Saudi Arabia Human Papillomavirus Vaccines Market Overview
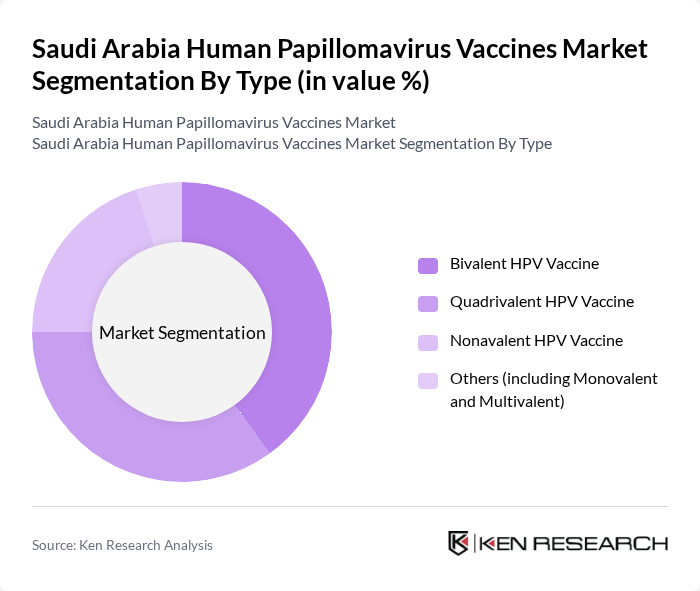
- The Saudi Arabia Human Papillomavirus Vaccines Market is valued at USD 40 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of HPV-related diseases, government health initiatives, and the rising prevalence of cervical cancer. The market is further supported by the integration of the HPV vaccine into the national immunization schedule, expansion to younger age groups, and subsidized screening programs that make preventive care more accessible. Notable trends include the launch of new vaccine formulations and clinical trials for broader protection against HPV types, as well as ongoing efforts to address vaccine hesitancy through targeted education campaigns .
- Key cities such as Riyadh, Jeddah, and Dammam continue to dominate the market due to their advanced healthcare infrastructure and higher population density. These urban centers benefit from significant investments in healthcare facilities and public health campaigns, which have contributed to increased vaccination rates and greater awareness about HPV and its associated risks .
- The Saudi Arabian government implemented a national HPV vaccination program, integrating the vaccine into the national immunization schedule in March 2022 and expanding coverage to younger age groups in September 2022. This initiative provides free vaccinations in schools and public health facilities, targeting both boys and girls to promote gender-neutral vaccination and reduce the incidence of HPV-related diseases. The program is governed by the Ministry of Health’s National Immunization Schedule (Ministry of Health, Kingdom of Saudi Arabia, 2022), which mandates HPV vaccination for adolescents and outlines operational details for coverage, compliance, and reporting .
Saudi Arabia Human Papillomavirus Vaccines Market Segmentation
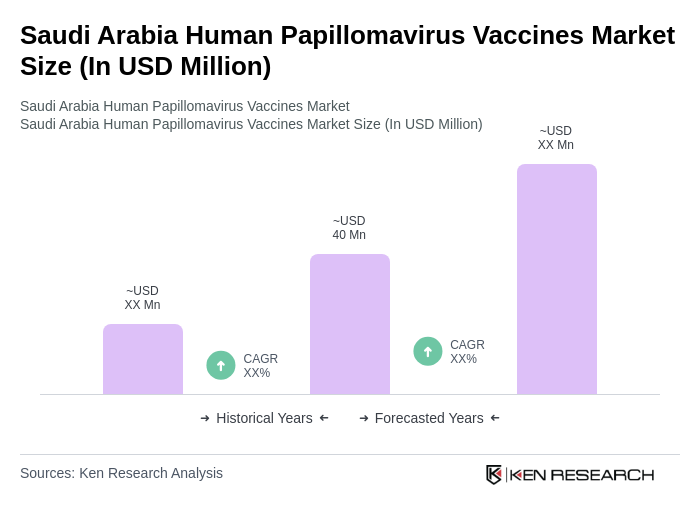
By Type:The market is segmented into Bivalent, Quadrivalent, Nonavalent, and others. The Bivalent HPV vaccine is primarily used for its effectiveness against the two most common cancer-causing HPV types (HPV-16 and HPV-18). The Quadrivalent HPV vaccine covers four types (HPV-6, 11, 16, 18), while the Nonavalent vaccine provides protection against nine types (HPV-6, 11, 16, 18, 31, 33, 45, 52, 58). The "Others" category includes Monovalent and Multivalent vaccines, though these are less commonly used in Saudi Arabia .
