Region:Middle East
Author(s):Dev
Product Code:KRAC1959
Pages:96
Published On:October 2025
By Service Type:The service type segmentation includes various testing services essential for ensuring the safety and efficacy of biological products. The subsegments include Bioburden Testing Services, Endotoxin Testing Services, Sterility Testing Services, Viral Clearance and Validation Services, Cell Line Authentication and Characterization Tests, Residual Host Contamination Testing Services, and Adventitious Agent Detection Testing Services. Among these, Bioburden Testing Services are currently dominating the market due to their critical role in the pharmaceutical and biopharmaceutical industries, where ensuring the absence of viable microorganisms is paramount for product safety.

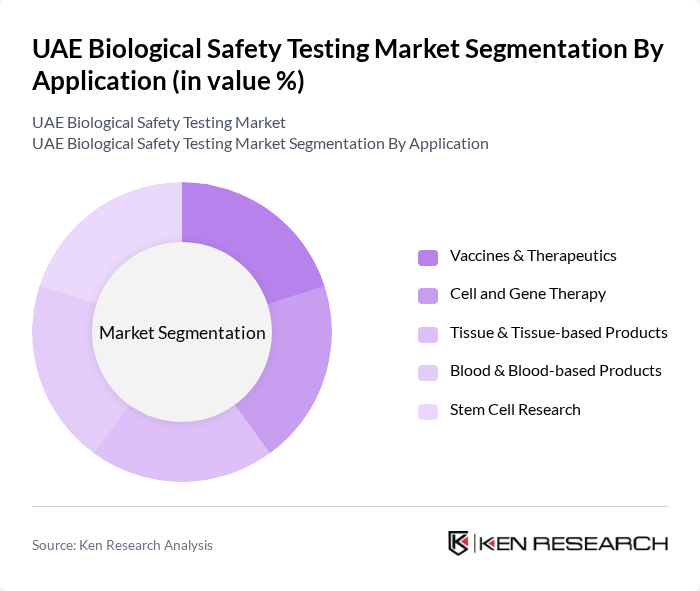
By Application:The application segmentation encompasses various fields where biological safety testing is crucial. This includes Vaccines & Therapeutics, Cell and Gene Therapy, Tissue & Tissue-based Products, Blood & Blood-based Products, and Stem Cell Research. The Vaccines & Therapeutics segment is leading the market, driven by the ongoing global health initiatives and the increasing focus on vaccine development, especially in the wake of recent health crises.
The UAE Biological Safety Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as SGS S.A., Eurofins Scientific, Charles River Laboratories, Merck KGaA, Intertek Group plc, WuXi AppTec, Labcorp Drug Development, Nelson Labs (part of Sotera Health), BioReliance (part of Merck), Toxikon Corporation, Medpace Holdings, Inc., BioAgilytix Labs, Pacific BioLabs, NAMSA (North American Science Associates), Viroclinics Xplore contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE biological safety testing market appears promising, driven by advancements in technology and increasing investments in healthcare infrastructure. As the sector evolves, automation and artificial intelligence are expected to play pivotal roles in enhancing testing efficiency and accuracy. Furthermore, the growing emphasis on personalized medicine will likely create new avenues for testing services, ensuring that the market remains dynamic and responsive to emerging healthcare needs and innovations.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Bioburden Testing Services Endotoxin Testing Services Sterility Testing Services Viral Clearance and Validation Services Cell Line Authentication and Characterization Tests Residual Host Contamination Testing Services Adventitious Agent Detection Testing Services |
| By Application | Vaccines & Therapeutics Cell and Gene Therapy Tissue & Tissue-based Products Blood & Blood-based Products Stem Cell Research |
| By End-User | Biopharmaceutical Companies Pharmaceutical Companies Biotechnology Firms Academic and Research Institutions Contract Research Organizations (CROs) |
| By Product Type | Reagents & Kits Services Instruments |
| By Region | Abu Dhabi Dubai Sharjah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Testing Facilities | 60 | Laboratory Managers, Quality Assurance Officers |
| Biotechnology Research Institutions | 50 | Research Scientists, Compliance Managers |
| Academic Laboratories | 40 | Principal Investigators, Lab Technicians |
| Regulatory Bodies | 40 | Regulatory Affairs Specialists, Policy Makers |
| Contract Research Organizations (CROs) | 45 | Project Managers, Business Development Executives |
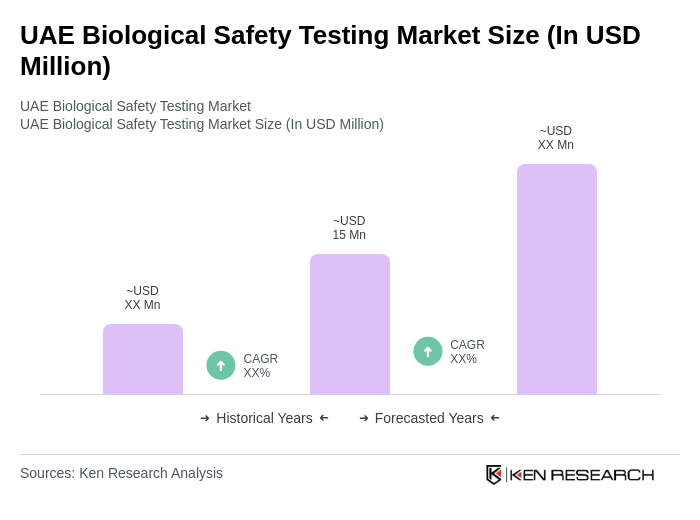
The UAE Biological Safety Testing Market is valued at approximately USD 15 million, reflecting a five-year historical analysis. This growth is driven by increasing demand for safety testing in pharmaceuticals and biotechnology, alongside stringent regulatory requirements for product safety and efficacy.