Region:Middle East
Author(s):Rebecca
Product Code:KRAC3178
Pages:89
Published On:October 2025
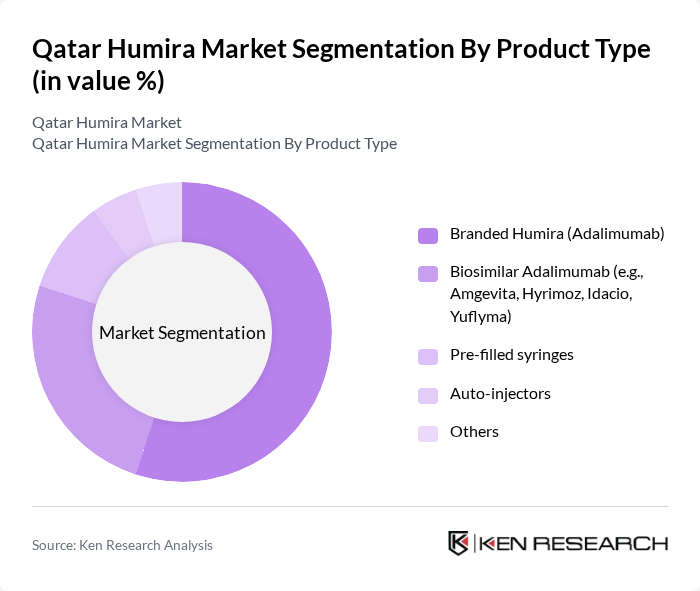
By Product Type:The product type segmentation reflects a market increasingly diversified by the entry of biosimilar adalimumab, which is gaining traction as a cost-effective alternative to branded Humira. Branded Humira (adalimumab) remains the leading sub-segment due to its established efficacy, but biosimilars such as Amgevita, Hyrimoz, Idacio, and Yuflyma are rapidly increasing their market share. Pre-filled syringes and auto-injectors are favored for their convenience, supporting patient adherence and self-administration. The “Others” category includes less common formulations and delivery systems tailored to specific patient requirements .
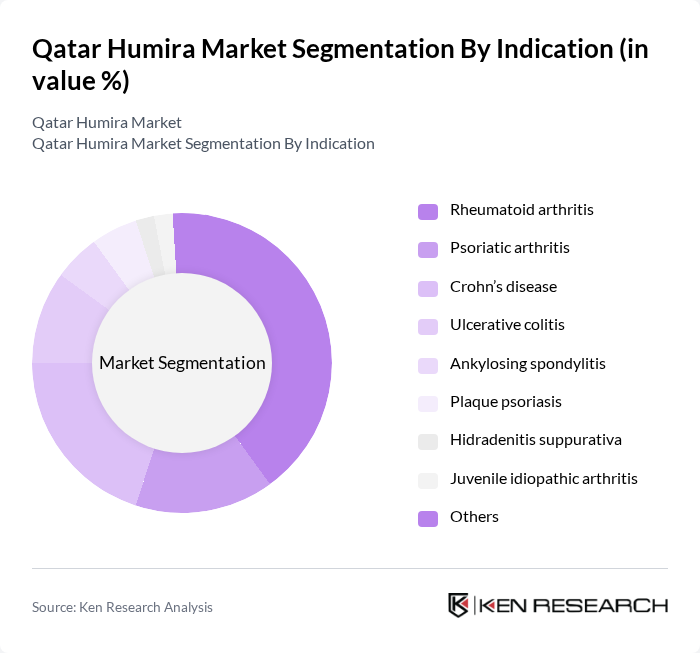
By Indication:The indication segmentation demonstrates that rheumatoid arthritis remains the primary driver of Humira prescriptions in Qatar, consistent with global trends. Crohn’s disease and ulcerative colitis are also significant indications, reflecting increased diagnosis rates and clinical awareness in the region. Other approved indications, including psoriatic arthritis, ankylosing spondylitis, plaque psoriasis, hidradenitis suppurativa, and juvenile idiopathic arthritis, contribute to the diversified use of adalimumab. The growing availability of biosimilars is further expanding access across these indications .
The Qatar Humira market is characterized by a dynamic mix of regional and international players. Leading participants such as AbbVie Inc., Amgen Inc., Sandoz International GmbH, Pfizer Inc., Celltrion Inc., Samsung Bioepis Co., Ltd., Biogen Inc., Fresenius Kabi AG, Mylan N.V. (Viatris Inc.), Coherus BioSciences, Inc., Alvotech, Boehringer Ingelheim Pharmaceuticals, Inc., Fujifilm Kyowa Kirin Biologics Co., Ltd., Stada Arzneimittel AG, Hikma Pharmaceuticals PLC contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Qatar Humira market appears promising, driven by increasing healthcare investments and a growing focus on personalized medicine. As the government continues to enhance healthcare infrastructure, patient access to advanced therapies is expected to improve significantly. Furthermore, the integration of telemedicine and home healthcare services will facilitate better management of autoimmune diseases, ensuring that patients receive timely treatment and support, ultimately fostering market growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Product Type | Branded Humira (Adalimumab) Biosimilar Adalimumab (e.g., Amgevita, Hyrimoz, Idacio, Yuflyma) Pre-filled syringes Auto-injectors Others |
| By Indication | Rheumatoid arthritis Psoriatic arthritis Crohn’s disease Ulcerative colitis Ankylosing spondylitis Plaque psoriasis Hidradenitis suppurativa Juvenile idiopathic arthritis Others |
| By End-User | Hospitals Specialty clinics Home care settings Others |
| By Distribution Channel | Hospital pharmacies Retail pharmacies Online pharmacies Others |
| By Patient Demographics | Age group (Children, Adults, Seniors) Gender (Male, Female) Disease type (Rheumatoid arthritis, Crohn's disease, etc.) |
| By Pricing Strategy | Premium pricing Competitive pricing Value-based pricing |
| By Geographic Distribution | Urban areas Rural areas Semi-urban areas |
| By Treatment Setting | Inpatient Outpatient Home-based care |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Rheumatology Clinics | 60 | Rheumatologists, Nurse Practitioners |
| Dermatology Practices | 50 | Dermatologists, Physician Assistants |
| Pharmacy Distribution Channels | 40 | Pharmacists, Pharmacy Managers |
| Patient Support Programs | 40 | Patient Advocates, Healthcare Coordinators |
| Health Insurance Providers | 40 | Claims Analysts, Policy Managers |
The Qatar Humira market is valued at approximately USD 85 million, driven by the rising prevalence of autoimmune diseases and the increasing adoption of biosimilar adalimumab, which offers cost-effective treatment options for patients.