Region:North America
Author(s):Geetanshi
Product Code:KRAC3093
Pages:87
Published On:October 2025
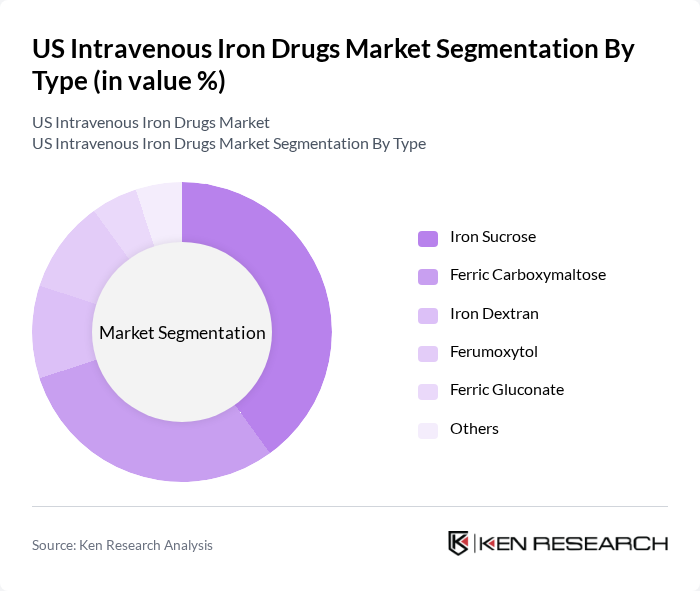
By Type:The market is segmented into various types of intravenous iron formulations, including Iron Sucrose, Ferric Carboxymaltose, Iron Dextran, Ferumoxytol, Ferric Gluconate, and Others. Among these, Iron Sucrose remains the most widely used due to its established safety profile and efficacy in treating iron deficiency anemia in diverse patient populations. Ferric Carboxymaltose is also gaining traction, favored for its convenient dosing regimen and suitability for outpatient care. The increasing adoption of newer formulations such as Ferumoxytol is driven by their favorable safety and efficacy profiles, especially in patients with chronic kidney disease and those intolerant to other iron formulations .
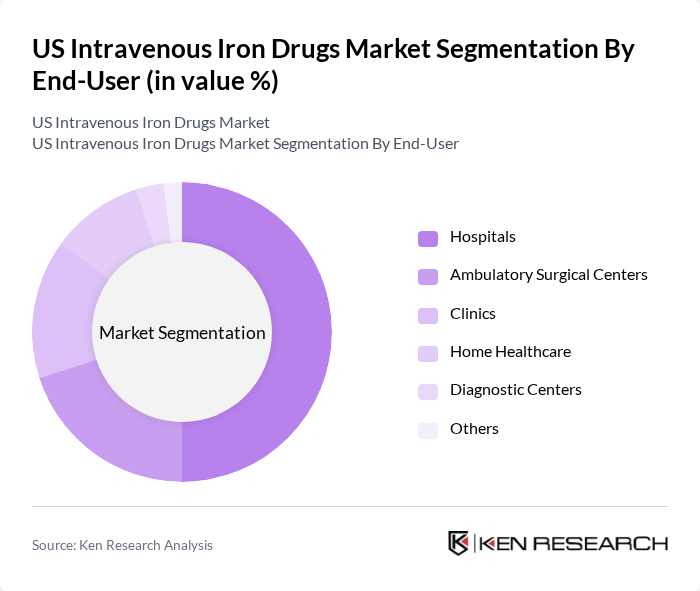
By End-User:The end-user segmentation includes Hospitals, Ambulatory Surgical Centers, Clinics, Home Healthcare, Diagnostic Centers, and Others. Hospitals are the leading end-users of intravenous iron drugs, primarily due to their capacity to manage complex cases and provide comprehensive care. Ambulatory Surgical Centers are also increasingly adopting these therapies, driven by the growing trend of outpatient treatments and the need for efficient patient management. The expansion of home healthcare and specialty clinics is further supporting the adoption of intravenous iron therapies outside traditional hospital settings .
The US Intravenous Iron Drugs Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Vifor Pharma AG, Fresenius Kabi AG, American Regent, Inc. (a Daiichi Sankyo Group company), Hikma Pharmaceuticals PLC, Sandoz International GmbH, Pfizer Inc., B. Braun Melsungen AG, Takeda Pharmaceutical Company Limited, Mallinckrodt Pharmaceuticals, Sanofi S.A., Aurobindo Pharma Limited, Pharmacosmos A/S, CSL Behring LLC, Grifols S.A. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the U.S. intravenous iron drugs market appears promising, driven by ongoing advancements in drug formulations and a growing emphasis on personalized medicine. As healthcare providers increasingly adopt technology-driven solutions, including telemedicine, the management of iron deficiency anemia is expected to become more efficient. Additionally, the expansion of home healthcare services will likely facilitate greater access to intravenous therapies, enhancing patient outcomes and overall market dynamics in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Iron Sucrose Ferric Carboxymaltose Iron Dextran Ferumoxytol Ferric Gluconate Others |
| By End-User | Hospitals Ambulatory Surgical Centers Clinics Home Healthcare Diagnostic Centers Others |
| By Administration Route | Intravenous Injection Intravenous Infusion |
| By Patient Demographics | Pediatric Patients Adult Patients Geriatric Patients |
| By Distribution Channel | Direct Sales Distributors Online Sales |
| By Pricing Tier | Premium Mid-range Economy |
| By Region | Northeast Midwest South West |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hematology Clinics | 60 | Hematologists, Nurse Practitioners |
| Hospital Pharmacy Departments | 50 | Pharmacy Directors, Clinical Pharmacists |
| Patient Advocacy Groups | 40 | Patient Representatives, Caregivers |
| Dialysis Centers | 45 | Nephrologists, Dialysis Nurses |
| Healthcare Policy Makers | 40 | Health Economists, Policy Analysts |
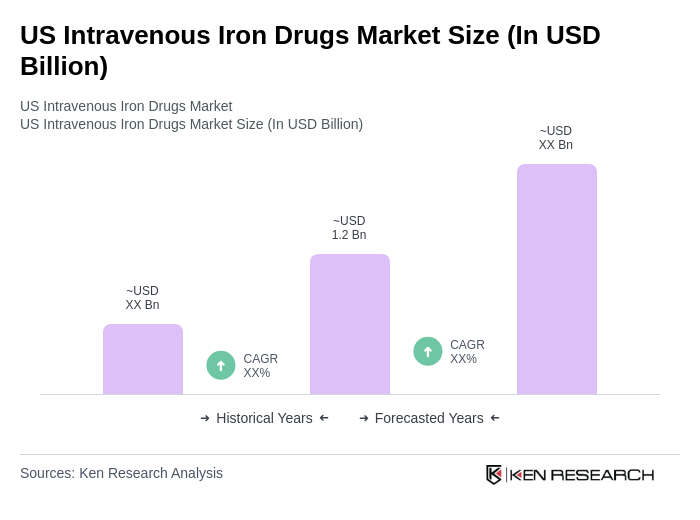
The US Intravenous Iron Drugs Market is valued at approximately USD 1.2 billion, reflecting a significant growth driven by the rising prevalence of iron deficiency anemia and advancements in intravenous iron therapies.