About the Report
Base Year 2024Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Overview
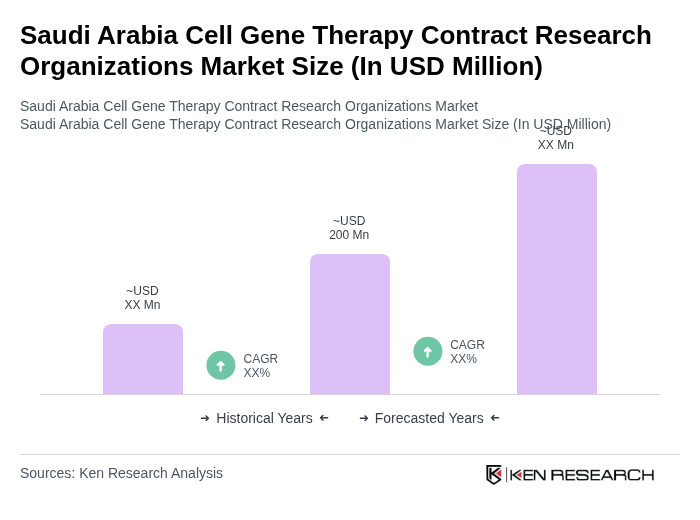
- The Saudi Arabia Cell Gene Therapy Contract Research Organizations Market is valued at USD 200 million, based on a five-year historical analysis. This market size is supported by multiple industry reports and reflects the combined value of cell and gene therapy services, clinical trials, and third-party logistics within Saudi Arabia. Growth is primarily driven by increasing investments in biotechnology, rapid advancements in gene editing technologies such as CRISPR, and a rising prevalence of genetic disorders and cancers. Additional drivers include government initiatives under Vision 2030, which prioritize healthcare infrastructure expansion, local biomanufacturing, and research and development in advanced therapies.
- Key cities dominating the market includeRiyadh,Jeddah, andDammam. Riyadh serves as the capital and central hub for healthcare and research institutions, hosting leading hospitals, universities, and clinical trial centers. Jeddah and Dammam are significant due to their advanced medical facilities, proximity to major pharmaceutical companies, and concentration of skilled professionals. These cities benefit from collaborative networks that foster innovation in cell and gene therapy, supported by regional government investment and international partnerships.
- In 2023, theSaudi Food and Drug Authority (SFDA)implemented the “Gene Therapy Products Registration Guidelines,” which streamline the approval process for gene therapy products. This regulation enhances patient access to innovative treatments while ensuring safety and efficacy. The SFDA guidelines require comprehensive clinical trial protocols, robust post-marketing surveillance, and detailed risk management plans for all gene therapy products, establishing high operational standards for clinical research organizations and sponsors.
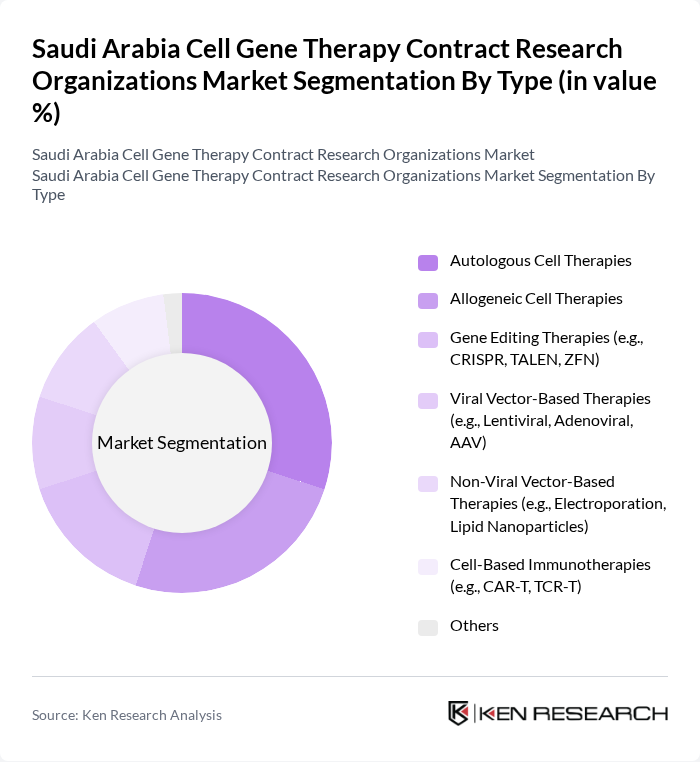
Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Segmentation
By Type:The market is segmented into various types of therapies, including Autologous Cell Therapies, Allogeneic Cell Therapies, Gene Editing Therapies, Viral Vector-Based Therapies, Non-Viral Vector-Based Therapies, Cell-Based Immunotherapies, and Others. Among these,Autologous Cell Therapiesare gaining significant traction due to their personalized approach, which enhances treatment efficacy and minimizes the risk of rejection. The increasing prevalence of chronic diseases, such as sickle cell disease and thalassemia, and advancements in gene editing and delivery technologies are driving demand for these therapies.
