About the Report
Base Year 2024US Allergy Autoimmune Disease Diagnostics Market Overview
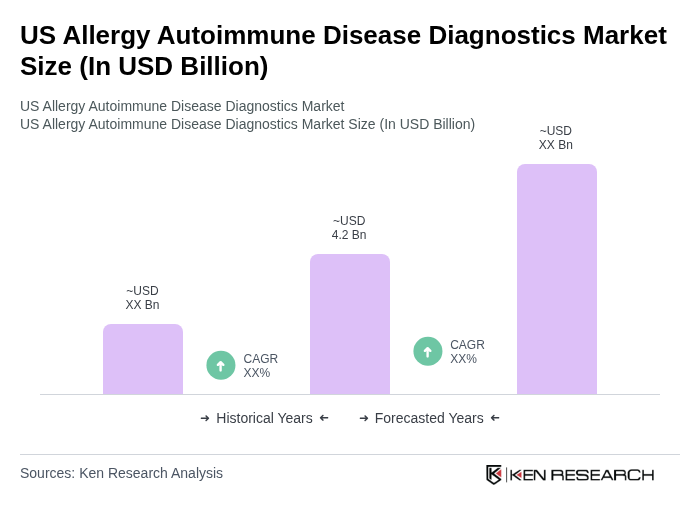
- The US Allergy Autoimmune Disease Diagnostics Market is valued at USD 4.2 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of allergies and autoimmune diseases, heightened awareness of diagnostic testing, and advancements in technology that enhance testing accuracy and efficiency. The market is also benefiting from the expansion of high-throughput diagnostic platforms, the adoption of AI-powered interpretation tools, and the integration of multiplex immunoassays for comprehensive disease profiling .
- The market is dominated by key regions such as California, New York, and Texas, which have a high population density and a significant number of healthcare facilities. These states also benefit from robust healthcare infrastructure and a growing focus on research and development in the field of diagnostics. Regional dominance is further supported by favorable reimbursement policies and concentrated investments in clinical research .
- The Clinical Laboratory Improvement Amendments (CLIA), 1988, issued by the Centers for Medicare & Medicaid Services (CMS), regulate laboratory testing quality standards in the US, including allergy and autoimmune disease diagnostics. CLIA requires laboratories to meet specific quality control, personnel qualification, and proficiency testing standards to ensure the accuracy, reliability, and timeliness of patient test results .
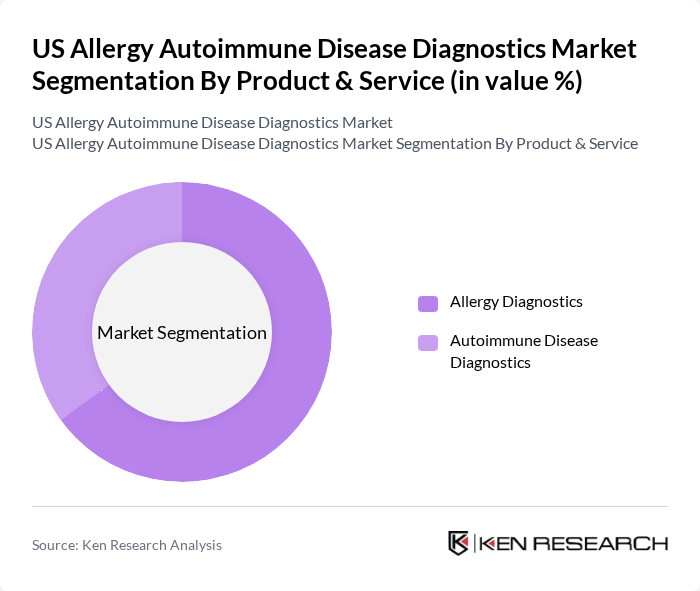
US Allergy Autoimmune Disease Diagnostics Market Segmentation
By Product & Service:The market is segmented into two primary categories: Allergy Diagnostics and Autoimmune Disease Diagnostics. Allergy Diagnostics includes tests and services specifically designed to identify allergic reactions, while Autoimmune Disease Diagnostics focuses on identifying autoimmune disorders through various testing methods. The Allergy Diagnostics segment is currently leading the market due to the rising incidence of allergies, the widespread use of skin prick and IgE blood tests, and the growing demand for rapid, accurate diagnostic solutions .
