Region:Middle East
Author(s):Shubham
Product Code:KRAD3515
Pages:100
Published On:November 2025
By Type:The market is segmented into various types of software that cater to different aspects of clinical trials. The primary subsegments includePatient Matching Software,Investigator Matching Software,Site Selection Software,Data Management Software, andOthers. Each of these subsegments plays a crucial role in enhancing the efficiency and effectiveness of clinical trials. Patient Matching Software leverages AI and big data analytics to identify eligible patients, while Investigator Matching Software connects qualified investigators with appropriate studies. Site Selection Software optimizes trial site identification, and Data Management Software ensures secure and compliant handling of trial data.
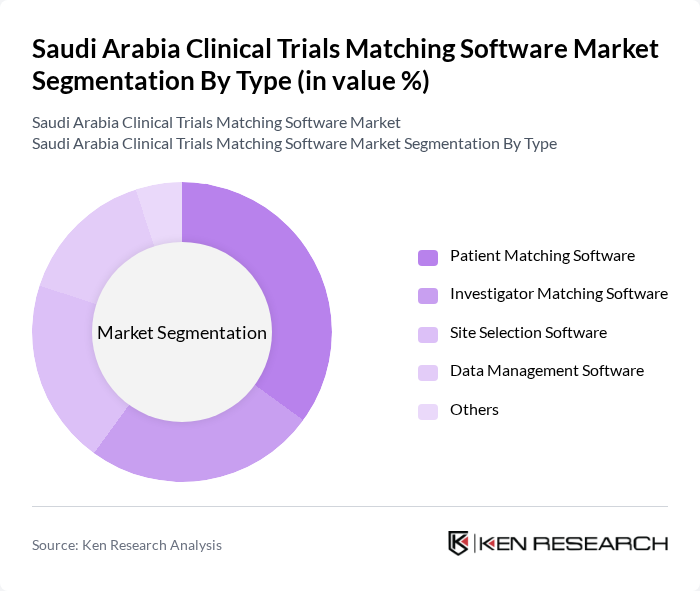
ThePatient Matching Softwaresubsegment is currently dominating the market due to its critical role in efficiently connecting patients with suitable clinical trials. This software utilizes advanced algorithms and data analytics to identify eligible patients based on specific criteria, significantly reducing the time and effort required for recruitment. The increasing focus on personalized medicine and patient-centric approaches in clinical research further drives the demand for such solutions. As a result, Patient Matching Software is expected to maintain its leadership position in the market.
By End-User:The market is segmented based on the end-users of clinical trials matching software, which includesPharmaceutical Companies,Clinical Research Organizations (CROs),Hospitals and Healthcare Providers,Academic Institutions,Government Health Agencies, andOthers. Each of these end-users has unique requirements and contributes to the overall demand for clinical trials matching software. Pharmaceutical companies and CROs require robust solutions for patient recruitment and trial management, while hospitals and academic institutions focus on compliance and data security. Government health agencies utilize these platforms to monitor and regulate clinical research activities.
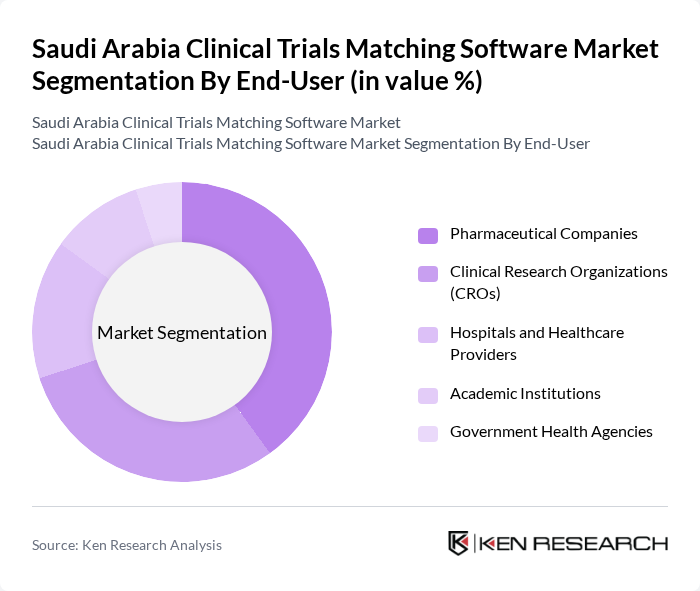
Pharmaceutical Companiesrepresent the leading end-user segment, driven by their need for efficient patient recruitment and trial management processes. These companies are increasingly investing in advanced software solutions to streamline their clinical trial operations, enhance data accuracy, and comply with regulatory requirements. The growing emphasis on drug development and the need for faster time-to-market further solidify the dominance of Pharmaceutical Companies in the clinical trials matching software market.