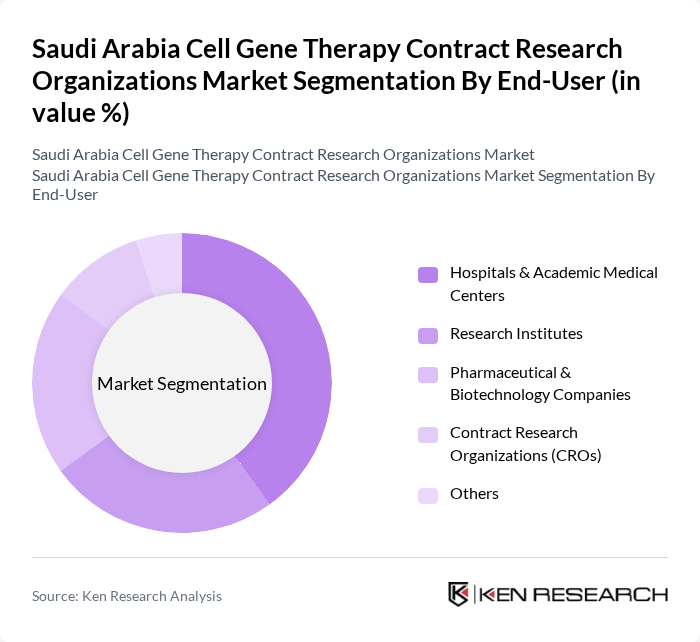
By End-User:The market is categorized by end-users, including Hospitals & Academic Medical Centers, Research Institutes, Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs), and Others.Hospitals and Academic Medical Centersare the leading end-users due to their direct involvement in patient care, clinical trials, and translational research. The increasing collaboration between hospitals, research institutions, and industry partners is further enhancing the adoption of gene therapies in clinical settings, with government incentives supporting these partnerships.
Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Competitive Landscape
The Saudi Arabia Cell Gene Therapy Contract Research Organizations Market is characterized by a dynamic mix of regional and international players. Leading participants such as King Faisal Specialist Hospital & Research Centre, Riyadh Pharma (Saudi Pharmaceutical Industries & Medical Appliances Corporation - SPIMACO), SaudiVax Ltd., Tabuk Pharmaceuticals Manufacturing Company, Sudair Pharma Company, National Center for Biotechnology (King Abdulaziz City for Science and Technology - KACST), Gulf Pharmaceutical Industries (Julphar), Advanced Cell Therapy Center (ACTC) - King Abdullah International Medical Research Center, IQVIA Saudi Arabia, Syneos Health Saudi Arabia, Labcorp Drug Development (Covance) Saudi Arabia, Charles River Laboratories Saudi Arabia, ICON plc Saudi Arabia, Medpace Saudi Arabia, Thermo Fisher Scientific Saudi Arabia contribute to innovation, geographic expansion, and service delivery in this space.
Sources:
Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Genetic Disorders:The prevalence of genetic disorders in Saudi Arabia is rising, with approximately 1 in 4 children affected by genetic conditions, according to the Saudi Ministry of Health. This alarming statistic drives demand for innovative therapies, including gene therapy. The increasing number of patients needing treatment creates a robust market for contract research organizations (CROs) specializing in cell and gene therapies, fostering growth in this sector.
- Advancements in Gene Editing Technologies:The rapid advancements in gene editing technologies, such as CRISPR-Cas9, have revolutionized the field of gene therapy. In future, the global market for gene editing is projected to reach $5.6 billion, with significant contributions from Saudi Arabia's biotechnology sector. These innovations enable more precise and effective treatments, attracting investments and enhancing the capabilities of local CROs to conduct cutting-edge research and clinical trials.
