Region:Middle East
Author(s):Rebecca
Product Code:KRAD4967
Pages:80
Published On:December 2025
By Service Type:
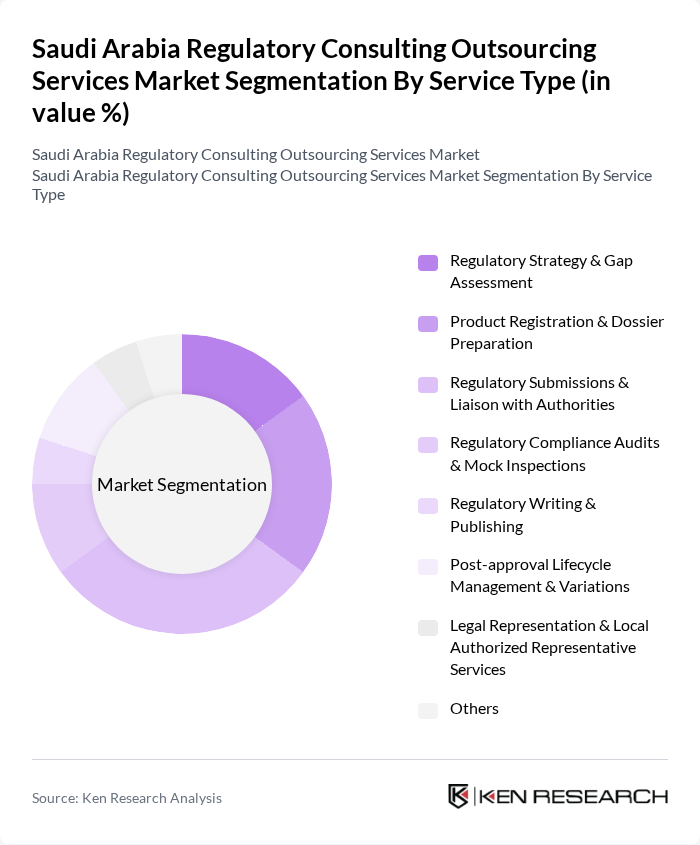
The service type segmentation includes various subsegments such as Regulatory Strategy & Gap Assessment, Product Registration & Dossier Preparation, Regulatory Submissions & Liaison with Authorities, Regulatory Compliance Audits & Mock Inspections, Regulatory Writing & Publishing, Post-approval Lifecycle Management & Variations, Legal Representation & Local Authorized Representative Services, and Others. Among these, Regulatory Submissions & Liaison with Authorities is the leading subsegment, driven by the increasing need for companies to ensure compliance with SFDA requirements across drugs, biologics, medical devices, and IVDs and to facilitate smooth interactions with regulatory bodies throughout the registration and post?approval phases. This subsegment is critical as it directly impacts the speed and success of product approvals, pricing and reimbursement decisions, and renewal activities, making it a priority for many multinational and local organizations.
By Category (Regulated Product):
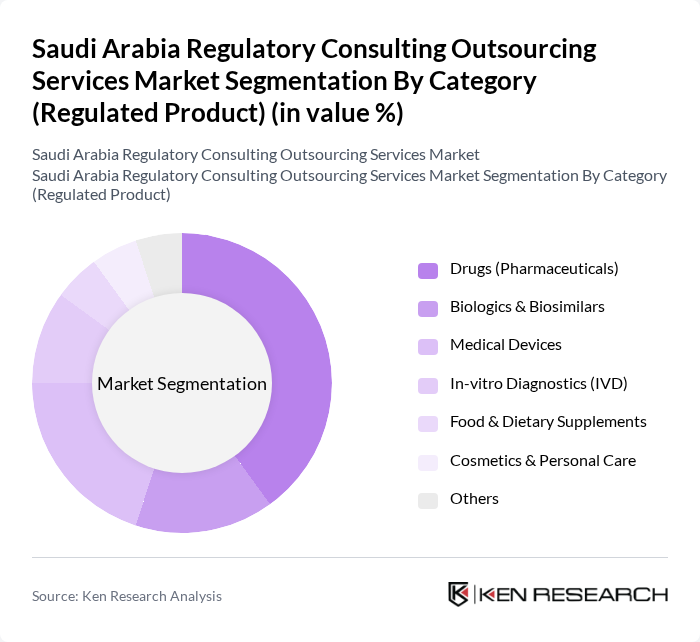
This segmentation includes Drugs (Pharmaceuticals), Biologics & Biosimilars, Medical Devices, In-vitro Diagnostics (IVD), Food & Dietary Supplements, Cosmetics & Personal Care, and Others. The Drugs (Pharmaceuticals) and Medical Devices subsegments are the most significant, reflecting the robust growth of the pharmaceutical and medtech industries in Saudi Arabia and the emphasis on local manufacturing, tendering, and importation under Vision 2030 health sector transformation. The increasing prevalence of chronic diseases, large public?sector healthcare spending, and ongoing healthcare reforms are driving demand for pharmaceutical, biologic, and device products, thereby necessitating comprehensive regulatory consulting services to ensure timely SFDA approvals, vigilance compliance, and sustained market access.