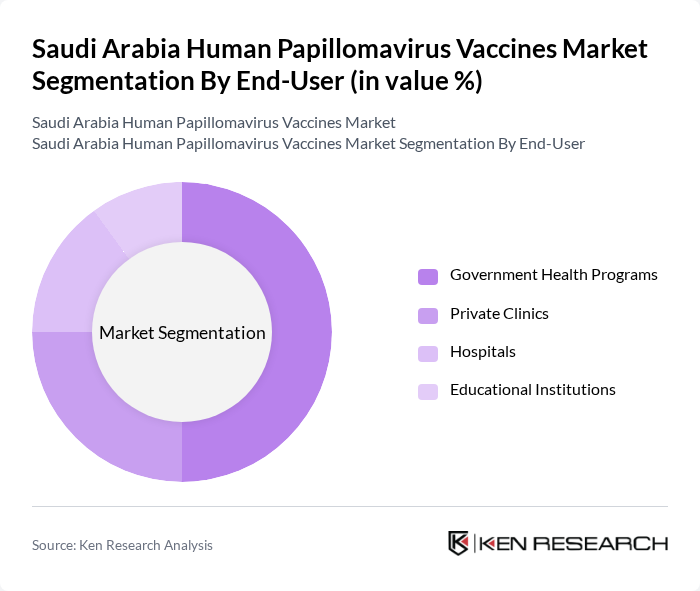
By End-User:The end-user segmentation includes Government Health Programs, Private Clinics, Hospitals, and Educational Institutions. Government health programs are the leading end-user segment, driven by national vaccination initiatives and public sector distribution. Private clinics and hospitals contribute significantly to vaccine administration, while educational institutions play a vital role in reaching adolescents through school-based vaccination campaigns .
Saudi Arabia Human Papillomavirus Vaccines Market Competitive Landscape
The Saudi Arabia Human Papillomavirus Vaccines Market is characterized by a dynamic mix of regional and international players. Leading participants such as GlaxoSmithKline plc, Merck & Co., Inc., Sanofi S.A., Pfizer Inc., AstraZeneca plc, Novartis AG, Bharat Biotech International Limited, Serum Institute of India Pvt. Ltd., BioNTech SE, Johnson & Johnson, Takeda Pharmaceutical Company Limited, CSL Limited, VBI Vaccines Inc., Inovio Pharmaceuticals, Inc., MedImmune LLC contribute to innovation, geographic expansion, and service delivery in this space.
Saudi Arabia Human Papillomavirus Vaccines Market Industry Analysis
Growth Drivers
- Increasing Awareness of HPV-Related Diseases:The awareness of HPV-related diseases has significantly increased in Saudi Arabia, with over 60% of the population now recognizing the link between HPV and cervical cancer. This heightened awareness is supported by educational campaigns from the Ministry of Health, which allocated approximately SAR 50 million in future for public health initiatives. As a result, more individuals are seeking vaccination, driving demand for HPV vaccines.
- Government Initiatives for Vaccination Programs:The Saudi government has implemented robust vaccination programs, including the introduction of HPV vaccines in school health programs. In future, the government reported administering over 1 million doses of HPV vaccines, reflecting a commitment to reducing cervical cancer rates. This initiative is part of a broader strategy to enhance public health, with an investment of SAR 200 million aimed at expanding vaccination outreach across the nation.
- Rising Healthcare Expenditure:Saudi Arabia's healthcare expenditure is projected to reach SAR 220 billion in future, reflecting a 10% increase from the previous period. This rise in spending is directed towards preventive healthcare measures, including vaccinations. The increased budget allows for better access to HPV vaccines, improved healthcare facilities, and enhanced public health campaigns, ultimately fostering a more favorable environment for HPV vaccination uptake.
Market Challenges
- Limited Public Awareness in Rural Areas:Despite overall awareness, rural areas in Saudi Arabia face significant challenges regarding HPV knowledge. Approximately 40% of individuals in these regions remain unaware of HPV and its associated risks. This lack of awareness is compounded by limited access to healthcare resources, making it difficult to implement effective vaccination programs. Addressing this gap is crucial for increasing vaccination rates in underserved populations.
- High Cost of Vaccination:The cost of HPV vaccination remains a barrier for many families, particularly in lower-income segments. The average cost of a complete vaccination series is around SAR 1,500, which can be prohibitive for many. Although government subsidies exist, they do not cover all expenses, leading to disparities in vaccination rates. This financial challenge hinders the overall effectiveness of public health initiatives aimed at reducing HPV-related diseases.
Saudi Arabia Human Papillomavirus Vaccines Market Future Outlook
The future of the HPV vaccine market in Saudi Arabia appears promising, driven by increasing government support and public health initiatives. The focus on preventive healthcare is expected to grow, with more resources allocated to vaccination programs. Additionally, advancements in vaccine technology and the integration of telehealth services for consultations and follow-ups will likely enhance accessibility. As awareness continues to rise, particularly among younger populations, the market is poised for significant growth in the coming years.
Market Opportunities
- Expansion of Vaccination Programs in Schools:Expanding HPV vaccination programs in schools presents a significant opportunity. With over 6 million students enrolled in Saudi schools, targeting this demographic can lead to increased vaccination rates. Collaborations with educational institutions can facilitate awareness and accessibility, ensuring that more children receive the vaccine at an early age, ultimately reducing HPV prevalence.
- Collaborations with International Health Organizations:Collaborating with international health organizations can enhance the effectiveness of HPV vaccination campaigns. Partnerships with entities like WHO can provide technical support and funding, enabling the implementation of best practices. Such collaborations can also facilitate knowledge sharing, leading to improved strategies for increasing vaccination coverage and addressing public health challenges in Saudi Arabia.