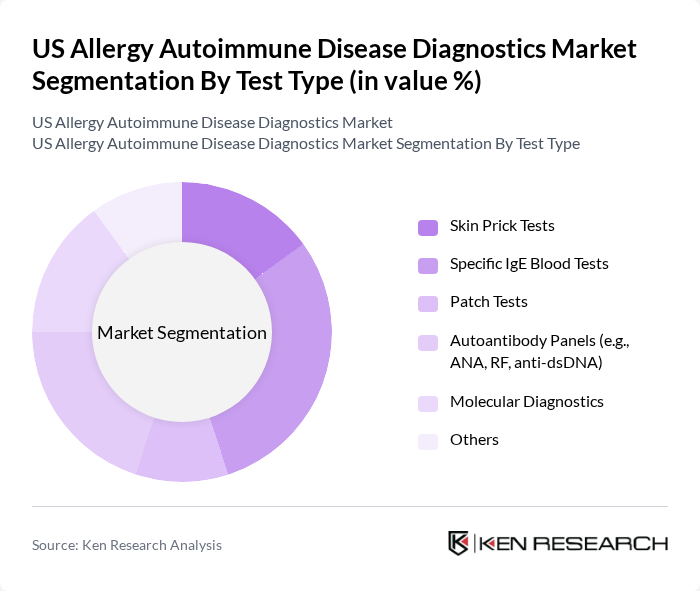
By Test Type:The market is further segmented by test types, including Skin Prick Tests, Specific IgE Blood Tests, Patch Tests, Autoantibody Panels (e.g., ANA, RF, anti-dsDNA), Molecular Diagnostics, and Others. Among these, Specific IgE Blood Tests are dominating the market due to their high accuracy, reliability, and preference among healthcare providers for identifying allergen sensitivities. Autoantibody panels, particularly antinuclear antibody (ANA) tests, are also widely adopted for autoimmune disease diagnostics, reflecting the need for early and precise detection .
US Allergy Autoimmune Disease Diagnostics Market Competitive Landscape
The US Allergy Autoimmune Disease Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific, Abbott Laboratories, Siemens Healthineers, Roche Diagnostics, Quest Diagnostics, Laboratory Corporation of America Holdings (Labcorp), Bio-Rad Laboratories, Danaher Corporation (Beckman Coulter), PerkinElmer (now Revvity, Inc.), Grifols, S.A., Hologic, Inc., Becton, Dickinson and Company (BD), Agilent Technologies, Exagen Inc., EUROIMMUN (a PerkinElmer company) contribute to innovation, geographic expansion, and service delivery in this space.
US Allergy Autoimmune Disease Diagnostics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Allergy and Autoimmune Diseases:The prevalence of allergies and autoimmune diseases in the U.S. has reached alarming levels, with approximately60 million Americanssuffering from allergies annually. According to the CDC, autoimmune diseases affect around30 million people, highlighting a significant demand for diagnostic solutions. This growing patient population drives the need for advanced diagnostic tests, as healthcare providers seek to identify and manage these conditions effectively, thereby propelling market growth.
- Advancements in Diagnostic Technologies:The U.S. diagnostics market is witnessing rapid technological advancements, with investments in innovative tools such as multiplex assays and next-generation sequencing. The FDA reported that over250new diagnostic tests were approved in future alone, enhancing the accuracy and speed of allergy and autoimmune disease detection. These advancements not only improve patient outcomes but also stimulate market growth by attracting healthcare providers to adopt cutting-edge diagnostic solutions.
