing is the dominant subsegment, as it is essential for the large-scale production of AAV products. The increasing number of clinical trials and the need for high-quality manufacturing processes have led to a significant rise in demand for manufacturing services. Process Development and Quality Control & Assurance also play crucial roles in ensuring the efficacy and safety of AAV products.
The Philippines AAV Contract Development Manufacturing Organization Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Thermo Fisher Scientific Inc. (Patheon), WuXi Advanced Therapies (WuXi AppTec), Syngene International Ltd., Gene Therapy Solutions Inc. (Philippines), Merck KGaA (MilliporeSigma), Catalent, Inc., Samsung Biologics, AGC Biologics, Philippine Genome Center, Batangas Life Science Park Biotech Hub, Fujifilm Diosynth Biotechnologies, Unilab, Inc. (Philippines), and Mabuhay Bioventures contribute to innovation, geographic expansion, and service delivery in this space.
The future of the AAV market in the Philippines appears promising, driven by increasing urbanization and government support for local manufacturing. In future, advancements in electric and hybrid AAV technologies are expected to gain traction, aligning with global sustainability trends. Furthermore, strategic partnerships with international aerospace firms will likely enhance local capabilities, fostering innovation and competitiveness. As the market matures, the Philippines could emerge as a key player in the Southeast Asian AAV landscape, attracting further investments and talent.
| Segment | Sub-Segments |
|---|---|
| By Product Type | AAV Vectors AAV Capsids Plasmids Others |
| By Service Type | Process Development Manufacturing Analytical Testing Fill & Finish Quality Control & Assurance Others |
| By Application | Gene Therapy Development Vaccine Development Biopharmaceutical & Pharmaceutical Discovery Biomedical Research Others |
| By End-User | Pharmaceutical Companies Biopharmaceutical Companies Academic & Research Institutes Others |
| By Manufacturing Workflow | Upstream Processing Vector Amplification & Expansion Vector Recovery & Harvesting Downstream Processing Purification Fill Finish Others |
| By Culture Type | Adherent Culture Suspension Culture Others |
| By Region (Philippines) | Luzon Visayas Mindanao Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| AAV Production Facilities | 60 | Production Managers, Quality Assurance Leads |
| Gene Therapy Research Institutions | 50 | Research Scientists, Lab Directors |
| Biopharmaceutical Companies Utilizing AAV | 55 | Product Development Managers, Regulatory Affairs Specialists |
| Healthcare Providers Administering AAV Therapies | 45 | Clinical Directors, Pharmacists |
| Investors in Biotech Startups | 40 | Venture Capitalists, Angel Investors |
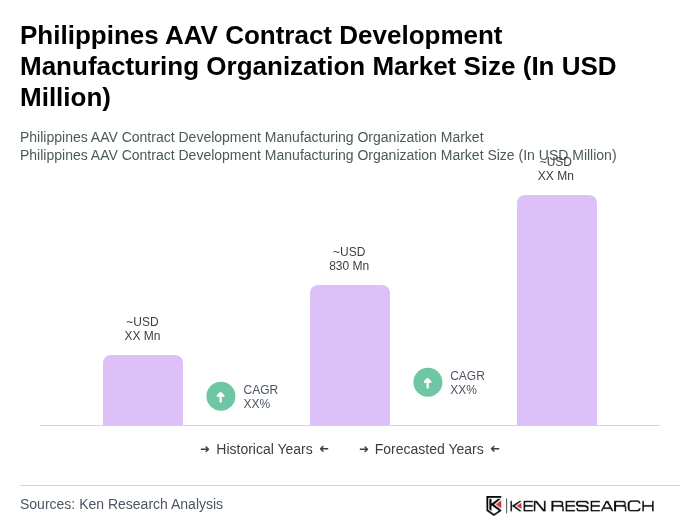
The Philippines AAV Contract Development Manufacturing Organization market is valued at approximately USD 830 million, driven by increasing demand for advanced therapies, particularly in gene and cell therapy, and rising investments in biopharmaceuticals and vaccine development.