The Saudi Arabia Regulatory Consulting Outsourcing Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as IQVIA, Parexel, ICON plc, Syneos Health, ProPharma Group, PharmaLex, Labcorp, Deloitte Middle East, PwC Middle East, KPMG Saudi Arabia, EY Saudi Arabia, Saudi Food and Drug Authority (SFDA)?focused Local Regulatory Consultancies, Biopharma Middle East (BPME), Regxon Consulting, Arab Center for Consultancy & Regulatory Services contribute to innovation, geographic expansion, and service delivery in this space.
The future of the regulatory consulting outsourcing services market in Saudi Arabia appears promising, driven by ongoing regulatory reforms and a growing emphasis on compliance. As businesses increasingly adopt digital solutions, the integration of advanced technologies like AI and data analytics will enhance service delivery. Furthermore, the government's commitment to economic diversification will likely create new opportunities for consulting firms, enabling them to expand their service offerings and cater to emerging sectors.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Regulatory Strategy & Gap Assessment Product Registration & Dossier Preparation Regulatory Submissions & Liaison with Authorities Regulatory Compliance Audits & Mock Inspections Regulatory Writing & Publishing Post?approval Lifecycle Management & Variations Legal Representation & Local Authorized Representative Services Others |
| By Category (Regulated Product) | Drugs (Pharmaceuticals) Biologics & Biosimilars Medical Devices In?vitro Diagnostics (IVD) Food & Dietary Supplements Cosmetics & Personal Care Others |
| By Client Type | Multinational Pharma & MedTech Companies Regional / Local Manufacturers Contract Research Organizations (CROs) Contract Development & Manufacturing Organizations (CDMOs) Distributors & Marketing Authorization Holders Others |
| By Engagement Model | Project?based Outsourcing Long?term Managed Services / Retainer Functional / Process Outsourcing Staff Augmentation & Secondment Others |
| By Company Size | Large Enterprises Mid?sized Companies Small & Emerging Companies Start?ups |
| By Therapeutic Area (for Life Sciences) | Oncology Cardiovascular & Metabolic Disorders Infectious Diseases & Vaccines Central Nervous System (CNS) Autoimmune & Rare Diseases Others |
| By Regulatory Authority Interface | Saudi Food and Drug Authority (SFDA) Ministry of Health (MOH) Saudi Center for Disease Prevention and Control (Weqaya) Gulf Health Council & GCC Regulatory Bodies Other Saudi Government Authorities |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Regulatory Compliance | 80 | Compliance Officers, Regulatory Affairs Managers |
| Financial Services Consulting | 70 | Risk Management Directors, Compliance Analysts |
| Manufacturing Sector Regulations | 60 | Quality Assurance Managers, Operations Directors |
| Environmental Compliance Services | 50 | Sustainability Managers, Environmental Compliance Officers |
| Telecommunications Regulatory Frameworks | 40 | Regulatory Affairs Specialists, Legal Advisors |
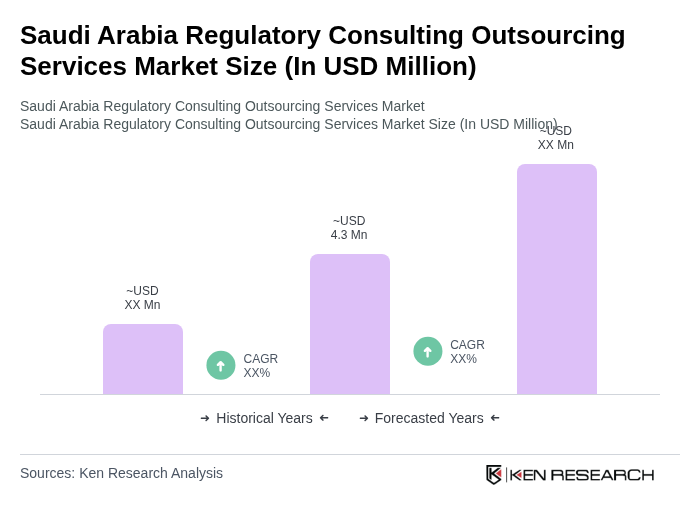
The Saudi Arabia Regulatory Consulting Outsourcing Services Market is valued at approximately USD 4.3 million, reflecting a historical analysis of regulatory consulting revenues over the past five years, driven by increasing regulatory complexities and demand for compliance.