Region:Asia
Author(s):Shubham
Product Code:KRAD3661
Pages:88
Published On:November 2025
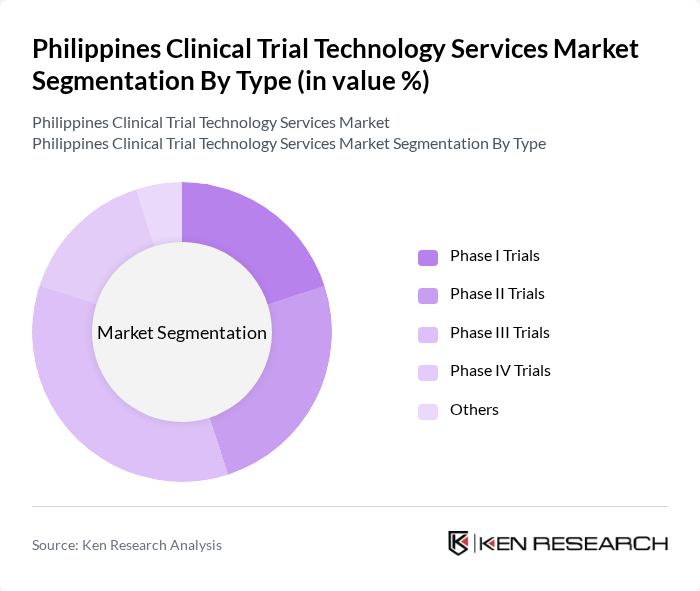
By Type:The market is segmented into various types of clinical trials, including Phase I, Phase II, Phase III, Phase IV, and others. Among these, Phase III trials dominate the market due to their critical role in determining the efficacy and safety of new treatments before they receive regulatory approval. The increasing complexity of drug development and the need for extensive patient data contribute to the prominence of this segment.
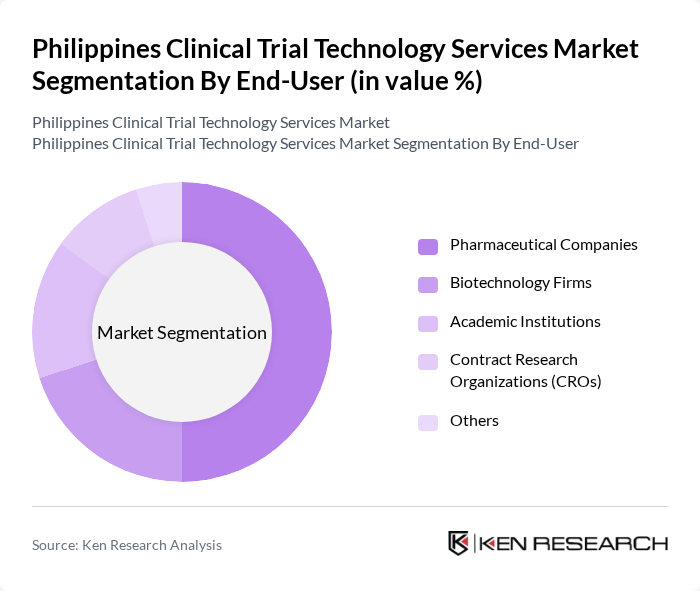
By End-User:The end-user segmentation includes pharmaceutical companies, biotechnology firms, academic institutions, contract research organizations (CROs), and others. Pharmaceutical companies are the leading end-users, driven by their need for extensive clinical data to support drug development and regulatory submissions. The increasing investment in R&D by these companies significantly influences the market dynamics.
The Philippines Clinical Trial Technology Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as IQVIA Holdings Inc., Medidata Solutions (Dassault Systèmes), Oracle Health Sciences, DATATRAK International Inc., Clario, PAREXEL International, Labcorp Drug Development, Veeva Systems Inc., Wipro Limited, ICON plc, Syneos Health, WuXi AppTec, Novotech CRO, Medpace, PRA Health Sciences (now part of ICON plc) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Philippines clinical trial technology services market appears promising, driven by advancements in digital health technologies and a growing emphasis on patient-centric approaches. As the healthcare landscape evolves, the integration of artificial intelligence and machine learning into clinical trials will enhance data analysis and patient engagement. Furthermore, the shift towards decentralized trials will facilitate broader participation, particularly in underserved regions, ultimately improving trial efficiency and outcomes.
| Segment | Sub-Segments |
|---|---|
| By Type | Phase I Trials Phase II Trials Phase III Trials Phase IV Trials Others |
| By End-User | Pharmaceutical Companies Biotechnology Firms Academic Institutions Contract Research Organizations (CROs) Others |
| By Therapeutic Area | Oncology Cardiovascular Neurology Infectious Diseases Others |
| By Study Design | Interventional Studies Observational Studies Expanded Access Studies Others |
| By Patient Population | Adult Patients Pediatric Patients Geriatric Patients Others |
| By Geographic Focus | Urban Areas Rural Areas Regional Trials Others |
| By Funding Source | Government Funding Private Investment Non-Profit Organizations Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 45 | Clinical Research Managers, Regulatory Affairs Specialists |
| Clinical Research Organizations (CROs) | 38 | Project Managers, Data Analysts |
| Healthcare Institutions | 42 | Clinical Trial Coordinators, Principal Investigators |
| Technology Providers | 35 | Product Managers, Technical Support Leads |
| Regulatory Bodies | 28 | Policy Makers, Compliance Officers |
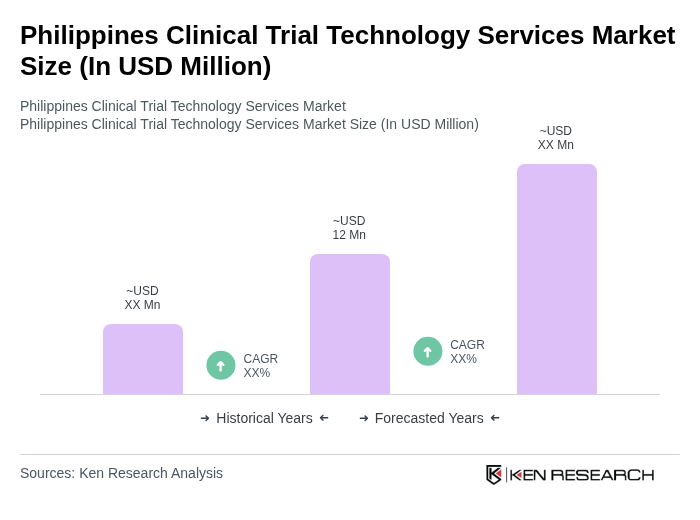
The Philippines Clinical Trial Technology Services Market is valued at approximately USD 12 million, reflecting a five-year historical analysis. This growth is driven by an increasing number of clinical trials and advancements in digital health technologies.