- Supportive Government Initiatives for Healthcare Innovation:The Saudi government has committed over $1 billion to healthcare innovation as part of its Vision 2030 initiative. This funding supports research and development in biotechnology, including gene therapy. The establishment of the National Biotechnology Strategy aims to foster collaboration between public and private sectors, creating a conducive environment for CROs to thrive and innovate in gene therapy solutions.
Market Challenges
- High Cost of Gene Therapy Development:The development of gene therapies is notoriously expensive, with costs often exceeding $1 million per patient. This financial burden poses a significant challenge for CROs in Saudi Arabia, limiting their ability to conduct extensive research and clinical trials. The high costs can deter investment and slow the pace of innovation, hindering the overall growth of the gene therapy market in the region.
- Regulatory Hurdles and Compliance Issues:Navigating the regulatory landscape for gene therapies in Saudi Arabia can be complex and time-consuming. The Saudi Food and Drug Authority (SFDA) has stringent guidelines that can delay the approval process for new therapies. In future, it is estimated that regulatory delays could extend timelines by up to 18 months, creating challenges for CROs seeking to bring innovative treatments to market efficiently.
Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Future Outlook
The future of the cell gene therapy market in Saudi Arabia appears promising, driven by technological advancements and increased funding. As the government continues to support biotechnology initiatives, CROs are likely to expand their capabilities and engage in more clinical trials. The integration of artificial intelligence in research processes will enhance efficiency and precision, while patient-centric approaches will shape treatment paradigms, ensuring that therapies are tailored to individual needs and improving overall healthcare outcomes.
Market Opportunities
- Expansion of Clinical Trials in Gene Therapy:The increasing number of clinical trials in gene therapy presents a significant opportunity for CROs. In future, Saudi Arabia is expected to host over 50 new clinical trials, focusing on innovative gene therapies. This expansion will enhance the local research landscape and attract international collaborations, positioning Saudi Arabia as a hub for gene therapy research.
- Development of Personalized Medicine Solutions:The growing demand for personalized medicine solutions is another key opportunity. With advancements in genomics, Saudi Arabia's CROs can develop tailored therapies that address specific genetic profiles. This trend is expected to drive market growth, as personalized treatments are increasingly recognized for their effectiveness in managing complex genetic disorders.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Autologous Cell Therapies Allogeneic Cell Therapies Gene Editing Therapies (e.g., CRISPR, TALEN, ZFN) Viral Vector-Based Therapies (e.g., Lentiviral, Adenoviral, AAV) Non-Viral Vector-Based Therapies (e.g., Electroporation, Lipid Nanoparticles) Cell-Based Immunotherapies (e.g., CAR-T, TCR-T) Others |
| By End-User | Hospitals & Academic Medical Centers Research Institutes Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) Others |
| By Application | Oncology (Solid Tumors, Hematological Malignancies) Rare Genetic Disorders (e.g., Sickle Cell Disease, Thalassemia) Cardiovascular Diseases Infectious Diseases Neurological Disorders Others |
| By Delivery Method | In Vivo Delivery Ex Vivo Delivery Others |
| By Region | Central Region (including Riyadh) Eastern Region (including Dammam, Khobar) Western Region (including Jeddah, Makkah) Southern Region Northern Region |
| By Research Phase | Preclinical Phase I Clinical Trials Phase II Clinical Trials Phase III Clinical Trials Commercialization Others |
| By Funding Source | Government Grants & Initiatives Private Investments Venture Capital Corporate Funding International Collaborations Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Saudi Food and Drug Authority, Ministry of Health)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations (CROs)
Healthcare Providers and Hospitals
Biomanufacturing Facilities
Health Insurance Companies
Industry Associations and Advocacy Groups
Players Mentioned in the Report:
King Faisal Specialist Hospital & Research Centre
Riyadh Pharma (Saudi Pharmaceutical Industries & Medical Appliances Corporation - SPIMACO)
SaudiVax Ltd.
