About the Report
Base Year 2024US Electronic Trial Master File eTMF Systems Market Overview
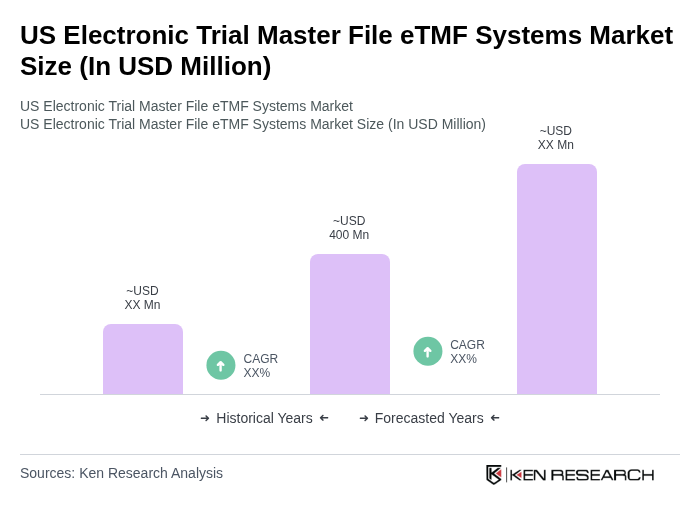
- The US Electronic Trial Master File eTMF Systems Market is valued at USD 400 million, based on a five-year historical analysis and North America’s revenue share within the global eTMF systems space. This growth is primarily driven by the increasing adoption of digital solutions in clinical trials, the need for regulatory compliance, and the demand for efficient data management systems. The shift towards remote monitoring, decentralized and hybrid trials, and the rising volume and complexity of clinical data have further accelerated the need for robust eTMF systems.
- Key demand clusters in this market are concentrated in major biopharmaceutical and research hubs such as the Greater Boston area, the San Francisco Bay Area, and the New York–New Jersey corridor, driven by their high density of pharmaceutical and biotechnology companies, academic medical centers, and contract research organizations. These regions are hubs for innovation and investment in clinical research, fostering a competitive environment that drives the adoption of eTMF systems, particularly cloud-based and integrated clinical trial platforms.
- Regulatory expectations for electronic systems in clinical trials are guided by instruments such as the Electronic Records; Electronic Signatures regulation (21 CFR Part 11) issued by the US Food and Drug Administration, which sets requirements for the use of electronic records and electronic signatures in FDA-regulated studies, and the Electronic Source Data in Clinical Investigations guidance issued by the US Food and Drug Administration in 2013, which addresses the capture, review, and retention of electronic data in clinical investigations. These frameworks, together with ICH E6 Good Clinical Practice requirements for essential documents, strongly encourage the use of validated electronic systems such as eTMF to enhance data integrity, audit readiness, and streamlined submissions.
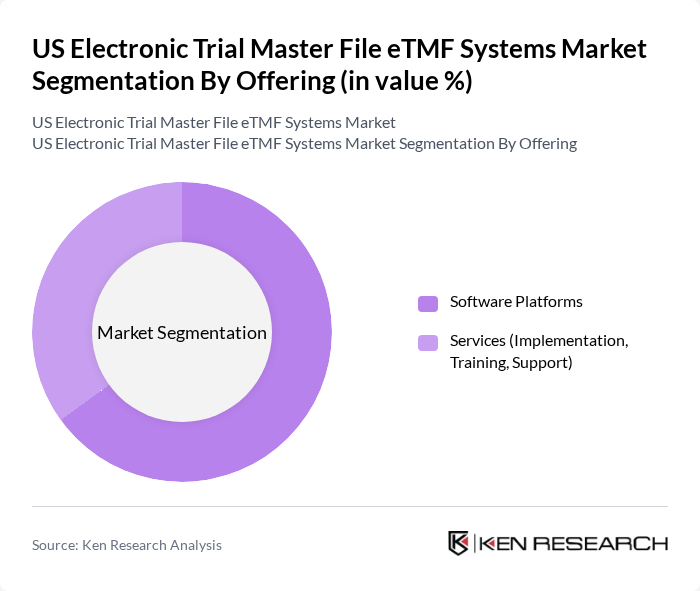
US Electronic Trial Master File eTMF Systems Market Segmentation
By Offering:The segmentation by offering includes Software Platforms and Services (Implementation, Training, Support). The Software Platforms segment is currently leading the market due to the increasing demand for comprehensive solutions that facilitate centralized document management, real-time tracking, and automated compliance with regulatory standards. Services, while essential, are often seen as supplementary to the core software offerings, providing configuration, integration, user training, and managed services that support efficient implementation and ongoing optimization of eTMF environments in clinical trials.
