Region:Asia
Author(s):Geetanshi
Product Code:KRAC3747
Pages:82
Published On:October 2025
 Monitoring Devices Market.png)
By Type:
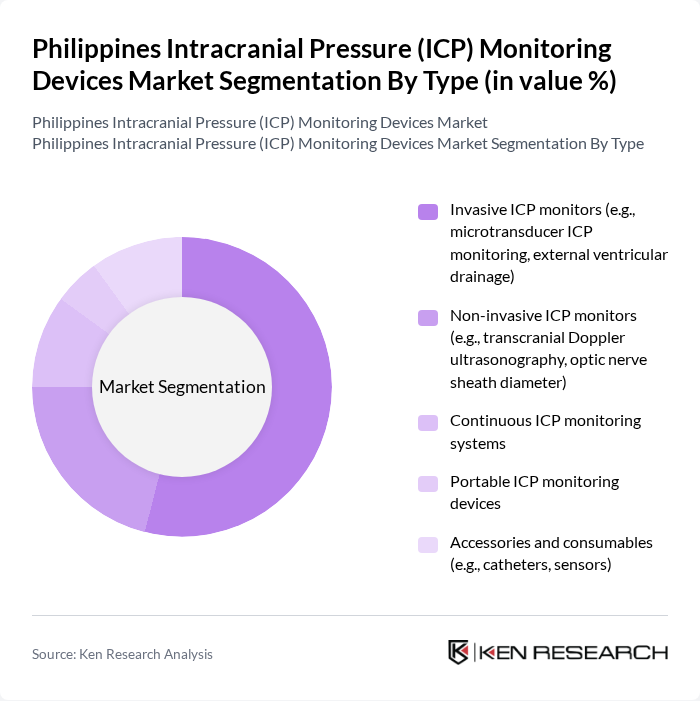
The ICP monitoring devices market can be segmented into various types, including invasive ICP monitors, non-invasive ICP monitors, continuous ICP monitoring systems, portable ICP monitoring devices, and accessories and consumables. Among these,invasive ICP monitors, such as microtransducer ICP monitoring and external ventricular drainage, dominate the market due to their accuracy and reliability in critical care settings. The increasing incidence of traumatic brain injuries and the need for precise monitoring in neurosurgery are driving the demand for these devices. Non-invasive monitors are gaining traction as well, but the invasive segment remains the leader due to its established efficacy and clinical preference in acute care .
By End-User:
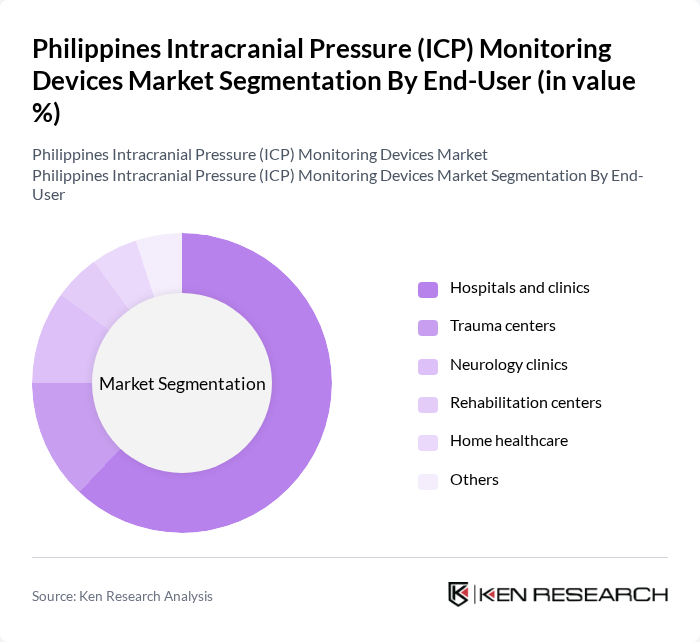
This market segment includes hospitals and clinics, trauma centers, neurology clinics, rehabilitation centers, home healthcare, and others.Hospitals and clinicsare the leading end-users, accounting for a significant share of the market. The increasing number of patients requiring critical care and the growing focus on advanced monitoring technologies in hospitals are key factors driving this segment. Trauma centers also play a crucial role, especially in regions with high incidences of accidents and injuries, further contributing to the demand for ICP monitoring devices. Hospitals and clinics remain the primary end-users globally, with a share exceeding 70% in most markets .
The Philippines Intracranial Pressure (ICP) Monitoring Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic, Codman Neuro (a Johnson & Johnson company), Natus Medical Incorporated, Integra LifeSciences, Raumedic AG, Spiegelberg GmbH & Co. KG, NeuroWave Systems Inc., ORA Medical, Sophysa, Aesculap AG (a B. Braun company), BrainScope Company, Inc., Cerenovus (a Johnson & Johnson company), B. Braun Melsungen AG, Stryker Corporation, Nihon Kohden Corporation contribute to innovation, geographic expansion, and service delivery in this space.
The future of the ICP monitoring devices market in the Philippines appears promising, driven by ongoing technological advancements and increased healthcare investments. As the government continues to prioritize healthcare infrastructure, the integration of telemedicine and remote monitoring solutions is expected to enhance patient care. Furthermore, the growing trend towards minimally invasive procedures will likely lead to the development of more efficient ICP devices, improving accessibility and affordability for healthcare providers across the nation.
| Segment | Sub-Segments |
|---|---|
| By Type | Invasive ICP monitors (e.g., microtransducer ICP monitoring, external ventricular drainage) Non-invasive ICP monitors (e.g., transcranial Doppler ultrasonography, optic nerve sheath diameter) Continuous ICP monitoring systems Portable ICP monitoring devices Accessories and consumables (e.g., catheters, sensors) |
| By End-User | Hospitals and clinics Trauma centers Neurology clinics Rehabilitation centers Home healthcare Others |
| By Application | Traumatic brain injury Stroke management Hydrocephalus Intracerebral hemorrhage Subarachnoid hemorrhage Post-operative monitoring CSF management Others |
| By Distribution Channel | Direct sales Distributors Online sales Others |
| By Region | Luzon Visayas Mindanao Others |
| By Price Range | Low range Mid range High range |
| By Technology | Fiber optic technology Microchip technology Wireless technology Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgery Departments | 60 | Neurosurgeons, Medical Directors |
| Critical Care Units | 50 | Intensivists, Critical Care Nurses |
| Hospital Procurement Teams | 40 | Procurement Managers, Supply Chain Officers |
| Biomedical Engineering Departments | 40 | Biomedical Engineers, Equipment Managers |
| Healthcare Regulatory Bodies | 40 | Regulatory Affairs Specialists, Compliance Officers |
The Philippines Intracranial Pressure (ICP) Monitoring Devices Market is valued at approximately USD 18 million, driven by the rising prevalence of neurological disorders and advancements in medical technology, particularly in critical care settings.