Tabuk Pharmaceuticals Manufacturing Company
Sudair Pharma Company
National Center for Biotechnology (King Abdulaziz City for Science and Technology - KACST)
Gulf Pharmaceutical Industries (Julphar)
Advanced Cell Therapy Center (ACTC) - King Abdullah International Medical Research Center
IQVIA Saudi Arabia
Syneos Health Saudi Arabia
Labcorp Drug Development (Covance) Saudi Arabia
Charles River Laboratories Saudi Arabia
ICON plc Saudi Arabia
Medpace Saudi Arabia
Thermo Fisher Scientific Saudi Arabia
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of genetic disorders
3.1.2 Advancements in gene editing technologies
3.1.3 Rising investment in biotechnology research
3.1.4 Supportive government initiatives for healthcare innovation
3.2 Market Challenges
3.2.1 High cost of gene therapy development
3.2.2 Regulatory hurdles and compliance issues
3.2.3 Limited awareness among healthcare providers
3.2.4 Shortage of skilled professionals in the field
3.3 Market Opportunities
3.3.1 Expansion of clinical trials in gene therapy
3.3.2 Collaborations with international research organizations
3.3.3 Development of personalized medicine solutions
3.3.4 Increasing demand for regenerative medicine
3.4 Market Trends
3.4.1 Growing focus on CAR-T cell therapies
3.4.2 Integration of AI in gene therapy research
3.4.3 Rise of patient-centric treatment approaches
3.4.4 Increased funding for gene therapy startups
3.5 Government Regulation
3.5.1 Implementation of the National Biotechnology Strategy
3.5.2 Establishment of regulatory frameworks for gene therapies
3.5.3 Guidelines for clinical trial approvals
3.5.4 Policies promoting public-private partnerships in healthcare
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Segmentation
8.1 By Type
8.1.1 Autologous Cell Therapies
8.1.2 Allogeneic Cell Therapies
8.1.3 Gene Editing Therapies (e.g., CRISPR, TALEN, ZFN)
8.1.4 Viral Vector-Based Therapies (e.g., Lentiviral, Adenoviral, AAV)
8.1.5 Non-Viral Vector-Based Therapies (e.g., Electroporation, Lipid Nanoparticles)
8.1.6 Cell-Based Immunotherapies (e.g., CAR-T, TCR-T)
8.1.7 Others
8.2 By End-User
8.2.1 Hospitals & Academic Medical Centers
8.2.2 Research Institutes
8.2.3 Pharmaceutical & Biotechnology Companies
8.2.4 Contract Research Organizations (CROs)
8.2.5 Others
8.3 By Application
8.3.1 Oncology (Solid Tumors, Hematological Malignancies)
8.3.2 Rare Genetic Disorders (e.g., Sickle Cell Disease, Thalassemia)
8.3.3 Cardiovascular Diseases
8.3.4 Infectious Diseases
8.3.5 Neurological Disorders
8.3.6 Others
8.4 By Delivery Method
8.4.1 In Vivo Delivery
8.4.2 Ex Vivo Delivery
8.4.3 Others
8.5 By Region
8.5.1 Central Region (including Riyadh)
8.5.2 Eastern Region (including Dammam, Khobar)
8.5.3 Western Region (including Jeddah, Makkah)
8.5.4 Southern Region
8.5.5 Northern Region
8.6 By Research Phase
8.6.1 Preclinical
8.6.2 Phase I Clinical Trials
8.6.3 Phase II Clinical Trials
8.6.4 Phase III Clinical Trials
8.6.5 Commercialization
8.6.6 Others
8.7 By Funding Source
8.7.1 Government Grants & Initiatives
8.7.2 Private Investments
8.7.3 Venture Capital
8.7.4 Corporate Funding
8.7.5 International Collaborations
8.7.6 Others
9. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, Small)
9.2.3 Revenue (USD Million, Latest Year)
9.2.4 Revenue Growth Rate (CAGR %)
9.2.5 Number of Ongoing/Completed Clinical Trials
9.2.6 Therapeutic Area Coverage (e.g., Oncology, Rare Diseases)
9.2.7 Market Penetration Rate (Share of Saudi Arabia Cell/Gene Therapy Trials)
9.2.8 Customer Retention Rate (%)
9.2.9 R&D Investment Ratio (% of Revenue)
9.2.10 Regulatory Compliance Rate (e.g., SFDA, GCP adherence)
9.2.11 Partnership and Collaboration Index (Number of Active Partnerships)
9.2.12 Innovation Index (Patents Filed, New Technologies Adopted)
9.2.13 Time-to-Approval for Clinical Trials (Months)
9.2.14 Local Manufacturing/Processing Capability
9.2.15 Pricing Strategy (Premium, Value-Based, Cost-Plus)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 King Faisal Specialist Hospital & Research Centre
