About the Report
Base Year 2024Listen to the audio summary
Saudi Arabia Bioanalytical Testing Services CRO Market Overview
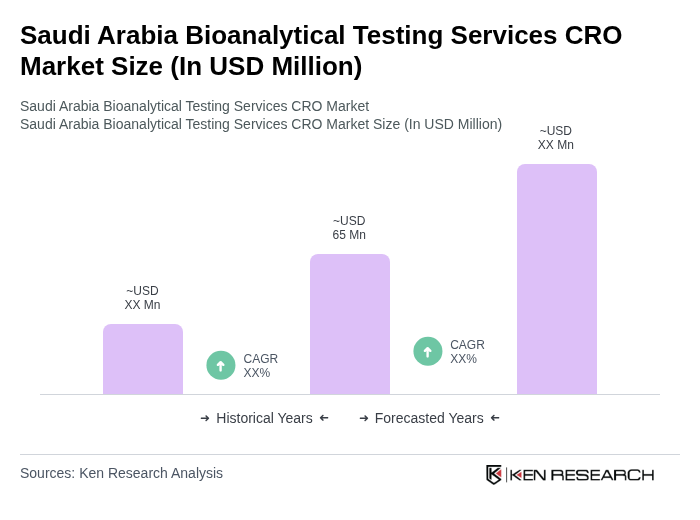
- The Saudi Arabia Bioanalytical Testing Services CRO Market is valued at USD 65 million, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for drug development and regulatory compliance, alongside the rising prevalence of chronic diseases such as diabetes and cardiovascular diseases that necessitate advanced testing services. The market is also supported by the expansion of pharmaceutical and biotechnology sectors under national initiatives such as Saudi Vision 2030, which emphasize local clinical research, biologics development, and strengthening of quality-control laboratories.
- Key cities such as Riyadh, Jeddah, and Dammam dominate the market due to their robust healthcare infrastructure and concentration of pharmaceutical and biopharmaceutical stakeholders. Riyadh, being the capital, serves as a hub for regulatory bodies and research institutions, including the Saudi Food and Drug Authority and major reference laboratories, while Jeddah and Dammam benefit from their strategic locations, port access, and links to industrial and logistics corridors, enhancing their roles in bioanalytical testing services and clinical trial support.
- In 2023, the Saudi Food and Drug Authority (SFDA) reinforced requirements for non?clinical and analytical laboratories to comply with Good Laboratory Practice standards through the “Guideline for Good Laboratory Practice (GLP) – Non?Clinical Health Studies” issued by the Saudi Food and Drug Authority in 2022, which sets obligations on study planning, quality assurance, facilities, equipment qualification, data integrity, and reporting for laboratories supporting medicinal product registration and regulatory submissions. This framework aims to ensure the reliability and quality of testing services, thereby enhancing the credibility of bioanalytical results in drug development and clinical trials and aligning Saudi Arabia with internationally recognized GLP practices.
