Region:Middle East
Author(s):Dev
Product Code:KRAA3927
Pages:93
Published On:January 2026
By Trial Phase:The trial phase segmentation includes various stages of clinical trials, which are crucial for the development of new therapies. The phases are designed to assess the safety, efficacy, and optimal dosing of new treatments. Phase III trials are particularly significant as they involve larger patient populations and are pivotal for regulatory approval.
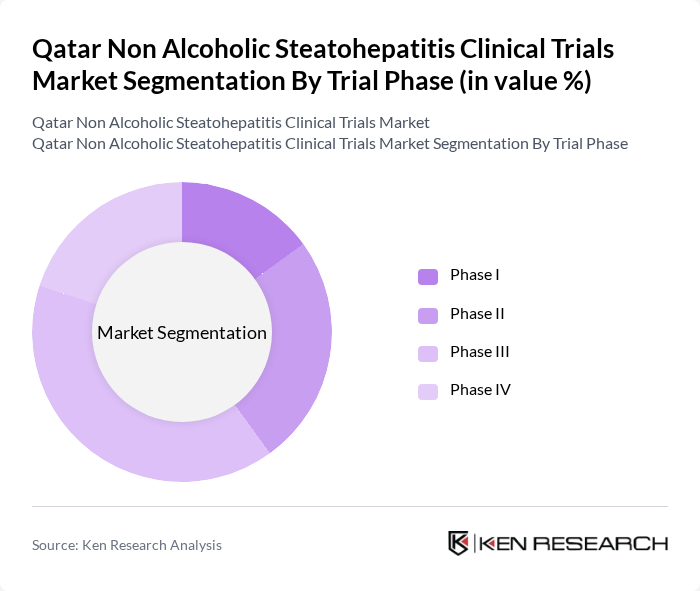
The Phase III trials dominate the market due to their critical role in determining the efficacy and safety of new treatments before they receive regulatory approval. These trials typically involve a larger and more diverse patient population, which enhances the reliability of the results. The increasing number of pharmaceutical companies focusing on NASH therapies has led to a surge in Phase III trials, making them the most significant segment in the market.
By Study Design:The study design segmentation categorizes clinical trials based on their methodology. This includes interventional trials, observational trials, and expanded access programs, each serving different purposes in the research landscape.
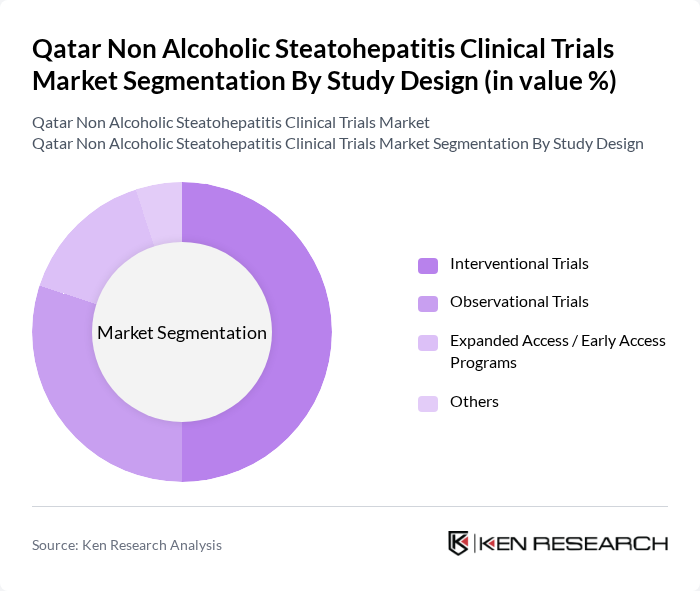
Interventional trials are the leading segment in the study design category, accounting for a significant portion of the market. These trials are essential for testing new drugs and therapies, providing critical data on their effectiveness and safety. The focus on developing innovative treatments for NASH has led to an increase in interventional studies, making them the backbone of clinical research in this area.