9.5.2 Riyadh Pharma (Saudi Pharmaceutical Industries & Medical Appliances Corporation - SPIMACO)
9.5.3 SaudiVax Ltd.
9.5.4 Tabuk Pharmaceuticals Manufacturing Company
9.5.5 Sudair Pharma Company
9.5.6 National Center for Biotechnology (King Abdulaziz City for Science and Technology - KACST)
9.5.7 Gulf Pharmaceutical Industries (Julphar)
9.5.8 Advanced Cell Therapy Center (ACTC) - King Abdullah International Medical Research Center
9.5.9 IQVIA Saudi Arabia
9.5.10 Syneos Health Saudi Arabia
9.5.11 Labcorp Drug Development (Covance) Saudi Arabia
9.5.12 Charles River Laboratories Saudi Arabia
9.5.13 ICON plc Saudi Arabia
9.5.14 Medpace Saudi Arabia
9.5.15 Thermo Fisher Scientific Saudi Arabia
10. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health
10.1.2 Ministry of Education
10.1.3 Ministry of Industry and Mineral Resources
10.1.4 Ministry of Investment
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Infrastructure
10.2.2 Funding for Research and Development
10.2.3 Expenditure on Clinical Trials
10.2.4 Budget Allocation for Gene Therapy
10.3 Pain Point Analysis by End-User Category
10.3.1 Hospitals
10.3.2 Research Institutions
10.3.3 Pharmaceutical Companies
10.3.4 Biotechnology Firms
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training and Education Needs
10.4.3 Infrastructure Readiness
10.4.4 Financial Preparedness
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Treatment Outcomes
10.5.2 Long-term Cost Savings
10.5.3 Expansion into New Therapeutic Areas
10.5.4 Patient Satisfaction Metrics
11. Saudi Arabia Cell Gene Therapy Contract Research Organizations Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Planning
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare and biotechnology associations in Saudi Arabia
- Review of published articles and white papers on cell gene therapy advancements
- Examination of regulatory frameworks and guidelines from the Saudi Food and Drug Authority (SFDA)
Primary Research
- Interviews with executives from leading Contract Research Organizations (CROs) specializing in gene therapy
- Surveys targeting clinical researchers and healthcare professionals involved in gene therapy trials
- Field interviews with regulatory affairs specialists to understand compliance challenges
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including market reports and expert opinions
- Triangulation of quantitative data with qualitative insights from industry experts
- Sanity checks conducted through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the overall healthcare expenditure in Saudi Arabia as a baseline for market potential
- Segmentation of the market by therapeutic areas and types of gene therapies offered
- Incorporation of government initiatives and funding for biotechnology research and development
Bottom-up Modeling
- Collection of data on the number of ongoing clinical trials and their respective phases
- Estimation of revenue generated by CROs based on service offerings and pricing models
- Volume and cost analysis of gene therapy products and services provided by CROs
Forecasting & Scenario Analysis
- Utilization of historical growth rates in the biotechnology sector to project future market trends
- Scenario modeling based on potential regulatory changes and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Gene Therapy Clinical Trials | 100 | Clinical Research Coordinators, Principal Investigators |
| Regulatory Compliance in Gene Therapy | 60 | Regulatory Affairs Managers, Quality Assurance Specialists |
| Market Trends in Cell Gene Therapy | 90 | Biotech Analysts, Market Research Professionals |
| Funding and Investment in Gene Therapy | 50 | Venture Capitalists, Investment Analysts |
| Patient Recruitment Strategies | 70 | Patient Advocacy Group Leaders, Clinical Trial Recruiters |
Frequently Asked Questions
What is the current value of the Saudi Arabia Cell Gene Therapy Contract Research Organizations Market?
The Saudi Arabia Cell Gene Therapy Contract Research Organizations Market is valued at approximately USD 200 million, reflecting the combined value of cell and gene therapy services, clinical trials, and third-party logistics within the country.