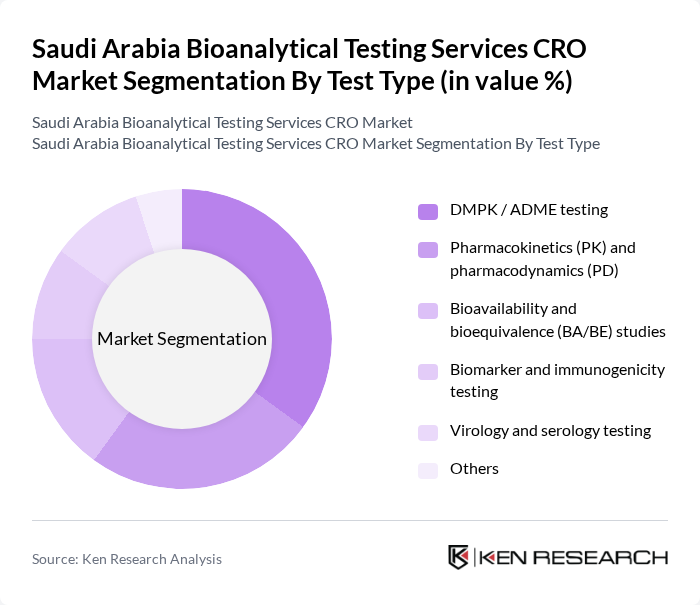
Saudi Arabia Bioanalytical Testing Services CRO Market Segmentation
By Test Type:The bioanalytical testing services market is segmented into various test types, each catering to specific needs in drug development and clinical research. The dominant sub-segment is pharmacokinetic (PK) testing, which is crucial for understanding drug absorption, distribution, metabolism, and excretion and is widely used across early and late-stage clinical trials. This segment is driven by the increasing focus on drug safety and efficacy, as well as regulatory requirements for comprehensive exposure–response and dose-optimization assessments. Other significant segments include pharmacodynamic (PD) and biomarker analysis, bioequivalence and bioavailability studies, immunogenicity and immunoassay-based testing, toxicokinetic and safety assessment, and cell-based and virology assays, which are essential for evaluating therapeutic response, comparability of generics and biosimilars, and safety in complex biologics and advanced therapies.
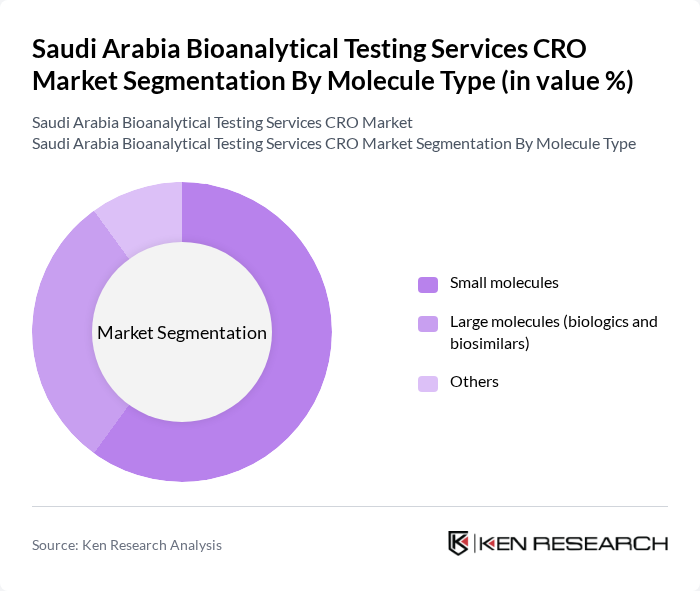
By Molecule Type:The market is also segmented by molecule type, which includes small molecules and large molecules (biologics and biosimilars). The small molecules segment remains a major revenue contributor globally due to their widespread use in traditional pharmaceuticals and generics, supported by extensive pipelines and ongoing research in areas such as cardiovascular, metabolic, and infectious diseases. The increasing number of small molecule drugs entering the market, coupled with the growing demand for generic drugs in Saudi Arabia’s expanding healthcare system, is driving this segment's growth. Large molecules (biologics and biosimilars) are gaining share, driven by rising adoption of monoclonal antibodies, vaccines, and other biologics, but they are associated with more complex assay requirements, stringent validation expectations, and specialized capabilities in immunogenicity and cell-based assays, which can moderate their pace of market penetration.
Saudi Arabia Bioanalytical Testing Services CRO Market Competitive Landscape
The Saudi Arabia Bioanalytical Testing Services CRO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Labcorp, Eurofins Scientific, Syneos Health, IQVIA, Charles River Laboratories, Wuxi AppTec, SGS, PRA Health Sciences / ICON, Medpace, Q2 Solutions, Celerion, PPD (Thermo Fisher Scientific), Local and regional Saudi-based CROs, Other niche bioanalytical service providers, Emerging GCC-based specialty labs contribute to innovation, geographic expansion, and service delivery in this space, often supporting pharmacokinetic studies, bioequivalence projects, biomarker programs, and complex biologics and cell and gene therapy trials.
