About the Report
Base Year 2024UAE Small Molecule CDMO Market Overview
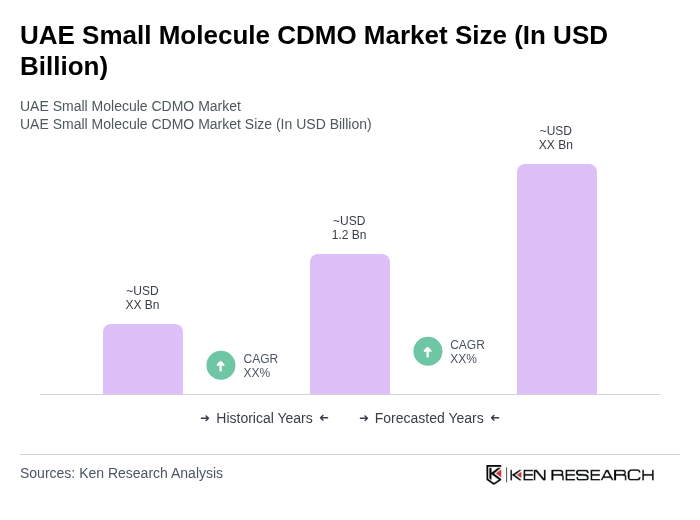
- The UAE Small Molecule CDMO Market is valued at USD 1.2 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing demand for pharmaceuticals, particularly in the oncology and chronic disease sectors, alongside a rise in outsourcing by pharmaceutical companies seeking cost-effective manufacturing solutions. Additional drivers include robust government support for local manufacturing initiatives, strategic global alliances, and the UAE’s competitive export advantage across the MENA region, all of which are accelerating market expansion and international CDMO collaborations .
- Key cities dominating this market include Dubai and Abu Dhabi, which serve as major hubs for pharmaceutical manufacturing and distribution. Their strategic location, advanced infrastructure, and supportive government policies—such as incentives for local production and the establishment of free zones—contribute to their prominence in the CDMO landscape .
- In 2023, the UAE government implemented Cabinet Decision No. 32 of 2023 Concerning the Regulation of Pharmaceutical Establishments and Products, issued by the Ministry of Health and Prevention. This binding regulation mandates that all pharmaceutical manufacturing facilities in the UAE comply with the latest Good Manufacturing Practices (GMP) standards, covering facility licensing, quality management systems, and regular inspections to ensure product quality and safety in line with international benchmarks .
UAE Small Molecule CDMO Market Segmentation
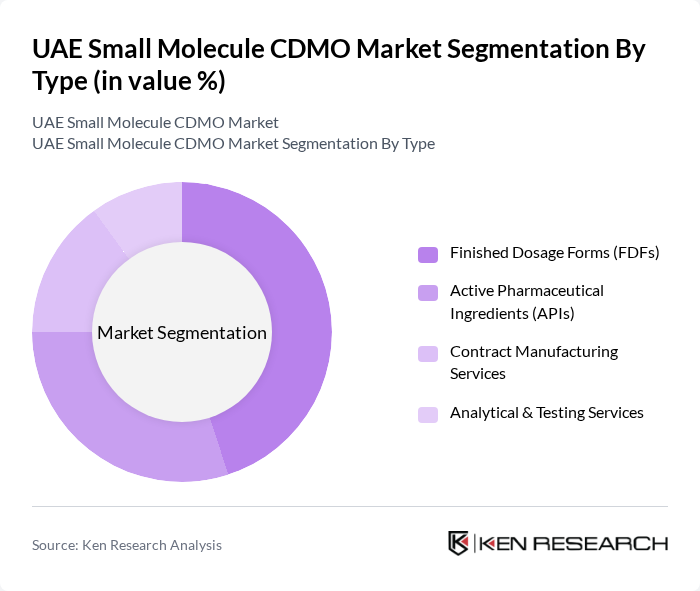
By Type:The market is segmented into four main types: Active Pharmaceutical Ingredients (APIs), Finished Dosage Forms (FDFs), Contract Manufacturing Services, and Analytical & Testing Services. Among these, Finished Dosage Forms (FDFs) currently dominate the UAE market, reflecting the region’s focus on advanced drug delivery systems and value-added formulations. The demand for FDFs is driven by the need for patient-centric products and the UAE’s emphasis on quality and innovation in healthcare. While APIs remain critical, the local market has seen a shift towards end-to-end solutions for drug product manufacturing, with CDMOs investing in formulation development and commercial-scale production .