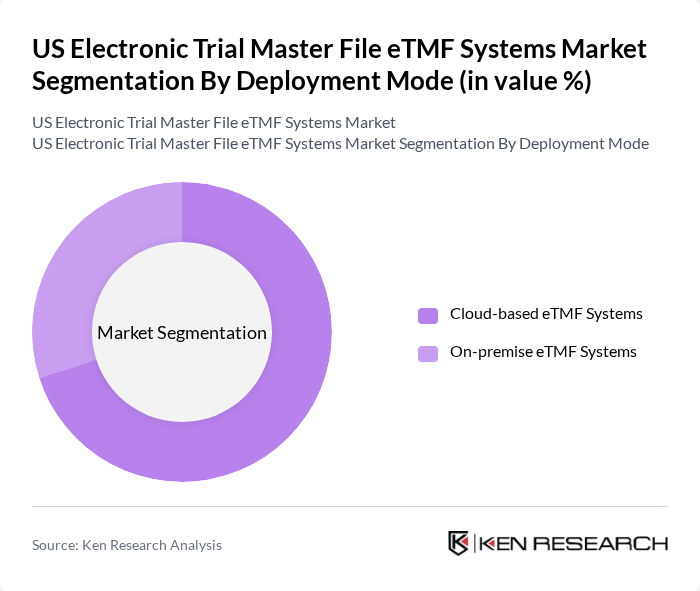
By Deployment Mode:The deployment mode segmentation includes Cloud-based eTMF Systems and On-premise eTMF Systems. Cloud-based solutions are dominating the market due to their scalability, cost-effectiveness, rapid deployment, and ease of access across dispersed study sites, which is particularly important for decentralized and hybrid clinical trial models. On-premise systems, while still relevant in organizations with specific data residency or legacy infrastructure requirements, are gradually declining as sponsors and CROs increasingly prefer the flexibility, automatic upgrades, and lower upfront capital expenditure associated with cloud solutions.
US Electronic Trial Master File eTMF Systems Market Competitive Landscape
The US Electronic Trial Master File eTMF Systems Market is characterized by a dynamic mix of regional and international players. Leading participants such as Veeva Systems, Medidata Solutions (Dassault Systèmes), Oracle Corporation, MasterControl, ArisGlobal, Phlexglobal, Trial Interactive (TransPerfect), Florence Healthcare, IQVIA, eClinical Solutions, Calyx, Mednet, Clinical Ink, MasterControl (eTMF & QMS Suite), Other Emerging US-focused eTMF Vendors contribute to innovation, geographic expansion, and service delivery in this space.
US Electronic Trial Master File eTMF Systems Market Industry Analysis
Growth Drivers
- Increasing Demand for Efficient Clinical Trial Management:The US clinical trial market is projected to reach $24 billion in future, driven by the need for streamlined processes. Efficient management of clinical trials is crucial, as the average cost of bringing a drug to market is approximately $2.8 billion. This demand for efficiency is pushing organizations to adopt eTMF systems that enhance collaboration and reduce time-to-market, ultimately improving patient outcomes and operational efficiency.
- Adoption of Cloud-Based Solutions:The cloud computing market in healthcare is expected to grow to $70 billion in future, reflecting a significant shift towards cloud-based eTMF solutions. These systems offer scalability, cost-effectiveness, and remote access, which are essential for managing clinical trials efficiently. With over 75% of clinical trial sponsors now utilizing cloud technologies, the trend is set to continue, enhancing data accessibility and collaboration among stakeholders.
- Regulatory Compliance Requirements:The US regulatory landscape mandates strict compliance with guidelines such as 21 CFR Part 11, which governs electronic records. In future, the FDA is expected to increase scrutiny on data integrity, pushing organizations to adopt eTMF systems that ensure compliance. With over 85% of clinical trials facing regulatory challenges, the need for compliant eTMF solutions is critical for maintaining operational integrity and avoiding costly penalties.
Market Challenges
- High Implementation Costs:The initial investment for eTMF systems can exceed $600,000, which poses a significant barrier for smaller organizations. This high cost includes software licensing, training, and integration with existing systems. As a result, many companies hesitate to transition from traditional paper-based systems, limiting their ability to leverage the benefits of digital solutions and hindering overall market growth.
- Resistance to Change from Traditional Systems:Approximately 65% of clinical trial professionals express reluctance to abandon traditional paper-based methods. This resistance stems from concerns about the learning curve associated with new technologies and the perceived risks of data loss during the transition. Such hesitance can slow the adoption of eTMF systems, ultimately impacting the efficiency and effectiveness of clinical trial management.