Saudi Arabia Bioanalytical Testing Services CRO Market Industry Analysis
Growth Drivers
- Increasing Demand for Pharmaceutical Development:The pharmaceutical sector in Saudi Arabia is projected to reach $8.9 billion in future, driven by a growing population and rising healthcare needs. This surge in demand for innovative drugs necessitates bioanalytical testing services, which are crucial for drug development and regulatory compliance. The Kingdom's Vision 2030 initiative further emphasizes the importance of local pharmaceutical production, enhancing the need for robust testing services to support this growth.
- Rise in Biologics and Biosimilars Research:The biologics market in Saudi Arabia is expected to grow significantly, with an estimated value of $1.5 billion in future. This growth is fueled by increasing investments in research and development, particularly in biosimilars, which are projected to account for 25% of the total pharmaceutical market. As the demand for these complex therapies rises, bioanalytical testing services will play a vital role in ensuring their safety and efficacy, driving market expansion.
- Expansion of Clinical Trials in the Region:Saudi Arabia has seen a notable increase in clinical trials, with over 300 trials registered in future alone. This expansion is supported by government initiatives aimed at enhancing the clinical research landscape. The growing number of trials necessitates comprehensive bioanalytical testing services to ensure compliance with international standards, thereby driving demand for these services in the region and contributing to market growth.
Market Challenges
- Stringent Regulatory Requirements:The bioanalytical testing services market in Saudi Arabia faces significant challenges due to stringent regulatory requirements imposed by the Saudi Food and Drug Authority (SFDA). Compliance with Good Manufacturing Practices (GMP) and other regulatory standards can be costly and time-consuming, creating barriers for new entrants. This regulatory landscape can hinder the growth of smaller CROs, limiting competition and innovation within the market.
- High Operational Costs:The operational costs associated with bioanalytical testing services are notably high, with estimates suggesting that establishing a fully equipped laboratory can exceed $1.2 million. These costs include advanced equipment, skilled personnel, and compliance with regulatory standards. As a result, many CROs struggle to maintain profitability, which can stifle growth and limit the availability of services in the market.
Saudi Arabia Bioanalytical Testing Services CRO Market Future Outlook
The future of the bioanalytical testing services market in Saudi Arabia appears promising, driven by advancements in technology and increasing collaboration between public and private sectors. The integration of digital technologies, such as AI and machine learning, is expected to enhance testing efficiency and accuracy. Additionally, the focus on personalized medicine will likely create new avenues for growth, as tailored therapies require specialized testing services to ensure optimal patient outcomes.
Market Opportunities
- Growth in Personalized Medicine:The personalized medicine market in Saudi Arabia is projected to reach $1.2 billion in future, driven by advancements in genomics and biotechnology. This growth presents significant opportunities for bioanalytical testing services, as tailored therapies require precise testing to ensure efficacy and safety, thereby expanding the market for specialized CROs.
