Region:Middle East
Author(s):Geetanshi
Product Code:KRAE0626
Pages:83
Published On:December 2025
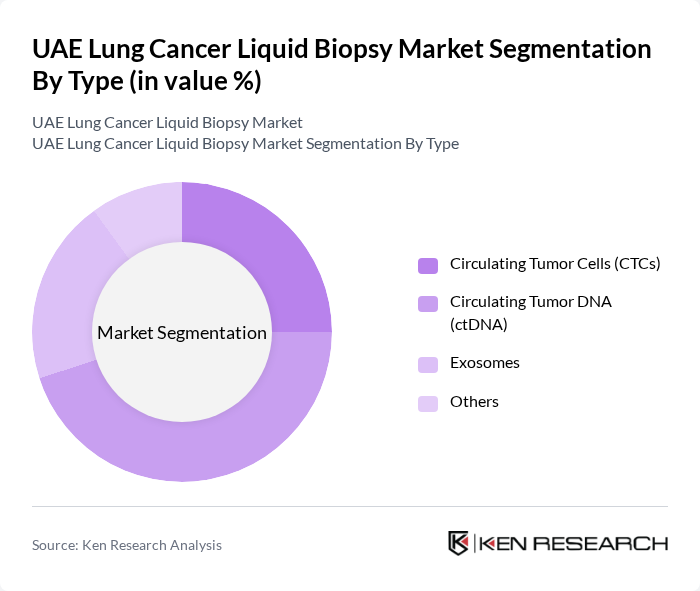
By Type:The market is segmented into Circulating Tumor Cells (CTCs), Circulating Tumor DNA (ctDNA), Exosomes, and Others. Among these, Circulating Tumor DNA (ctDNA) is the leading sub-segment due to its high sensitivity and specificity in detecting genetic mutations associated with lung cancer. The increasing preference for ctDNA assays in clinical settings is driven by their non-invasive nature and ability to provide real-time insights into tumor dynamics, making them essential for personalized treatment approaches.
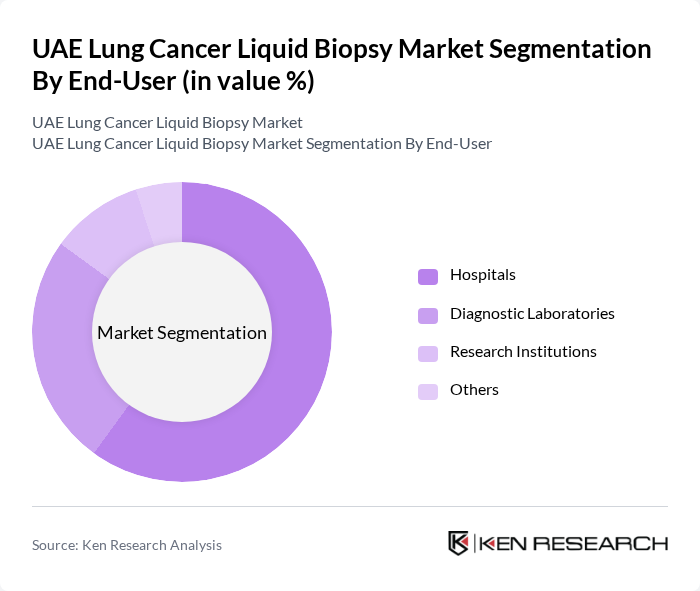
By End-User:The market is categorized into Hospitals, Diagnostic Laboratories, Research Institutions, and Others. Hospitals are the dominant end-user segment, accounting for a significant share of the market. This is primarily due to the increasing adoption of liquid biopsy tests in clinical settings for early detection and monitoring of lung cancer, as well as the growing emphasis on personalized medicine in hospital environments.
The UAE Lung Cancer Liquid Biopsy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Guardant Health, Foundation Medicine, Biocept, Roche Diagnostics, Qiagen, Illumina, Sysmex Corporation, Exact Sciences, Natera, LabCorp, Myriad Genetics, Caris Life Sciences, Freenome, C2i Genomics, OncoOne contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE lung cancer liquid biopsy market appears promising, driven by ongoing technological advancements and increasing healthcare investments. As the government prioritizes cancer care, initiatives to enhance healthcare infrastructure and promote research are expected to flourish. Furthermore, the integration of artificial intelligence in diagnostics is anticipated to streamline processes, improving accuracy and efficiency. These trends will likely foster a more robust market environment, encouraging the adoption of liquid biopsy technologies in clinical practice.
| Segment | Sub-Segments |
|---|---|
| By Type | Circulating Tumor Cells (CTCs) Circulating Tumor DNA (ctDNA) Exosomes Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Others |
| By Sample Type | Blood Samples Urine Samples Tissue Samples Others |
| By Application | Early Detection Treatment Monitoring Recurrence Monitoring Others |
| By Technology | Next-Generation Sequencing (NGS) Polymerase Chain Reaction (PCR) Digital PCR Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncologists and Hematologists | 100 | Medical Doctors, Cancer Specialists |
| Laboratory Technicians and Managers | 80 | Lab Managers, Diagnostic Technologists |
| Healthcare Administrators | 60 | Hospital Administrators, Policy Makers |
| Patients with Lung Cancer | 75 | Patients, Caregivers, Support Group Members |
| Health Insurance Providers | 50 | Insurance Analysts, Claims Managers |
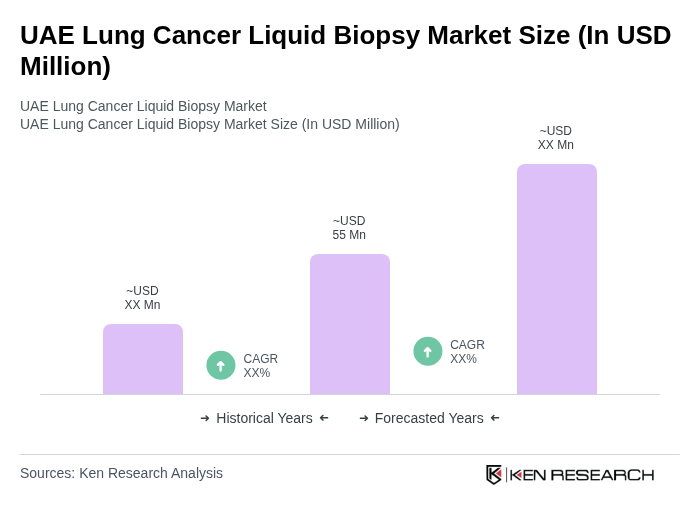
The UAE Lung Cancer Liquid Biopsy Market is valued at approximately USD 55 million, reflecting a significant growth trend driven by the increasing incidence of lung cancer and the demand for non-invasive diagnostic methods.