The Qatar Non Alcoholic Steatohepatitis Clinical Trials Market is characterized by a dynamic mix of regional and international players. Leading participants such as Hamad Medical Corporation (HMC), Sidra Medicine, Qatar University, Weill Cornell Medicine–Qatar, Qatar Biomedical Research Institute (QBRI), Qatar Foundation, Qatar National Research Fund (QNRF), Ministry of Public Health – Qatar, Qatar Metabolic Institute / Specialized Liver & Metabolic Centers, Selected Global Pharma & Biotech Sponsors Active in Qatar NASH Trials contribute to innovation, geographic expansion, and service delivery in this space.
The future of the NASH clinical trials market in Qatar appears promising, driven by increasing investments in healthcare infrastructure and a growing focus on personalized medicine. As the government continues to support research initiatives, the integration of digital health technologies is expected to enhance trial efficiency. Furthermore, the emphasis on patient-centric designs will likely improve recruitment and retention rates, fostering a more robust clinical trial environment that can adapt to emerging therapeutic needs.
| Segment | Sub-Segments |
|---|---|
| By Trial Phase | Phase I Phase II Phase III Phase IV |
| By Study Design | Interventional Trials Observational Trials Expanded Access / Early Access Programs Others |
| By Drug Class / Mechanism of Action | FXR Agonists PPAR Agonists GLP-1 Receptor Agonists and Related Metabolic Agents Anti-fibrotic Agents Others |
| By NASH Disease Stage | Early-Stage NASH (F0–F1) Intermediate Fibrosis (F2–F3) Advanced Fibrosis / Cirrhosis (F4) Others |
| By Patient Profile | With Type 2 Diabetes Mellitus Non-Diabetic with Metabolic Syndrome / Obesity Other Comorbid Conditions Others |
| By Sponsor Type | Global Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Others |
| By Trial Setting in Qatar | Tertiary Care & Academic Hospitals Specialized Liver & Metabolic Centers Multi-specialty Hospitals & Clinics Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Trial Investigators | 60 | Principal Investigators, Clinical Research Coordinators |
| Healthcare Providers | 80 | Hepatologists, General Practitioners |
| Patients with NASH | 120 | Individuals diagnosed with NASH, Caregivers |
| Pharmaceutical Industry Experts | 70 | Medical Affairs Managers, Market Access Specialists |
| Regulatory Authorities | 40 | Health Policy Makers, Regulatory Affairs Officers |
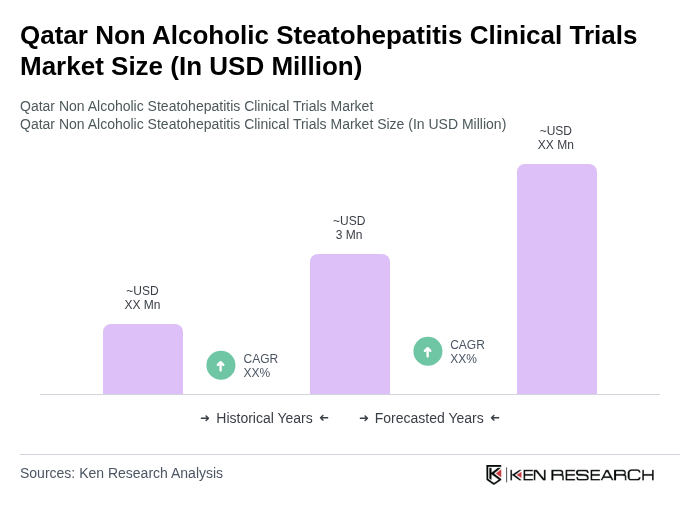
The Qatar Non Alcoholic Steatohepatitis Clinical Trials Market is valued at approximately USD 3 million, reflecting a significant growth driven by the rising prevalence of non-alcoholic fatty liver disease and increased awareness of liver health in the region.