The Saudi Arabia Clinical Trials Matching Software Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medidata Solutions, Oracle Health Sciences, Veeva Systems, Parexel International, Labcorp Drug Development (formerly Covance), CRF Health (now part of Signant Health), BioClinica (now part of ERT), eClinical Solutions, Medpace, Syneos Health, Clinipace (now part of dMed-Clinipace), PPD (now part of Thermo Fisher Scientific), ICON plc, WCG Clinical, TrialSpark, myTomorrows, Signant Health, ERT, Saudi Clinical Trials Registry (SCTR), King Faisal Specialist Hospital & Research Centre (KFSH&RC) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the Saudi Arabia clinical trials matching software market appears promising, driven by ongoing technological advancements and supportive government policies. As the healthcare sector increasingly embraces digital transformation, the integration of AI and real-time data analytics will enhance patient engagement and streamline trial processes. Furthermore, the expansion of telemedicine services is expected to facilitate remote patient recruitment, creating new avenues for software developers to innovate and meet the evolving needs of clinical research.
| Segment | Sub-Segments |
|---|---|
| By Type | Patient Matching Software Investigator Matching Software Site Selection Software Data Management Software Others |
| By End-User | Pharmaceutical Companies Clinical Research Organizations (CROs) Hospitals and Healthcare Providers Academic Institutions Government Health Agencies Others |
| By Deployment Model | On-Premise Cloud-Based Hybrid Others |
| By Functionality | Patient Recruitment Data Analysis Reporting Compliance Management Integration with Electronic Health Records (EHR) Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Software Type | Standalone Software Integrated Software Solutions Mobile Applications Others |
| By Pricing Model | Subscription-Based One-Time License Fee Pay-Per-Use Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 60 | Clinical Research Managers, Regulatory Affairs Specialists |
| Biotechnology Firms | 40 | Product Development Managers, Clinical Operations Directors |
| Healthcare Institutions | 50 | Clinical Trial Coordinators, Research Nurses |
| Regulatory Bodies | 40 | Policy Makers, Compliance Officers |
| Technology Providers | 40 | Software Developers, IT Managers in Healthcare |
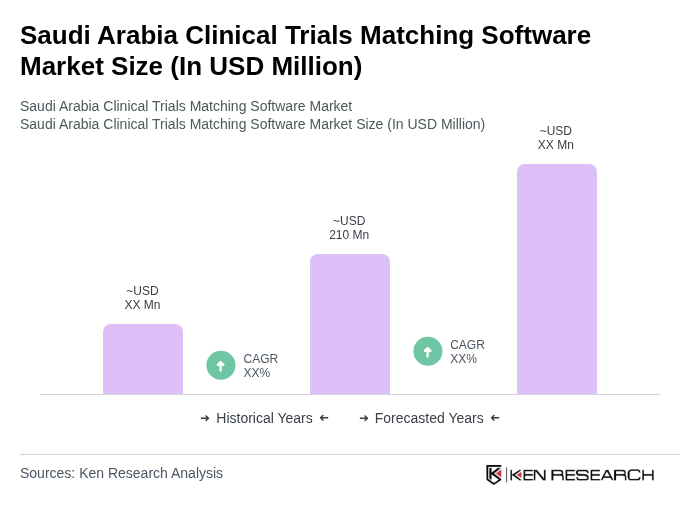
The Saudi Arabia Clinical Trials Matching Software Market is valued at approximately USD 210 million, reflecting a significant growth driven by the increasing number of clinical trials and advancements in healthcare technology.