- Rising Awareness and Education about Allergies:Increased public awareness regarding allergies and autoimmune diseases has led to a surge in demand for diagnostic testing. Educational campaigns by organizations like the American Academy of Allergy, Asthma & Immunology have reached millions, emphasizing the importance of early diagnosis. In future, over70%of adults reported understanding the symptoms of allergies, which correlates with a20%increase in diagnostic test requests, further driving market expansion.
Market Challenges
- High Costs of Diagnostic Tests:The financial burden of allergy and autoimmune disease diagnostics remains a significant challenge, with average test costs ranging from$250 to $1,800. Many patients face out-of-pocket expenses due to inadequate insurance coverage, leading to delayed diagnoses. According to the National Institute of Health, nearly35%of patients forgo necessary tests due to cost, which hampers market growth and limits access to essential diagnostic services.
- Limited Access to Healthcare Facilities:Access to healthcare facilities, particularly in rural areas, poses a challenge for allergy and autoimmune disease diagnostics. The U.S. Health Resources and Services Administration reported that25%of rural residents live more than40 milesfrom the nearest diagnostic center. This geographical disparity results in lower testing rates and delayed diagnoses, ultimately affecting patient outcomes and hindering market growth in underserved regions.
US Allergy Autoimmune Disease Diagnostics Market Future Outlook
The future of the U.S. allergy autoimmune disease diagnostics market appears promising, driven by technological innovations and increasing healthcare investments. The integration of artificial intelligence in diagnostics is expected to enhance accuracy and efficiency, while the rise of telemedicine will facilitate remote testing solutions. As healthcare systems prioritize preventive measures, the demand for early diagnostic tools will likely grow, fostering a more proactive approach to managing allergies and autoimmune diseases in future.
Market Opportunities
- Growth in Telemedicine and Remote Diagnostics:The telemedicine sector is projected to reach$200 billionby future, creating opportunities for remote diagnostic solutions. This growth allows patients to access allergy testing from home, increasing convenience and compliance. As healthcare providers adopt telehealth platforms, the demand for remote diagnostics will likely rise, enhancing market accessibility and patient engagement.
- Expansion of Personalized Medicine:The personalized medicine market is expected to grow to$3 trillionby future, offering significant opportunities for tailored diagnostic solutions. By focusing on individual patient profiles, diagnostic companies can develop specific tests that cater to unique genetic and environmental factors. This trend will likely enhance treatment efficacy and patient satisfaction, driving further investment in the diagnostics market.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product & Service | Allergy Diagnostics Autoimmune Disease Diagnostics |
| By Test Type | Skin Prick Tests Specific IgE Blood Tests Patch Tests Autoantibody Panels (e.g., ANA, RF, anti-dsDNA) Molecular Diagnostics Others |
| By Diagnostics Type | Allergy Diagnostics Autoimmune Disease Diagnostics |
| By End-User | Hospitals Clinics Diagnostic Laboratories Research Institutions Homecare Settings Others |
| By Disease Type | Allergic Rhinitis Asthma Eczema Food Allergies Celiac Disease Rheumatoid Arthritis Systemic Lupus Erythematosus (SLE) Type 1 Diabetes Others |
| By Distribution Channel | Direct Sales Online Sales Retail Pharmacies Hospitals and Clinics Others |
| By Geography | Northeast Midwest South West Others |
| By Technology | Enzyme-Linked Immunosorbent Assay (ELISA) Polymerase Chain Reaction (PCR) Next-Generation Sequencing (NGS) Multiplex Immunoassays Indirect Immunofluorescence Digital & AI-powered Diagnostics Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, Centers for Disease Control and Prevention)
Manufacturers and Producers of Diagnostic Devices
Distributors and Retailers of Medical Equipment
Healthcare Providers and Clinics Specializing in Allergy and Autoimmune Diseases
Biotechnology and Pharmaceutical Companies
Health Insurance Companies and Payers
Industry Associations and Advocacy Groups
Players Mentioned in the Report:
Thermo Fisher Scientific
Abbott Laboratories
Siemens Healthineers
Roche Diagnostics
Quest Diagnostics
Laboratory Corporation of America Holdings (Labcorp)
Bio-Rad Laboratories
Danaher Corporation (Beckman Coulter)
PerkinElmer (now Revvity, Inc.)
