The $100B Biosimilars Shift in APAC – How Asia is Redefining Global Biopharma Economics
Asia is Reshaping Global Biopharma Dynamics Through Affordable Innovation, Expiring Patents, and Strategic Local Leverage.

















Built for Leaders Across
If you’re leading product strategy, portfolio planning, market access, or regional commercialization in biopharma, this is your field guide to thenext $100 billion global disruption.
This POV is designed specifically for:
- CXOs & Global Expansion Leaders in biologics, biosimilars, and chronic therapies
- VCs & PE Funds evaluating biotech returns and M&A possibilities in Asia
- CDMOs, API Suppliers, and Local Biotech Firms scaling production in India, China, Korea, and Japan
- Health Economists and Policy Regulators working on biosimilar guidelines and cost frameworks in emerging health systems
Executive Summary
Biosimilars are no longer a regulatory side story. In APAC, they are becoming the foundation for a new era of accessible biopharmaceutical innovation.
The Asia-Pacific region is expected to add$77.7 billionin new biopharma value between 2023 and 2029, with biosimilars driving much of this growth. APAC's market—currently worth$170.5 billion—is projected to hit$248.2 billionby 2029. But what's more important ishowthis growth is happening:
- Biosimilars in APACare priced at30–70% lessthan branded originator therapies
- Local productionand regulatory acceleration are enabling faster market access
- Patent cliffsacross blockbuster biologics are creatinga $100B opportunity window
From oncology to autoimmune and cardiovascular, the region is scaling biosimilar production at a pace never seen before.
The Market Snapshot – How Big Is This Shift?
Market Size & CAGR:
- Global Biopharma (2023): USD 905.3B
- APAC Biopharma (2023): USD 170.5B
- APAC Forecast (2029): USD 248.2B
- CAGR (2023–2029): 6.5% (faster than global biopharma average of 5.1%)

Affordability Metrics:
- Biosimilars cost30–70% lessthan their innovator equivalents
- Countries like India, Vietnam, and China offer a6–8x pricing differentialvs Western markets
- Treatment costs for HER2+ cancer in India have dropped from USD 40,000 to under USD 4,000 due to biosimilars
Policy & Production Levers:
- Government-linked price caps and tax incentives in China and Korea
- Japan’s prescribing incentives for biosimilar switches
- India’s regulatory acceleration through CDSCO and make-in-India provisions
Biosimilar Penetration – Where Is The Growth Real?
Penetration in key markets isexpected to triple by 2029, with clear leadership emerging:
- India: from0.6% → 1.7%
- China: from2.1 % → 6.1 %
- South Korea: from9.7% → 32.8%
- Japan: from5.5% → 14.8%
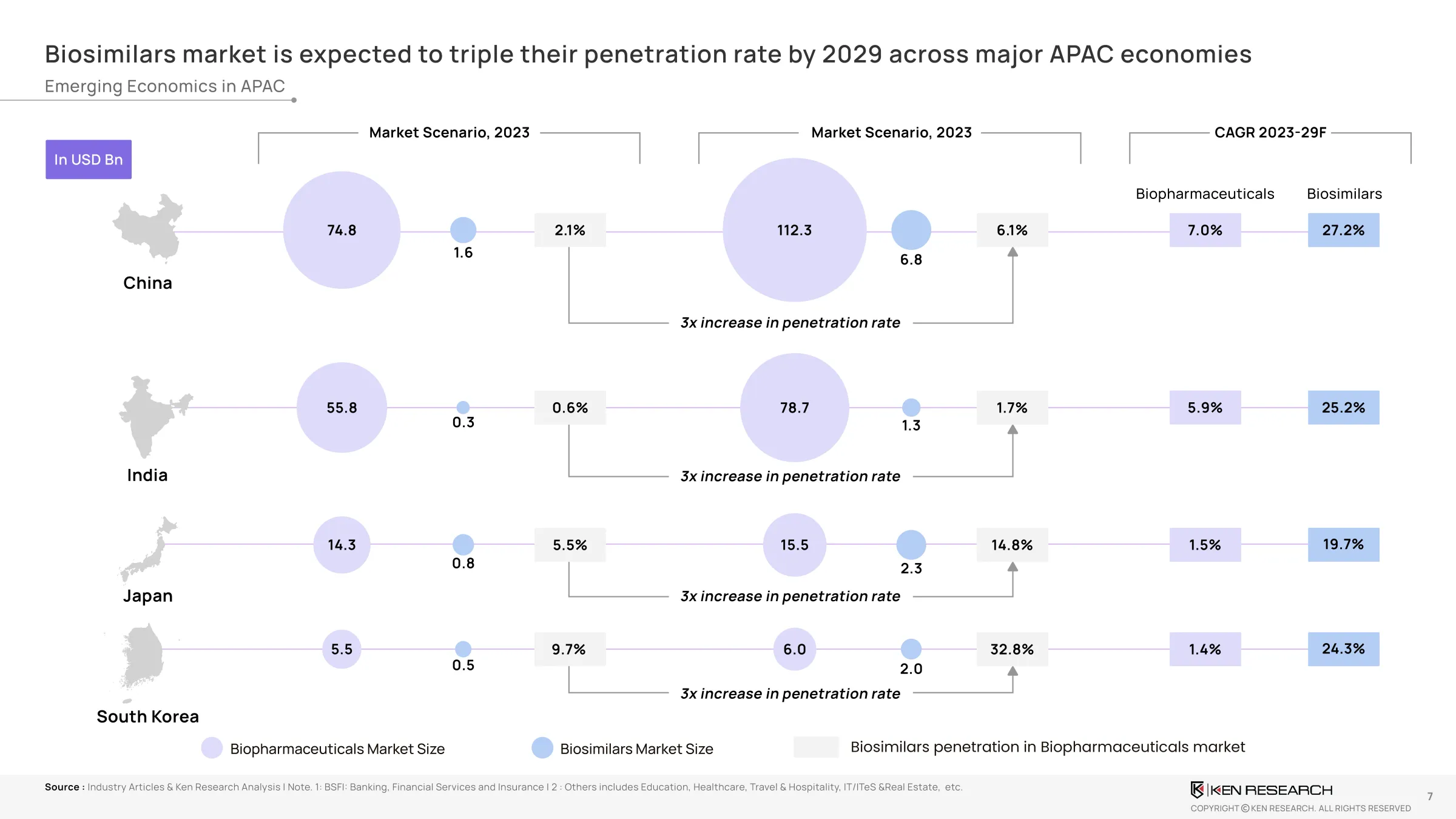
This is not just statistical growth—it’s astructural reallocation of market share. Biosimilars are now replacing innovator mAbs in public procurement. National health systems are integrating biosimilar-led therapy options into their formularies.
