UAE Duchenne Muscular Dystrophy (DMD) Drugs Market Overview
- The UAE Duchenne Muscular Dystrophy (DMD) Drugs Market is valued at approximately USD 5 million, based on a five-year historical analysis. This growth is primarily driven by increasing awareness of DMD, advancements in drug development—particularly gene and RNA therapies—and the rising prevalence of the disease in the region. The market is also supported by government initiatives aimed at improving healthcare access and funding for rare disease research, with a notable focus on expanding newborn screening and fostering multinational collaborations for innovative therapies .
- Key cities such as Dubai and Abu Dhabi dominate the UAE DMD drugs market due to their advanced healthcare infrastructure, high concentration of medical research institutions, and availability of specialized healthcare services. These cities also attract international pharmaceutical companies, enhancing the market's growth potential through collaborations and innovative treatment options .
- In 2023, the UAE government implemented the “Guidelines for Clinical Trials in the UAE,” issued by the Ministry of Health and Prevention (MOHAP), mandating that all pharmaceutical companies must conduct local clinical trials for DMD drugs before obtaining market approval. This regulation requires companies to submit trial protocols for approval, enroll UAE patients, and report safety/efficacy data specific to the local population, thereby enhancing patient trust and improving treatment outcomes .
 Drugs Market.png)
UAE Duchenne Muscular Dystrophy (DMD) Drugs Market Segmentation
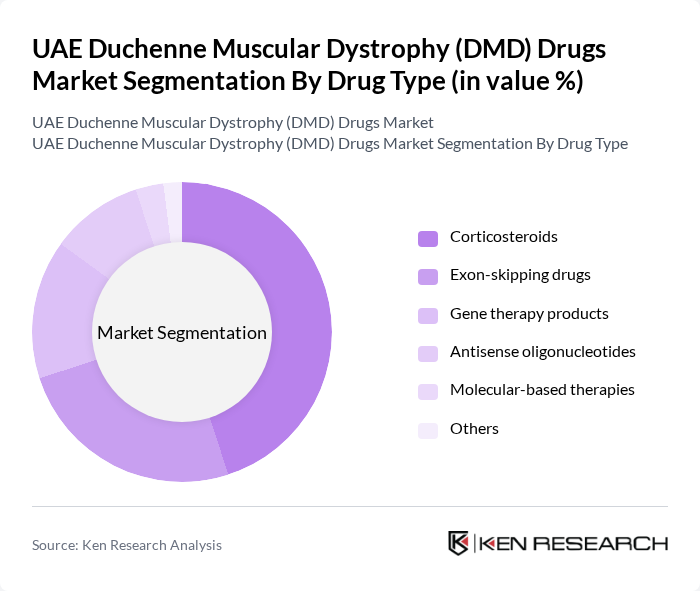
By Drug Type:The market is segmented into various drug types, including corticosteroids, exon-skipping drugs, gene therapy products, antisense oligonucleotides, molecular-based therapies, and others. Each of these drug types plays a crucial role in the treatment of DMD, with corticosteroids remaining the most widely used due to their effectiveness in slowing disease progression. Exon-skipping drugs and gene therapy products are gaining traction as innovative therapies that target the underlying genetic causes of DMD, appealing to both patients and healthcare providers. Recent trends highlight the growing adoption of molecular-based therapies and gene therapies, driven by improved clinical outcomes and regulatory approvals .
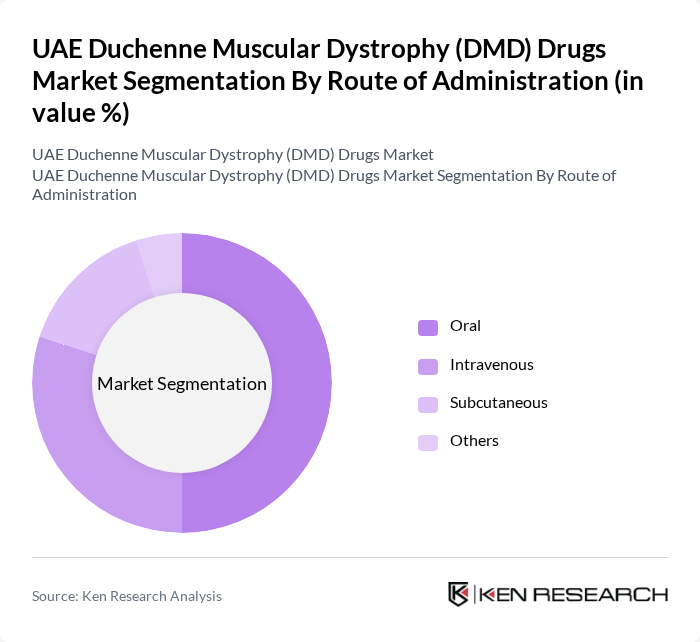
By Route of Administration:The drugs are administered through various routes, including oral, intravenous, subcutaneous, and others. Oral administration is the most preferred route due to its convenience and ease of use, particularly for pediatric patients. Intravenous and subcutaneous routes are also significant, especially for drugs that require rapid absorption or continuous delivery, making them essential in the treatment regimen for DMD. Recent market trends indicate a steady increase in intravenous and subcutaneous formulations, reflecting the introduction of advanced biologics and gene therapies .