Grifols, S.A.
Hologic, Inc.
Becton, Dickinson and Company (BD)
Agilent Technologies
Exagen Inc.
EUROIMMUN (a PerkinElmer company)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Allergy Autoimmune Disease Diagnostics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Allergy Autoimmune Disease Diagnostics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Allergy Autoimmune Disease Diagnostics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of allergy and autoimmune diseases
3.1.2 Advancements in diagnostic technologies
3.1.3 Rising awareness and education about allergies
3.1.4 Government initiatives and funding for research
3.2 Market Challenges
3.2.1 High costs of diagnostic tests
3.2.2 Limited access to healthcare facilities
3.2.3 Regulatory hurdles in product approvals
3.2.4 Competition from alternative diagnostic methods
3.3 Market Opportunities
3.3.1 Growth in telemedicine and remote diagnostics
3.3.2 Expansion of personalized medicine
3.3.3 Development of novel biomarkers
3.3.4 Strategic partnerships and collaborations
3.4 Market Trends
3.4.1 Increasing use of AI and machine learning in diagnostics
3.4.2 Shift towards home-based testing solutions
3.4.3 Integration of digital health technologies
3.4.4 Focus on preventive healthcare measures
3.5 Government Regulation
3.5.1 FDA guidelines for diagnostic tests
3.5.2 CLIA regulations for laboratory testing
3.5.3 Reimbursement policies for allergy diagnostics
3.5.4 State-level regulations impacting diagnostics
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Allergy Autoimmune Disease Diagnostics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Allergy Autoimmune Disease Diagnostics Market Segmentation
8.1 By Product & Service
8.1.1 Allergy Diagnostics
8.1.2 Autoimmune Disease Diagnostics
8.2 By Test Type
8.2.1 Skin Prick Tests
8.2.2 Specific IgE Blood Tests
8.2.3 Patch Tests
8.2.4 Autoantibody Panels (e.g., ANA, RF, anti-dsDNA)
8.2.5 Molecular Diagnostics
8.2.6 Others
8.3 By Diagnostics Type
8.3.1 Allergy Diagnostics
8.3.2 Autoimmune Disease Diagnostics
8.4 By End-User
8.4.1 Hospitals
8.4.2 Clinics
8.4.3 Diagnostic Laboratories
8.4.4 Research Institutions
8.4.5 Homecare Settings
8.4.6 Others
8.5 By Disease Type
8.5.1 Allergic Rhinitis
8.5.2 Asthma
8.5.3 Eczema
8.5.4 Food Allergies
8.5.5 Celiac Disease
8.5.6 Rheumatoid Arthritis
8.5.7 Systemic Lupus Erythematosus (SLE)
8.5.8 Type 1 Diabetes
8.5.9 Others
8.6 By Distribution Channel
8.6.1 Direct Sales
8.6.2 Online Sales
8.6.3 Retail Pharmacies
8.6.4 Hospitals and Clinics
8.6.5 Others
8.7 By Geography
8.7.1 Northeast
8.7.2 Midwest
8.7.3 South
8.7.4 West
8.7.5 Others
8.8 By Technology
8.8.1 Enzyme-Linked Immunosorbent Assay (ELISA)
8.8.2 Polymerase Chain Reaction (PCR)
8.8.3 Next-Generation Sequencing (NGS)
8.8.4 Multiplex Immunoassays
8.8.5 Indirect Immunofluorescence
8.8.6 Digital & AI-powered Diagnostics
8.8.7 Others
9. US Allergy Autoimmune Disease Diagnostics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 KPIs for Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 US Market Revenue (USD, latest year)
9.2.4 Revenue Growth Rate (CAGR %)
9.2.5 Market Share (%)
9.2.6 Number of Diagnostic Tests Performed (Annual Volume)
9.2.7 R&D Expenditure (% of Revenue)
9.2.8 Product Portfolio Breadth (Number of Diagnostic Panels/Tests)
9.2.9 Regulatory Approvals (Number of FDA/CLIA Cleared Products)
9.2.10 Geographic Coverage (Number of US States/Regions Served)
9.2.11 Distribution Network Strength (Number of Partner Labs/Facilities)
9.2.12 Customer Retention Rate (%)
9.2.13 Customer Satisfaction Index (Survey Score)
9.2.14 Pricing Strategy (Premium/Value/Volume)
9.2.15 Product Innovation Rate (New Launches per Year)
9.2.16 Digital Integration Level (AI/Telehealth/Remote Diagnostics)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Thermo Fisher Scientific