Patent Cliffs & Pipeline – Where The Value Is Shifting
More thanUSD 100 billion in biologic drug revenue is at riskfrom expiring patents between 2023–2030.
Top Drugs Losing Exclusivity:
- Keytruda (USD 17.2B)s
- Humira (USD 16.1B)
- Opdivo, Eliquis, Enbrel, Avastin, Rituxan…
Impact:
- 50+ biosimilar candidates are expected to enter the market by 2030
- Oncology, autoimmune, and cardiovascular dominate this pipeline
- APAC firms like Celltrion, Biocon, Dr. Reddy’s, and Samsung Bioepis are leading filings
Strategic Shift – The Third Global Wave Of Biosimilars
The world has seen two biosimilar waves before:
- Wave 1: Europe – Cytokines, hormones (2005–2015)
- Wave 2: US – Rheumatology, mAbs (2015–2020)
Now comesWave 3 – APAC, focused on:
Oncology
- High-cost chronic therapies
- Multi-indication biologics
- Regional IP-driven manufacturing
This is APAC’s moment tolead global biosimilar affordability and accessibility, just as it once led generics in small molecules.
TAM – The $102.6b Biosimilar Opportunity By 2029
Therapy Area Breakdown:
- Oncology: USD 64.3B
- Cardio-Metabolic: USD 16.4B
- Autoimmune: USD 2.9B
- Neuro/Hormonal/Others: USD 19B+
Top 4 Focus Countries:
- India: Biosimilar exports and CDMO play
- China: Local approvals + global licensing
- Japan: Prescriber shift via centralized incentives
- South Korea: Leading on biosimilar APIs and bulk drug capacity
Who’s Winning – M&A, Jv, And Launch Landscape 2024
Recent strategic deals include:
Celltrion’s $278M acquisition of Takeda’s biosimilar assets
Samsung Bioepis expanding via Biogen divestiture (~$775M)
Biocon’s five-year alliance with Sandoz (Australia roll-out)
Zydus + Dr. Reddy’s co-developing HER2+ oncology biosimilars
Track strategic plays shaping the biosimilar map across APAC and beyond
FAQ's
Still Got Questions? Connect Via Mail
What is the size of the APAC biopharmaceuticals market by 2029?
The APAC biopharma market is projected to reach USD 248.2 billion by 2029, growing at a 6.5% CAGR from its 2023 base of USD 170.5 billion.
How much is the biosimilar opportunity in APAC worth?
Over USD 100 billion by 2029, with oncology contributing 64% of the total, driven by patent expiries and growing acceptance.
What makes APAC critical for global biosimilar growth?
Its cost advantage (up to 8x), policy acceleration, and strong local manufacturing make APAC the ideal biosimilar export and consumption hub.
Which countries are leading biosimilar adoption?
India, China, Japan, and South Korea are investing in production incentives, biosimilar switching, and global export models.
How are global players entering the APAC biosimilar ecosystem?
Through M&A (Celltrion, Biocon), JVs (Sandoz), and licensing strategies, especially in oncology and autoimmunity biosimilars.