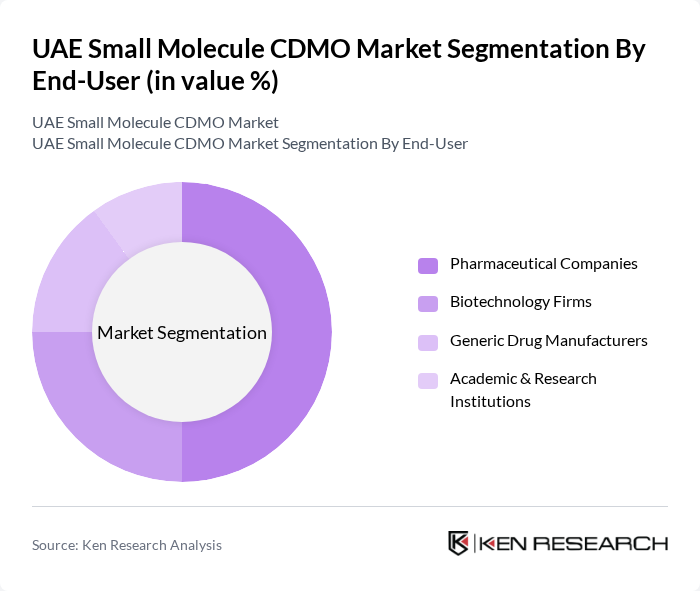
By End-User:The end-user segmentation includes Pharmaceutical Companies, Biotechnology Firms, Generic Drug Manufacturers, and Academic & Research Institutions. Pharmaceutical Companies are the leading end-users, driven by the need for reliable manufacturing partners to support their extensive product pipelines. The increasing complexity of drug formulations, the need for compliance with stringent regulatory standards, and the UAE’s push for local production have made CDMOs essential for these companies .
UAE Small Molecule CDMO Market Competitive Landscape
The UAE Small Molecule CDMO Market is characterized by a dynamic mix of regional and international players. Leading participants such as Globalpharma Co. LLC (Dubai Investments), Neopharma LLC, Pharmax Pharmaceuticals FZ LLC, Julphar Gulf Pharmaceutical Industries, LifePharma FZE, Tabuk Pharmaceuticals Manufacturing Co., Lonza Group, Catalent, Inc., Recipharm AB, Aenova Group, Siegfried Holding AG, Piramal Pharma Solutions, Famar, Alcami Corporation, Jubilant HollisterStier contribute to innovation, geographic expansion, and service delivery in this space.
UAE Small Molecule CDMO Market Industry Analysis
Growth Drivers
- Increasing Demand for Generic Drugs:The UAE's generic drug market is projected to reach AED 4.9 billion in future, driven by rising healthcare costs and a growing aging population. The government’s initiatives to promote affordable healthcare have led to a 15% increase in generic drug prescriptions over the past two years. This trend is expected to continue, as patients and healthcare providers increasingly favor cost-effective alternatives to branded medications, thereby boosting the small molecule CDMO sector.
- Rise in Biopharmaceuticals:The biopharmaceutical sector in the UAE is anticipated to grow significantly, with investments reaching AED 1.2 billion in future. This growth is fueled by advancements in biotechnology and an increasing focus on personalized medicine. The UAE's strategic location and investment in research facilities have attracted global biopharmaceutical companies, leading to a 20% increase in demand for small molecule CDMO services to support drug development and manufacturing processes.
- Government Support for Healthcare Initiatives:The UAE government has allocated AED 4.9 billion for healthcare initiatives in future, emphasizing the importance of local pharmaceutical manufacturing. This funding is aimed at enhancing healthcare infrastructure and promoting local production of medicines. As a result, small molecule CDMOs are expected to benefit from increased contracts and collaborations with government entities, leading to a more robust market environment and improved service offerings.
Market Challenges
- Regulatory Compliance Complexities:The UAE's pharmaceutical industry faces stringent regulatory requirements, which can complicate the operations of small molecule CDMOs. Compliance with the Ministry of Health and Prevention's regulations requires significant investment in quality assurance and control systems. In future, the cost of compliance is projected to rise by 10%, placing additional financial strain on CDMOs and potentially hindering their ability to compete effectively in the market.
- High Competition Among CDMOs:The UAE small molecule CDMO market is characterized by intense competition, with over 50 active players vying for market share. This competitive landscape has led to price wars, reducing profit margins for many companies. In future, the average profit margin for CDMOs is expected to decline by 5%, as companies invest heavily in marketing and technology to differentiate their services, making it challenging for smaller firms to sustain operations.