9.5.2 Abbott Laboratories
9.5.3 Siemens Healthineers
9.5.4 Roche Diagnostics
9.5.5 Quest Diagnostics
9.5.6 Laboratory Corporation of America Holdings (Labcorp)
9.5.7 Bio-Rad Laboratories
9.5.8 Danaher Corporation (Beckman Coulter)
9.5.9 PerkinElmer (now Revvity, Inc.)
9.5.10 Grifols, S.A.
9.5.11 Hologic, Inc.
9.5.12 Becton, Dickinson and Company (BD)
9.5.13 Agilent Technologies
9.5.14 Exagen Inc.
9.5.15 EUROIMMUN (a PerkinElmer company)
10. US Allergy Autoimmune Disease Diagnostics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Allergy Diagnostics
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers and Vendors
10.1.4 Compliance and Regulatory Considerations
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Diagnostic Equipment
10.2.2 Funding for Research and Development
10.2.3 Budget for Training and Development
10.2.4 Expenditure on Marketing and Outreach
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Diagnostic Laboratories
10.3.3 Concerns of Research Institutions
10.3.4 Homecare User Challenges
10.4 User Readiness for Adoption
10.4.1 Awareness of Diagnostic Options
10.4.2 Training Needs for Healthcare Providers
10.4.3 Acceptance of New Technologies
10.4.4 Financial Readiness for Adoption
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Diagnostic Accuracy
10.5.2 Cost Savings from Early Detection
10.5.3 Expansion into New Diagnostic Areas
10.5.4 User Feedback and Improvement Cycles
11. US Allergy Autoimmune Disease Diagnostics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of published market reports from healthcare organizations and industry associations
- Review of academic journals and articles focusing on allergy and autoimmune disease diagnostics
- Examination of government health statistics and regulatory frameworks related to diagnostics
Primary Research
- Interviews with leading allergists and immunologists to gather insights on diagnostic trends
- Surveys with laboratory managers and technicians involved in allergy testing
- Focus groups with patients to understand their experiences and preferences regarding diagnostics
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of market data with clinical guidelines and treatment protocols
- Sanity checks through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure on allergy and autoimmune diagnostics
- Segmentation by diagnostic type, including in vitro and in vivo tests
- Incorporation of trends in telemedicine and home testing kits for allergy diagnostics
Bottom-up Modeling
- Collection of sales data from major diagnostic manufacturers and laboratories
- Estimation of market share based on product offerings and regional presence
- Volume and pricing analysis for various diagnostic tests and services
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and emerging market trends
- Scenario analysis based on potential changes in healthcare policies and patient demographics
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Allergy Testing Laboratories | 100 | Laboratory Directors, Quality Control Managers |
| Immunology Clinics | 60 | Allergists, Nurse Practitioners |
| Healthcare Providers | 110 | General Practitioners, Family Medicine Physicians |
| Patient Advocacy Groups | 50 | Patient Coordinators, Community Outreach Managers |
| Health Insurance Companies | 40 | Policy Analysts, Claims Managers |
Frequently Asked Questions
What is the current value of the US Allergy Autoimmune Disease Diagnostics Market?
The US Allergy Autoimmune Disease Diagnostics Market is valued at approximately USD 4.2 billion, reflecting a significant growth driven by the rising prevalence of allergies and autoimmune diseases, increased awareness of diagnostic testing, and advancements in technology.