Saudi Arabia Automated Cell Counting Market Overview
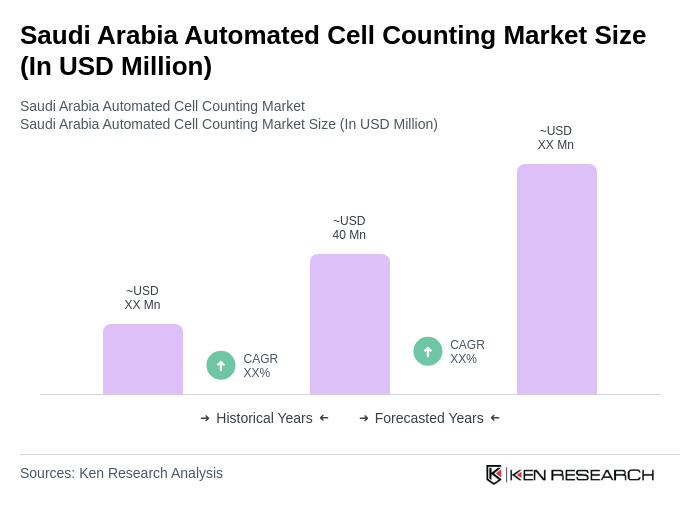
- The Saudi Arabia Automated Cell Counting Market is valued at USD 40 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic diseases such as diabetes (18.3% adult prevalence), cardiovascular conditions, and cancer, which are fueling demand for advanced diagnostic tools. Additional growth drivers include Vision 2030 healthcare modernization initiatives, a USD 65 billion government investment in healthcare infrastructure, and the expansion of private hospital networks and diagnostic chains. The market also benefits from the establishment of biotech hubs and pharmaceutical manufacturing facilities, which require precise cell counting for research, clinical trials, and drug development.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their advanced healthcare infrastructure and concentration of medical facilities. Riyadh, as the capital, hosts major hospitals, research institutions, and biotechnology centers. Jeddah is recognized as a commercial hub with a growing number of diagnostic laboratories, while Dammam’s strategic location supports access to healthcare services and facilitates regional distribution of cell counting technologies. These cities are pivotal in driving adoption and utilization of automated cell counting devices, supported by both public and private sector investments.
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Medical Devices Interim Regulation, 2023” issued by the Saudi Food and Drug Authority. This binding regulation mandates that all diagnostic laboratories utilize SFDA-approved automated cell counting devices, requiring compliance with international standards for accuracy, calibration, and safety. The regulation covers device registration, periodic calibration, operator training, and reporting of device performance, aiming to standardize laboratory practices and improve patient outcomes across the Kingdom.
Saudi Arabia Automated Cell Counting Market Segmentation
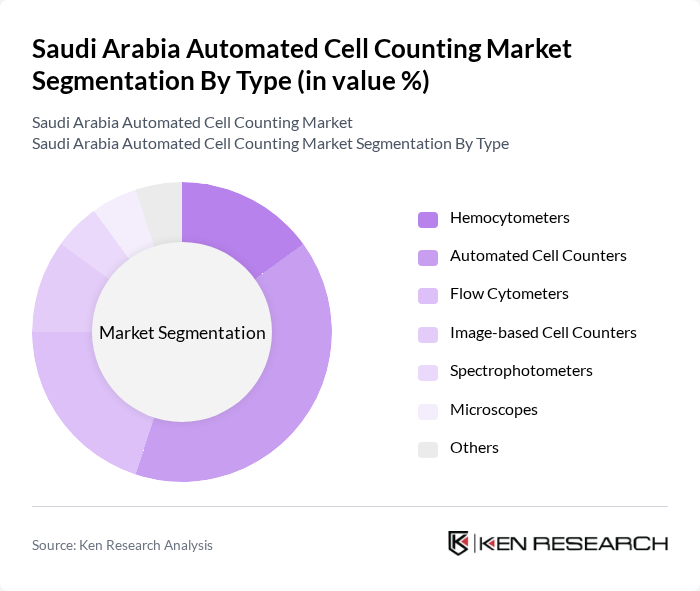
By Type:The market is segmented into Hemocytometers, Automated Cell Counters, Flow Cytometers, Image-based Cell Counters, Spectrophotometers, Microscopes, and Others. Among these, Automated Cell Counters are leading the market, driven by government mandates for healthcare quality and efficiency in high-volume facilities. Flow Cytometers are increasingly adopted in research institutions and specialized cancer centers, while Hemocytometers remain prevalent in smaller facilities and rural hospitals. Image-based Cell Counters and Spectrophotometers are gaining traction for specialized applications in research and bioprocessing. The demand for rapid, reliable results in hospitals and laboratories continues to accelerate the adoption of automated solutions.