- Increasing Investment in R&D:Saudi Arabia's investment in research and development is expected to exceed $2.5 billion in future, focusing on innovative healthcare solutions. This influx of funding will likely boost the demand for bioanalytical testing services, as increased R&D activities necessitate comprehensive testing to support new drug development and regulatory submissions, creating a favorable environment for CROs.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Test Type | **DMPK / ADME testing** **Pharmacokinetics (PK) and pharmacodynamics (PD)** **Bioavailability and bioequivalence (BA/BE) studies** **Biomarker and immunogenicity testing** **Virology and serology testing** Others |
| By Molecule Type | **Small molecules** **Large molecules (biologics and biosimilars)** Others |
| By Therapeutic Area | **Oncology** **Cardiovascular** **Neurology** **Infectious diseases** Others |
| By Workflow | **Sample preparation** **Sample analysis** Others |
| By End-User | **Pharmaceutical companies** **Biotechnology firms** **Contract research organizations** **Academic and research institutions** Others |
| By Technology | **Mass spectrometry and chromatography-based assays** **Ligand-binding and immunoassays** **Molecular and cell-based assays** Others |
| By Region | **Central Region** **Eastern Region** **Western Region** **Southern Region** |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Contract Manufacturing Organizations (CMOs)
Biomarker and Diagnostic Companies
Pharmaceutical Supply Chain Managers
Players Mentioned in the Report:
**Labcorp**
**Eurofins Scientific**
**Syneos Health**
**IQVIA**
**Charles River Laboratories**
**Wuxi AppTec**
**SGS**
**PRA Health Sciences / ICON**
**Medpace**
**Q2 Solutions**
**Celerion**
**PPD (Thermo Fisher Scientific)**
**Local and regional Saudi-based CROs**
**Other niche bioanalytical service providers**
**Emerging GCC-based specialty labs**
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Bioanalytical Testing Services CRO Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Bioanalytical Testing Services CRO Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Bioanalytical Testing Services CRO Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for **pharmaceutical development**
3.1.2 Rise in **biologics and biosimilars** research
3.1.3 Expansion of **clinical trials** in the region
3.1.4 Advancements in **analytical technologies**
3.2 Market Challenges
3.2.1 Stringent **regulatory requirements**
3.2.2 High **operational costs**
3.2.3 Limited **skilled workforce**
3.2.4 **Market competition** from established players
3.3 Market Opportunities
3.3.1 Growth in **personalized medicine**
3.3.2 Increasing investment in **R&D**
3.3.3 Collaborations with **academic institutions**
3.3.4 Expansion into **emerging markets**
3.4 Market Trends
3.4.1 Shift towards **outsource testing services**
3.4.2 Adoption of **digital technologies** in testing
3.4.3 Focus on **sustainability** in operations
3.4.4 Increasing demand for **integrated services**
3.5 Government Regulation
3.5.1 Implementation of **GMP standards**
3.5.2 Compliance with **ISO certifications**
3.5.3 Regulatory support for **clinical trials**
3.5.4 Policies promoting **biotechnology** advancements
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Bioanalytical Testing Services CRO Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Bioanalytical Testing Services CRO Market Segmentation
8.1 By Test Type
8.1.1 **DMPK / ADME testing**
8.1.2 **Pharmacokinetics (PK) and pharmacodynamics (PD)**
8.1.3 **Bioavailability and bioequivalence (BA/BE) studies**
8.1.4 **Biomarker and immunogenicity testing**
8.1.5 **Virology and serology testing**
8.1.6 Others
8.2 By Molecule Type
8.2.1 **Small molecules**
8.2.2 **Large molecules (biologics and biosimilars)**
8.2.3 Others
8.3 By Therapeutic Area
8.3.1 **Oncology**
8.3.2 **Cardiovascular**
8.3.3 **Neurology**
8.3.4 **Infectious diseases**
8.3.5 Others
8.4 By Workflow
8.4.1 **Sample preparation**
8.4.2 **Sample analysis**
8.4.3 Others
8.5 By End-User
8.5.1 **Pharmaceutical companies**
8.5.2 **Biotechnology firms**
8.5.3 **Contract research organizations**
8.5.4 **Academic and research institutions**
8.5.5 Others
8.6 By Technology
8.6.1 **Mass spectrometry and chromatography-based assays**
8.6.2 **Ligand-binding and immunoassays**
8.6.3 **Molecular and cell-based assays**
8.6.4 Others
8.7 By Region
8.7.1 **Central Region**
8.7.2 **Eastern Region**
8.7.3 **Western Region**
8.7.4 **Southern Region**
9. Saudi Arabia Bioanalytical Testing Services CRO Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Saudi Arabia Revenue (USD million)
9.2.4 3-Year Revenue CAGR (%)
