Region:Middle East
Author(s):Shubham
Product Code:KRAD3477
Pages:89
Published On:November 2025
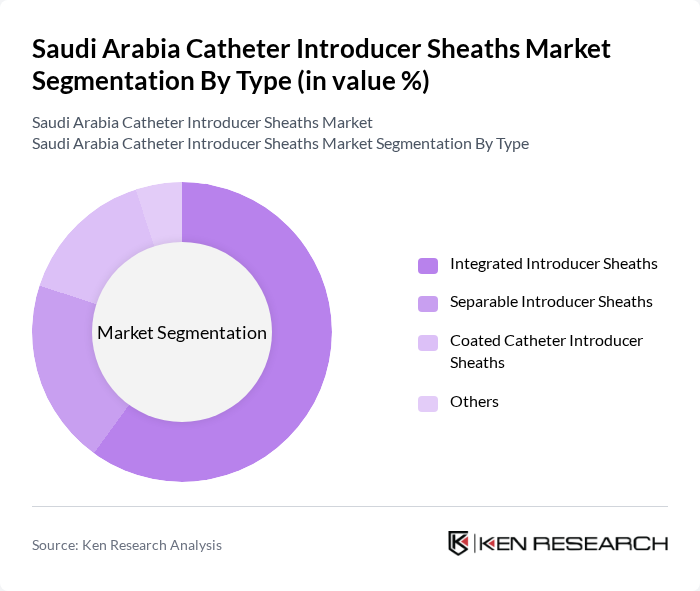
By Type:The market is segmented into Integrated Introducer Sheaths, Separable Introducer Sheaths, Coated Catheter Introducer Sheaths, and Others. Integrated Introducer Sheaths are the most widely adopted due to their all-in-one design, ease of use, and procedural efficiency, particularly in cardiovascular and interventional radiology procedures. Separable Introducer Sheaths are preferred in specialized cases requiring device customization or staged interventions. The demand for Coated Catheter Introducer Sheaths is rising, driven by their improved biocompatibility and reduced infection risk, especially in patients with higher susceptibility to vascular complications .
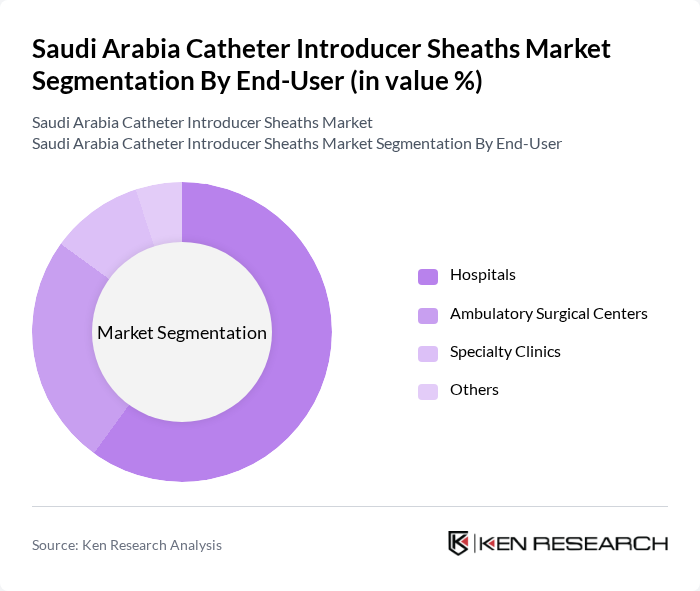
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Specialty Clinics, and Others. Hospitals represent the largest end-user segment, attributed to the high volume of cardiovascular and interventional procedures and the need for advanced vascular access devices. Ambulatory Surgical Centers are experiencing increased adoption due to the shift toward outpatient minimally invasive interventions. Specialty Clinics, particularly those focused on cardiology and vascular medicine, contribute to market diversity by catering to targeted patient populations .
The Saudi Arabia Catheter Introducer Sheaths Market is characterized by a dynamic mix of regional and international players. Leading participants such as Boston Scientific Corporation, Medtronic plc, Abbott Laboratories, B. Braun Melsungen AG, Terumo Corporation, Cook Medical LLC, Johnson & Johnson (Biosense Webster), Cardinal Health, Inc., Smiths Medical (ICU Medical, Inc.), Teleflex Incorporated, ConvaTec Group PLC, Owens & Minor, Inc. (formerly Halyard Health), Merit Medical Systems, Inc., AngioDynamics, Inc., Medline Industries, LP contribute to innovation, geographic expansion, and service delivery in this space.
The future of the catheter introducer sheaths market in Saudi Arabia appears promising, driven by increasing healthcare investments and a growing focus on minimally invasive procedures. As the healthcare infrastructure expands, particularly in urban areas, the demand for advanced medical devices is expected to rise. Additionally, the integration of telemedicine and remote monitoring technologies will likely enhance patient care, further propelling the adoption of catheter introducer sheaths in various medical settings.
| Segment | Sub-Segments |
|---|---|
| By Type | Integrated Introducer Sheaths Separable Introducer Sheaths Coated Catheter Introducer Sheaths Others |
| By End-User | Hospitals Ambulatory Surgical Centers Specialty Clinics Others |
| By Material | Polyurethane Silicone PVC Others |
| By Size | Fr Fr Fr Others |
| By Application | Cardiovascular Procedures Urological Procedures Gastrointestinal Procedures Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiovascular Procedures | 50 | Cardiologists, Interventional Radiologists |
| General Surgery Applications | 40 | General Surgeons, Surgical Nurses |
| Emergency Medical Services | 30 | Emergency Room Physicians, Paramedics |
| Hospital Procurement Departments | 45 | Procurement Managers, Supply Chain Coordinators |
| Medical Device Distributors | 25 | Sales Representatives, Distribution Managers |
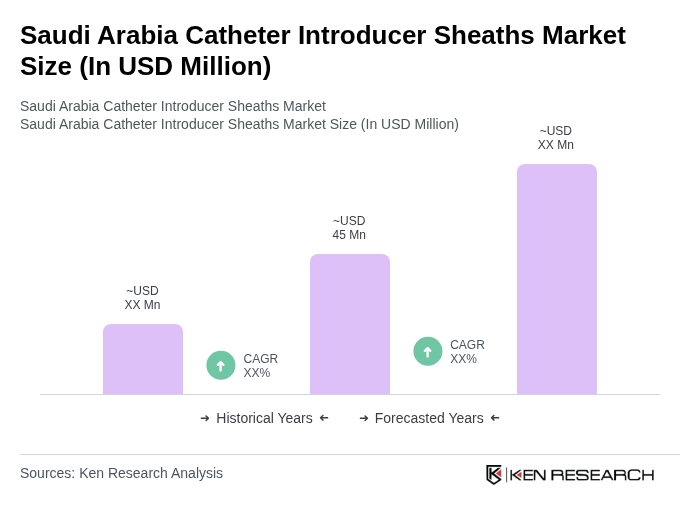
The Saudi Arabia Catheter Introducer Sheaths Market is valued at approximately USD 45 million, reflecting a five-year historical analysis. This growth is driven by factors such as the rising prevalence of cardiovascular diseases and advancements in minimally invasive medical technologies.