UAE Small Molecule CDMO Market Future Outlook
The future of the UAE small molecule CDMO market appears promising, driven by increasing investments in healthcare and a growing emphasis on local manufacturing. As the demand for personalized medicine rises, CDMOs will need to adapt to new technologies and processes. Additionally, strategic partnerships with biopharmaceutical companies will likely enhance service offerings and expand market reach. The focus on sustainability and digital transformation will further shape the industry landscape, fostering innovation and efficiency in drug development and production.
Market Opportunities
- Growth in Contract Manufacturing:The contract manufacturing segment is expected to expand, with an estimated value of AED 1 billion in future. This growth is driven by pharmaceutical companies seeking to outsource production to reduce costs and increase efficiency. CDMOs that can offer flexible and scalable manufacturing solutions will be well-positioned to capture this opportunity and enhance their market presence.
- Technological Advancements in Drug Development:Innovations in drug development technologies, such as AI and machine learning, are set to revolutionize the small molecule CDMO landscape. In future, investments in these technologies are projected to exceed AED 500 million, enabling CDMOs to streamline processes and improve product quality. Companies that leverage these advancements will gain a competitive edge and attract more clients seeking efficient and effective drug development solutions.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Active Pharmaceutical Ingredients (APIs) Finished Dosage Forms (FDFs) Contract Manufacturing Services Analytical & Testing Services |
| By End-User | Pharmaceutical Companies Biotechnology Firms Generic Drug Manufacturers Academic & Research Institutions |
| By Therapeutic Area | Oncology Cardiovascular Infectious Diseases Metabolic Disorders |
| By Service Type | Process Development & Optimization Analytical & Quality Control Services Regulatory Support & Compliance Packaging & Logistics |
| By Scale of Operation | Commercial Scale Clinical Scale Pilot Scale Small Batch/Custom Manufacturing |
| By Region | Abu Dhabi Dubai Sharjah Northern Emirates |
| By Client Type | Startups Established Local Firms Multinational Corporations Government & Public Sector |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health and Prevention, UAE Food and Drug Authority)
Pharmaceutical Manufacturers
Biotechnology Companies
Contract Research Organizations (CROs)
Supply Chain and Logistics Providers
Pharmaceutical Distributors
Healthcare Providers and Hospitals
Players Mentioned in the Report:
Globalpharma Co. LLC (Dubai Investments)
Neopharma LLC
Pharmax Pharmaceuticals FZ LLC
Julphar Gulf Pharmaceutical Industries
LifePharma FZE
Tabuk Pharmaceuticals Manufacturing Co.
Lonza Group
Catalent, Inc.
Recipharm AB
Aenova Group
Siegfried Holding AG
Piramal Pharma Solutions
Famar
Alcami Corporation
Jubilant HollisterStier
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. UAE Small Molecule CDMO Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 UAE Small Molecule CDMO Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. UAE Small Molecule CDMO Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for generic drugs
3.1.2 Rise in biopharmaceuticals
3.1.3 Expansion of pharmaceutical companies in the UAE
3.1.4 Government support for healthcare initiatives
3.2 Market Challenges
3.2.1 Regulatory compliance complexities
3.2.2 High competition among CDMOs
3.2.3 Fluctuating raw material prices
3.2.4 Limited skilled workforce
3.3 Market Opportunities
3.3.1 Growth in contract manufacturing
3.3.2 Technological advancements in drug development
3.3.3 Strategic partnerships and collaborations
3.3.4 Expansion into emerging markets
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Increased outsourcing of R&D activities
3.4.3 Adoption of digital technologies in manufacturing
3.4.4 Focus on sustainability in production processes
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced approval processes for new drugs
3.5.3 Incentives for local manufacturing
3.5.4 Compliance with international standards
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. UAE Small Molecule CDMO Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. UAE Small Molecule CDMO Market Segmentation
8.1 By Type
8.1.1 Active Pharmaceutical Ingredients (APIs)
8.1.2 Finished Dosage Forms (FDFs)
8.1.3 Contract Manufacturing Services
8.1.4 Analytical & Testing Services
8.2 By End-User
8.2.1 Pharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Generic Drug Manufacturers