US Electronic Trial Master File eTMF Systems Market Future Outlook
The future of the US eTMF systems market is poised for significant transformation, driven by technological advancements and evolving regulatory landscapes. As organizations increasingly embrace decentralized clinical trials, the demand for real-time data access and patient-centric approaches will intensify. Furthermore, the integration of artificial intelligence and machine learning into eTMF systems is expected to enhance data management capabilities, streamline processes, and improve compliance, ultimately shaping the future of clinical trial management.
Market Opportunities
- Expansion in Emerging Markets:The growing pharmaceutical sectors in regions like Asia-Pacific are projected to reach $350 billion in future. This expansion presents opportunities for eTMF providers to penetrate new markets, offering tailored solutions that address local regulatory requirements and operational challenges, thereby enhancing global reach and revenue potential.
- Development of Mobile eTMF Solutions:With mobile device usage in healthcare expected to surpass 85% in future, the demand for mobile-compatible eTMF solutions is rising. These solutions facilitate on-the-go access to trial data, improving collaboration among stakeholders and enhancing overall trial efficiency, thus creating a lucrative opportunity for innovative eTMF providers.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Offering | Software Platforms Services (Implementation, Training, Support) |
| By Deployment Mode | Cloud-based eTMF Systems On-premise eTMF Systems |
| By Functionality | Core TMF Document Management Workflow & Process Management Audit Trails, Inspection Readiness & Compliance Reporting, Dashboards & Analytics |
| By Application | Clinical Trial Management Regulatory Compliance & Inspection Readiness Quality & Risk Management |
| By End-User | Pharmaceutical Companies Biotechnology Companies Contract Research Organizations (CROs) Academic Medical Centers & Research Institutions Other Sponsors & Clinical Sites |
| By Integration Capability | Integration with CTMS Integration with EDC/eCOA and Other eClinical Systems Integration via APIs & Middleware |
| By Region (US Census Regions) | Northeast Midwest South West |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, National Institutes of Health)
Pharmaceutical and Biotechnology Companies
Clinical Research Organizations
Healthcare Providers and Institutions
Technology Providers and Software Developers
Data Management and Compliance Specialists
Insurance Companies and Payers
Players Mentioned in the Report:
Veeva Systems
Medidata Solutions (Dassault Systemes)
Oracle Corporation
MasterControl
ArisGlobal
Phlexglobal
Trial Interactive (TransPerfect)
Florence Healthcare
IQVIA
eClinical Solutions
Calyx
Mednet
Clinical Ink
MasterControl (eTMF & QMS Suite)
Other Emerging US-focused eTMF Vendors
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. US Electronic Trial Master File eTMF Systems Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 US Electronic Trial Master File eTMF Systems Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. US Electronic Trial Master File eTMF Systems Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for efficient clinical trial management
3.1.2 Adoption of cloud-based solutions
3.1.3 Regulatory compliance requirements
3.1.4 Rising focus on data integrity and security
3.2 Market Challenges
3.2.1 High implementation costs
3.2.2 Resistance to change from traditional systems
3.2.3 Data privacy concerns
3.2.4 Limited interoperability with existing systems
3.3 Market Opportunities
3.3.1 Expansion in emerging markets
3.3.2 Integration of AI and machine learning
3.3.3 Increasing partnerships with CROs
3.3.4 Development of mobile eTMF solutions
3.4 Market Trends
3.4.1 Shift towards decentralized clinical trials
3.4.2 Growing emphasis on patient-centric approaches
3.4.3 Enhanced focus on real-time data access
3.4.4 Rise of collaborative platforms for trial management
3.5 Government Regulation
3.5.1 FDA guidelines on electronic records
3.5.2 HIPAA compliance for data protection
3.5.3 21 CFR Part 11 regulations
3.5.4 GCP guidelines for clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. US Electronic Trial Master File eTMF Systems Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. US Electronic Trial Master File eTMF Systems Market Segmentation
8.1 By Offering
8.1.1 Software Platforms
8.1.2 Services (Implementation, Training, Support)
8.2 By Deployment Mode
8.2.1 Cloud-based eTMF Systems
8.2.2 On-premise eTMF Systems
8.3 By Functionality
8.3.1 Core TMF Document Management
8.3.2 Workflow & Process Management
8.3.3 Audit Trails, Inspection Readiness & Compliance
8.3.4 Reporting, Dashboards & Analytics
8.4 By Application