UAE Duchenne Muscular Dystrophy (DMD) Drugs Market Competitive Landscape
The UAE Duchenne Muscular Dystrophy (DMD) Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Sarepta Therapeutics, PTC Therapeutics, Pfizer, Sanofi, F. Hoffmann-La Roche AG, Novartis AG, Vertex Pharmaceuticals Incorporated, BioMarin Pharmaceutical Inc., Astellas Pharma Inc., Eli Lilly and Company, Amgen Inc., GlaxoSmithKline plc, Teva Pharmaceutical Industries Ltd., Merck & Co., Inc., AbbVie Inc. contribute to innovation, geographic expansion, and service delivery in this space.
UAE Duchenne Muscular Dystrophy (DMD) Drugs Market Industry Analysis
Growth Drivers
- Increasing Prevalence of DMD:The UAE has reported approximately 1 in 3,500 live male births affected by Duchenne Muscular Dystrophy (DMD), translating to around 200 new cases annually. This rising prevalence is driving demand for effective treatments. The UAE's population of 9.5 million includes a significant number of young males, further emphasizing the need for targeted therapies. As awareness grows, healthcare providers are increasingly focused on addressing this urgent health issue, thereby propelling market growth.
- Advancements in Gene Therapy:The UAE is witnessing significant advancements in gene therapy, with investments exceeding AED 600 million in research and development for rare diseases. Innovative treatments, such as exon-skipping therapies, are showing promise in clinical trials, potentially improving patient outcomes. The UAE's commitment to becoming a hub for biotechnology is evident, as it aims to attract global pharmaceutical companies to develop and commercialize DMD therapies, thus enhancing treatment options for patients.
- Government Support and Funding:The UAE government allocated AED 1.2 billion in future for rare disease initiatives, including DMD. This funding supports research, treatment accessibility, and patient care programs. Additionally, the establishment of the UAE Rare Disease Alliance aims to streamline efforts in combating rare diseases, fostering collaboration among stakeholders. Such government initiatives are crucial in creating a conducive environment for the development and distribution of DMD drugs, ultimately benefiting patients and healthcare providers alike.
Market Challenges
- High Cost of DMD Treatments:The average annual cost of DMD treatments in the UAE can exceed AED 1.2 million per patient, creating a significant financial burden on families and healthcare systems. This high cost limits access to essential therapies, particularly for low-income families. The lack of comprehensive insurance coverage for rare diseases further exacerbates this challenge, hindering the overall growth of the DMD drugs market in the region.
- Limited Availability of Specialized Healthcare Facilities:The UAE has only a handful of specialized centers for DMD treatment, with fewer than 12 facilities equipped to provide comprehensive care. This scarcity leads to long wait times for diagnosis and treatment, negatively impacting patient outcomes. Furthermore, the uneven distribution of healthcare resources across the emirates complicates access for patients, particularly those in remote areas, posing a significant challenge to market expansion.
UAE Duchenne Muscular Dystrophy (DMD) Drugs Market Future Outlook
The future of the UAE DMD drugs market appears promising, driven by ongoing advancements in gene therapy and increased government funding. As the healthcare landscape evolves, the integration of digital health technologies is expected to enhance patient management and treatment adherence. Additionally, the focus on personalized medicine will likely lead to more effective therapies tailored to individual patient needs, improving overall care and outcomes for those affected by DMD in the UAE.
Market Opportunities
- Development of New Therapeutic Agents:The UAE's investment in biotechnology presents a significant opportunity for the development of innovative therapeutic agents targeting DMD. With over AED 400 million allocated for research in future, pharmaceutical companies can explore novel treatment pathways, potentially leading to breakthroughs that improve patient quality of life and treatment efficacy.
- Expansion of Telemedicine for Patient Management:The rise of telemedicine in the UAE, with a projected growth rate of 30% in future, offers a unique opportunity for enhancing DMD patient management. By leveraging digital platforms, healthcare providers can offer remote consultations, monitoring, and support, improving access to care for patients in underserved areas and facilitating timely interventions.
 Drugs Market.png)