9.2.5 EBITDA Margin (%)
9.2.6 R&D Spend as % of Revenue
9.2.7 Share of Saudi Arabia in Global Revenue (%)
9.2.8 Active Bioanalytical Projects in Saudi Arabia (Number)
9.2.9 Key Saudi / GCC Clients (Number)
9.2.10 Average Contract Value (USD, per project)
9.2.11 Proposal Win Rate (%)
9.2.12 On-time Delivery Rate (%)
9.2.13 Quality / Audit Findings per Inspection (Number)
9.2.14 Client Retention Rate (%)
9.2.15 Net Promoter Score (NPS)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 **Labcorp**
9.5.2 **Eurofins Scientific**
9.5.3 **Syneos Health**
9.5.4 **IQVIA**
9.5.5 **Charles River Laboratories**
9.5.6 **Wuxi AppTec**
9.5.7 **SGS**
9.5.8 **PRA Health Sciences / ICON**
9.5.9 **Medpace**
9.5.10 **Q2 Solutions**
9.5.11 **Celerion**
9.5.12 **PPD (Thermo Fisher Scientific)**
9.5.13 **Local and regional Saudi-based CROs**
9.5.14 **Other niche bioanalytical service providers**
9.5.15 **Emerging GCC-based specialty labs**
10. Saudi Arabia Bioanalytical Testing Services CRO Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 **Budget allocation trends**
10.1.2 **Decision-making processes**
10.1.3 **Preferred service providers**
10.1.4 **Contracting practices**
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 **Investment in laboratory facilities**
10.2.2 **Funding for research initiatives**
10.2.3 **Expenditure on technology upgrades**
10.2.4 **Partnerships with CROs**
10.3 Pain Point Analysis by End-User Category
10.3.1 **Regulatory compliance challenges**
10.3.2 **Cost management issues**
10.3.3 **Quality assurance concerns**
10.3.4 **Timeliness of results**
10.4 User Readiness for Adoption
10.4.1 **Awareness of bioanalytical services**
10.4.2 **Training and support needs**
10.4.3 **Integration with existing systems**
10.4.4 **Feedback mechanisms**
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 **Performance metrics**
10.5.2 **Scalability of services**
10.5.3 **Long-term partnerships**
10.5.4 **Future service needs**
11. Saudi Arabia Bioanalytical Testing Services CRO Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Tracking
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regulatory bodies such as the Saudi Food and Drug Authority (SFDA)
- Review of published market studies and white papers on bioanalytical testing trends in Saudi Arabia
- Examination of academic journals and publications focusing on advancements in bioanalytical methodologies
Primary Research
- Interviews with laboratory directors and managers at leading bioanalytical testing facilities
- Surveys targeting pharmaceutical companies and biotech firms utilizing bioanalytical services
- Field interviews with regulatory affairs specialists to understand compliance requirements
Validation & Triangulation
- Cross-validation of findings through multiple data sources including industry reports and expert opinions
- Triangulation of market data with insights from academic research and industry conferences
- Sanity checks conducted through expert panel reviews comprising industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the bioanalytical testing market size based on national healthcare expenditure and R&D investment
- Segmentation of the market by service type, including pharmacokinetics, toxicology, and biomarker analysis
- Incorporation of government initiatives promoting biotechnology and pharmaceutical research
Bottom-up Modeling
- Collection of service pricing data from leading bioanalytical testing laboratories
- Volume estimates based on the number of clinical trials and research projects in Saudi Arabia
- Calculation of market size using a volume x price approach for each service segment
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating factors such as drug development timelines and regulatory changes
- Scenario modeling based on potential shifts in healthcare policies and funding for biotechnology
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Bioanalytical Services | 100 | Clinical Research Managers, Regulatory Affairs Specialists |
| Biotechnology Testing Services | 80 | Laboratory Directors, R&D Scientists |
| Contract Research Organizations (CROs) | 70 | Business Development Managers, Project Managers |
| Academic Research Institutions | 60 | Principal Investigators, Research Coordinators |
| Regulatory Compliance and Quality Assurance | 90 | Quality Control Managers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Saudi Arabia Bioanalytical Testing Services CRO Market?
The Saudi Arabia Bioanalytical Testing Services CRO Market is valued at approximately USD 65 million, reflecting a five-year historical analysis. This growth is driven by increasing drug development demands and regulatory compliance needs.