8.4.1 Clinical Trial Management
8.4.2 Regulatory Compliance & Inspection Readiness
8.4.3 Quality & Risk Management
8.5 By End-User
8.5.1 Pharmaceutical Companies
8.5.2 Biotechnology Companies
8.5.3 Contract Research Organizations (CROs)
8.5.4 Academic Medical Centers & Research Institutions
8.5.5 Other Sponsors & Clinical Sites
8.6 By Integration Capability
8.6.1 Integration with CTMS
8.6.2 Integration with EDC/eCOA and Other eClinical Systems
8.6.3 Integration via APIs & Middleware
8.7 By Region (US Census Regions)
8.7.1 Northeast
8.7.2 Midwest
8.7.3 South
8.7.4 West
9. US Electronic Trial Master File eTMF Systems Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Revenue Tier: Large / Mid / Small)
9.2.3 US eTMF-Specific Revenue (Latest Year)
9.2.4 3-year Revenue CAGR in eTMF Segment
9.2.5 Number of Active eTMF Deployments / Clients in the US
9.2.6 Average Annual Contract Value (ACV)
9.2.7 Gross Margin (%) on eTMF Offerings
9.2.8 Customer Retention / Net Revenue Retention (%)
9.2.9 Win Rate in Competitive Bids (%)
9.2.10 Average Implementation Time (Months)
9.2.11 Uptime / SLA Performance (%)
9.2.12 R&D Spend on eTMF as % of Revenue
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Veeva Systems
9.5.2 Medidata Solutions (Dassault Systèmes)
9.5.3 Oracle Corporation
9.5.4 MasterControl
9.5.5 ArisGlobal
9.5.6 Phlexglobal
9.5.7 Trial Interactive (TransPerfect)
9.5.8 Florence Healthcare
9.5.9 IQVIA
9.5.10 eClinical Solutions
9.5.11 Calyx
9.5.12 Mednet
9.5.13 Clinical Ink
9.5.14 MasterControl (eTMF & QMS Suite)
9.5.15 Other Emerging US-focused eTMF Vendors
10. US Electronic Trial Master File eTMF Systems Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Vendor Selection Criteria
10.1.4 Contracting Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Patterns
10.2.3 Budget Constraints
10.2.4 Future Spending Projections
10.3 Pain Point Analysis by End-User Category
10.3.1 Common Challenges Faced
10.3.2 Technology Adoption Barriers
10.3.3 Support and Maintenance Issues
10.3.4 Compliance and Regulatory Hurdles
10.4 User Readiness for Adoption
10.4.1 Training and Support Needs
10.4.2 Change Management Strategies
10.4.3 Technology Familiarity Levels
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Case Opportunities
10.5.4 Lessons Learned from Deployments
11. US Electronic Trial Master File eTMF Systems Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategies
9.1.3 Packaging Approaches
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnership Dynamics
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from regulatory bodies such as the FDA and EMA
- Review of published white papers and case studies on eTMF adoption
- Examination of market trends and forecasts from research institutions and consultancy firms
Primary Research
- Interviews with clinical trial managers and regulatory affairs professionals
- Surveys targeting IT managers in pharmaceutical and biotech companies
- Field interviews with end-users of eTMF systems in clinical research organizations
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews
- Triangulation of data from primary and secondary sources to ensure consistency
- Sanity checks through feedback from industry panels and focus groups
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total market size based on overall clinical trial expenditure in the US
- Segmentation of the market by therapeutic area and trial phase
- Incorporation of growth rates from historical data and emerging trends in digitalization
Bottom-up Modeling
- Collection of data on the number of active clinical trials and their average duration
- Estimation of eTMF system adoption rates among different trial sponsors
- Calculation of average spending on eTMF solutions per trial
Forecasting & Scenario Analysis
- Multi-variable forecasting based on regulatory changes and technological advancements
- Scenario analysis considering varying rates of eTMF adoption across sectors
- Development of baseline, optimistic, and pessimistic market growth projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Companies | 120 | Clinical Trial Managers, Regulatory Affairs Specialists |
| Biotechnology Firms | 90 | IT Managers, Project Managers |
| Clinical Research Organizations (CROs) | 70 | Operations Directors, Data Managers |
| Academic Research Institutions | 60 | Research Coordinators, Compliance Officers |
| Regulatory Bodies | 40 | Policy Makers, Compliance Analysts |
Frequently Asked Questions
What is the current value of the US Electronic Trial Master File eTMF Systems Market?
The US Electronic Trial Master File eTMF Systems Market is valued at approximately USD 400 million, reflecting a significant growth driven by the increasing adoption of digital solutions in clinical trials and the need for efficient data management systems.