8.2.4 Academic & Research Institutions
8.3 By Therapeutic Area
8.3.1 Oncology
8.3.2 Cardiovascular
8.3.3 Infectious Diseases
8.3.4 Metabolic Disorders
8.4 By Service Type
8.4.1 Process Development & Optimization
8.4.2 Analytical & Quality Control Services
8.4.3 Regulatory Support & Compliance
8.4.4 Packaging & Logistics
8.5 By Scale of Operation
8.5.1 Commercial Scale
8.5.2 Clinical Scale
8.5.3 Pilot Scale
8.5.4 Small Batch/Custom Manufacturing
8.6 By Region
8.6.1 Abu Dhabi
8.6.2 Dubai
8.6.3 Sharjah
8.6.4 Northern Emirates
8.7 By Client Type
8.7.1 Startups
8.7.2 Established Local Firms
8.7.3 Multinational Corporations
8.7.4 Government & Public Sector
9. UAE Small Molecule CDMO Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate (3-year CAGR)
9.2.4 Market Penetration (Number of UAE-based clients/contracts)
9.2.5 Capacity Utilization Rate (%)
9.2.6 Regulatory Compliance Track Record (e.g., GMP, UAE MoHAP approvals)
9.2.7 On-time Delivery Rate (%)
9.2.8 Quality Assurance Metrics (e.g., batch failure rate, audit outcomes)
9.2.9 R&D Investment as % of Revenue
9.2.10 Supply Chain Resilience (e.g., local sourcing ratio, lead time)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Globalpharma Co. LLC (Dubai Investments)
9.5.2 Neopharma LLC
9.5.3 Pharmax Pharmaceuticals FZ LLC
9.5.4 Julphar Gulf Pharmaceutical Industries
9.5.5 LifePharma FZE
9.5.6 Tabuk Pharmaceuticals Manufacturing Co.
9.5.7 Lonza Group
9.5.8 Catalent, Inc.
9.5.9 Recipharm AB
9.5.10 Aenova Group
9.5.11 Siegfried Holding AG
9.5.12 Piramal Pharma Solutions
9.5.13 Famar
9.5.14 Alcami Corporation
9.5.15 Jubilant HollisterStier
10. UAE Small Molecule CDMO Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Ministry of Health and Prevention
10.1.2 Ministry of Industry and Advanced Technology
10.1.3 Ministry of Economy
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Pharmaceutical Infrastructure Investments
10.2.2 Energy Efficiency Initiatives
10.2.3 R&D Funding
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Cost Management
10.3.2 Quality Assurance
10.3.3 Regulatory Compliance
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Awareness of CDMO Services
10.4.2 Training and Support Needs
10.4.3 Financial Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Scalability of Solutions
10.5.3 Long-term Partnerships
10.5.4 Others
11. UAE Small Molecule CDMO Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from the UAE Ministry of Health and Prevention
- Market analysis from local pharmaceutical associations and trade publications
- Regulatory frameworks and guidelines from the UAE Ministry of Health and Prevention (MOHAP)
Primary Research
- Interviews with executives from small molecule CDMO firms operating in the UAE
- Surveys targeting R&D managers in pharmaceutical companies
- Focus groups with industry experts and consultants specializing in contract manufacturing
Validation & Triangulation
- Cross-validation of data through multiple industry sources and reports
- Triangulation of insights from primary interviews with secondary data findings
- Sanity checks conducted through expert panel discussions and feedback sessions
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of the overall pharmaceutical market size in the UAE and its growth trajectory
- Segmentation of the market by therapeutic areas and types of small molecules
- Incorporation of government initiatives promoting local manufacturing and innovation
Bottom-up Modeling
- Data collection on production capacities and output from key CDMO players
- Cost analysis based on service offerings and pricing structures in the market
- Estimation of market share based on client contracts and project pipelines
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project future market trends
- Scenario modeling based on potential regulatory changes and market dynamics
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Small Molecule CDMO Services | 60 | Business Development Managers, Operations Directors |
| Pharmaceutical Manufacturing Insights | 80 | Quality Assurance Managers, Regulatory Affairs Specialists |
| Research & Development Trends | 50 | R&D Directors, Formulation Scientists |
| Market Entry Strategies | 40 | Market Analysts, Strategic Planning Executives |
| Client Satisfaction and Feedback | 70 | Procurement Managers, Product Managers |
Frequently Asked Questions
What is the current value of the UAE Small Molecule CDMO Market?
The UAE Small Molecule CDMO Market is valued at approximately USD 1.2 billion, driven by increasing pharmaceutical demand, particularly in oncology and chronic diseases, along with a rise in outsourcing by pharmaceutical companies for cost-effective manufacturing solutions.