By End-User:The end-user segmentation includes Hospitals & Diagnostic Laboratories, Pharmaceutical & Biotechnology Companies, Research & Academic Institutes, and Others. Hospitals and diagnostic laboratories are the primary users, accounting for over half of market demand, supported by Vision 2030’s expansion of healthcare facilities and systematic upgrades from manual to automated cell counting. Pharmaceutical and biotechnology companies are significant contributors, driven by local manufacturing, clinical trials, and R&D investments. Research and academic institutes, including major centers like KAUST, are emerging buyers for specialized applications in regenerative medicine and bioprocessing.
Saudi Arabia Automated Cell Counting Market Competitive Landscape
The Saudi Arabia Automated Cell Counting Market is characterized by a dynamic mix of regional and international players. Leading participants such as Beckman Coulter, Inc. (Danaher Corporation), Sysmex Corporation, Abbott Laboratories, Siemens Healthineers, Roche Diagnostics, Thermo Fisher Scientific Inc., Becton, Dickinson and Company (BD), Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., PerkinElmer, Inc., Merck KGaA, Mindray Medical International Limited, ChemoMetec A/S, Horiba Ltd., Eppendorf AG, GE Healthcare contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Automated Cell Counting Market Industry Analysis
Growth Drivers
- Increasing Demand for Accurate Diagnostics:The Saudi healthcare sector is witnessing a surge in demand for precise diagnostic tools, driven by a 15% annual increase in laboratory tests. This growth is fueled by the rising awareness of early disease detection, particularly in chronic conditions. The Ministry of Health's investment of approximately SAR 12 billion in healthcare technology in future further supports this trend, enhancing the adoption of automated cell counting systems for improved diagnostic accuracy.
- Rising Prevalence of Chronic Diseases:Chronic diseases, such as diabetes and cardiovascular disorders, are on the rise in Saudi Arabia, with diabetes prevalence reaching 19.2% in future. This alarming statistic necessitates advanced diagnostic tools, including automated cell counters, to monitor and manage these conditions effectively. The government's commitment to reducing chronic disease rates through enhanced healthcare services is expected to drive demand for automated solutions in the coming years.
- Technological Advancements in Cell Counting:The automated cell counting market is benefiting from rapid technological advancements, with innovations such as AI-driven analytics and high-throughput systems. In future, investments in R&D for medical devices in Saudi Arabia exceeded SAR 2 billion, fostering the development of more efficient and accurate cell counting technologies. These advancements are crucial for laboratories aiming to improve operational efficiency and diagnostic capabilities, thereby propelling market growth.
Market Challenges
- High Cost of Automated Cell Counting Systems:The initial investment required for automated cell counting systems can be a significant barrier, with prices ranging from SAR 250,000 to SAR 1.2 million. This high cost limits accessibility for smaller healthcare facilities and laboratories, which may opt for manual counting methods instead. As a result, the market faces challenges in achieving widespread adoption, particularly in less affluent regions of Saudi Arabia.
- Limited Awareness Among Healthcare Professionals:Despite the benefits of automated cell counting, many healthcare professionals in Saudi Arabia remain unaware of these technologies. A survey conducted in future indicated that only 35% of lab technicians were familiar with automated systems. This lack of awareness can hinder the adoption of advanced technologies, as professionals may prefer traditional methods they are more comfortable with, thus stalling market growth.
Saudi Arabia Automated Cell Counting Market Future Outlook
The future of the automated cell counting market in Saudi Arabia appears promising, driven by ongoing investments in healthcare infrastructure and technological innovations. As the government continues to prioritize healthcare improvements, the integration of AI and machine learning into diagnostic processes is expected to enhance efficiency and accuracy. Furthermore, the expansion of healthcare facilities will likely create a greater demand for automated solutions, positioning the market for significant growth in the coming years.
Market Opportunities
- Expansion of Healthcare Facilities:The Saudi government plans to increase the number of healthcare facilities by 25% in future, creating a substantial opportunity for automated cell counting systems. This expansion will necessitate the implementation of advanced diagnostic technologies to meet the growing patient demand, thereby driving market growth and adoption.
- Increasing Research Activities in Biotechnology:With biotechnology research funding reaching SAR 600 million in future, there is a significant opportunity for automated cell counting systems in research settings. Enhanced research activities will require precise cell counting technologies, fostering collaborations between research institutions and technology providers to innovate and improve diagnostic capabilities.