Region:Asia
Author(s):Shubham
Product Code:KRAC8930
Pages:94
Published On:November 2025
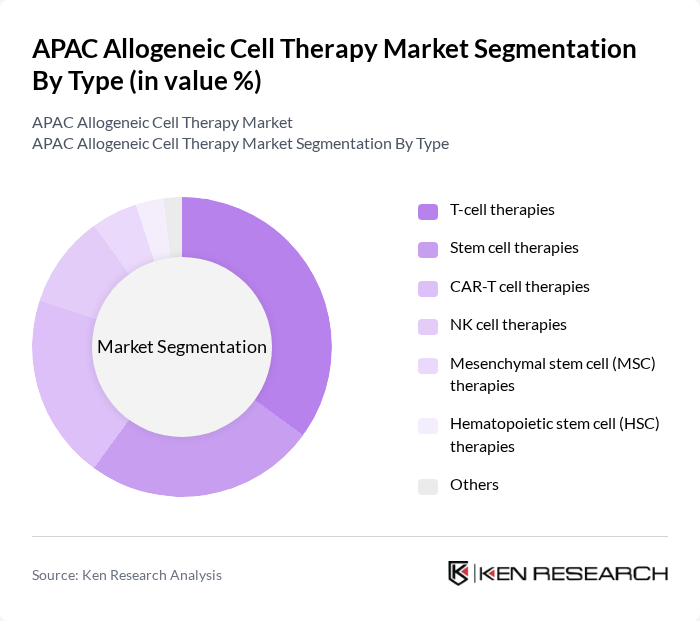
By Type:The market is segmented into various types of therapies, including T-cell therapies, stem cell therapies, CAR-T cell therapies, NK cell therapies, mesenchymal stem cell (MSC) therapies, hematopoietic stem cell (HSC) therapies, and others. Among these, T-cell therapies are currently leading the market due to their effectiveness in treating hematologic malignancies and solid tumors. The increasing number of clinical trials and regulatory approvals for T-cell therapies has significantly contributed to their dominance, as they are widely recognized for their potential to provide long-lasting remissions in patients. CAR-T cell therapies and NK cell therapies are also experiencing rapid adoption, driven by breakthroughs in oncology and immunotherapy.
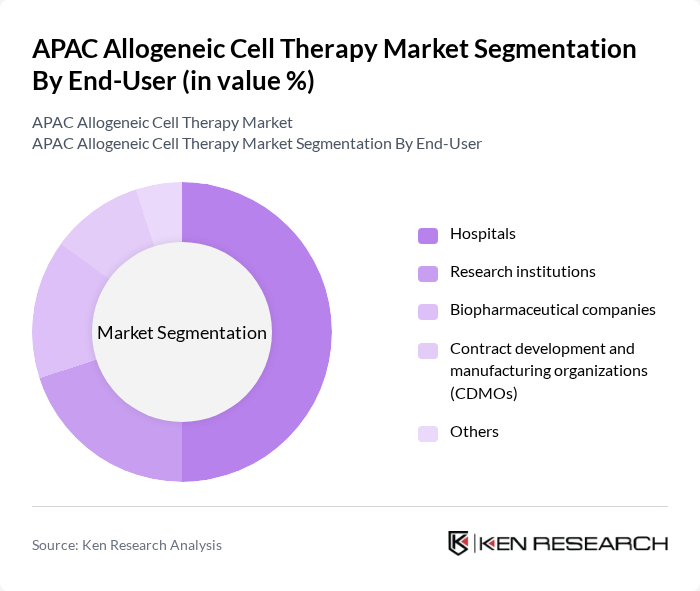
By End-User:The end-user segmentation includes hospitals, research institutions, biopharmaceutical companies, contract development and manufacturing organizations (CDMOs), and others. Hospitals are the leading end-users, primarily due to the increasing number of patients requiring advanced therapies and the growing adoption of cell therapies in clinical practice. The demand for innovative treatment options in hospitals is driving investments in infrastructure and training, further solidifying their position as the dominant end-user in the market. Biopharmaceutical companies and CDMOs are also expanding their roles, reflecting the region’s focus on R&D and scalable manufacturing.
The APAC Allogeneic Cell Therapy Market is characterized by a dynamic mix of regional and international players. Leading participants such as Takeda Pharmaceutical Company Limited, Fosun Kite Biotechnology Co., Ltd., JW Therapeutics Co. Ltd., Shanghai Cellular Biopharmaceutical Group Co., Ltd. (CBMG), Kolon Life Science, Inc., JCR Pharmaceuticals Co., Ltd., Medipost Co., Ltd., GC Cell Corporation, Chugai Pharmaceutical Co., Ltd., Sumitomo Pharma Co., Ltd., Novartis AG, Gilead Sciences, Inc., Bristol-Myers Squibb Company, Allogene Therapeutics, Inc., BlueRock Therapeutics LP contribute to innovation, geographic expansion, and service delivery in this space.
The future of the APAC allogeneic cell therapy market appears promising, driven by ongoing technological advancements and increasing healthcare investments. As the region continues to prioritize healthcare innovation, the integration of artificial intelligence in therapy development is expected to enhance efficiency and outcomes. Furthermore, the growing emphasis on patient-centric approaches will likely lead to more tailored therapies, improving patient engagement and satisfaction in treatment processes.
| Segment | Sub-Segments |
|---|---|
| By Type | T-cell therapies Stem cell therapies CAR-T cell therapies NK cell therapies Mesenchymal stem cell (MSC) therapies Hematopoietic stem cell (HSC) therapies Others |
| By End-User | Hospitals Research institutions Biopharmaceutical companies Contract development and manufacturing organizations (CDMOs) Others |
| By Application | Oncology (hematologic and solid tumors) Autoimmune diseases Cardiovascular diseases Neurological disorders Orthopedic disorders Others |
| By Delivery Method | Intravenous Intratumoral Subcutaneous Intra-arterial Others |
| By Source of Cells | Donor-derived cells (peripheral blood, bone marrow, umbilical cord, adipose tissue) Induced pluripotent stem cells (iPSCs) Others |
| By Region | East Asia (China, Japan, South Korea) Southeast Asia (Singapore, Malaysia, Thailand, Indonesia, Vietnam, Philippines) South Asia (India, Pakistan, Bangladesh) Oceania (Australia, New Zealand) Others |
| By Regulatory Pathway | Fast track approval Standard approval Conditional approval Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hematopoietic Stem Cell Therapy | 60 | Hematologists, Oncologists |
| T-cell Therapy Applications | 55 | Immunologists, Clinical Researchers |
| Regulatory Perspectives on Cell Therapies | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Patient Experience and Outcomes | 50 | Patients, Caregivers, Patient Advocacy Groups |
| Market Access and Reimbursement Strategies | 45 | Health Economists, Payers, Policy Makers |
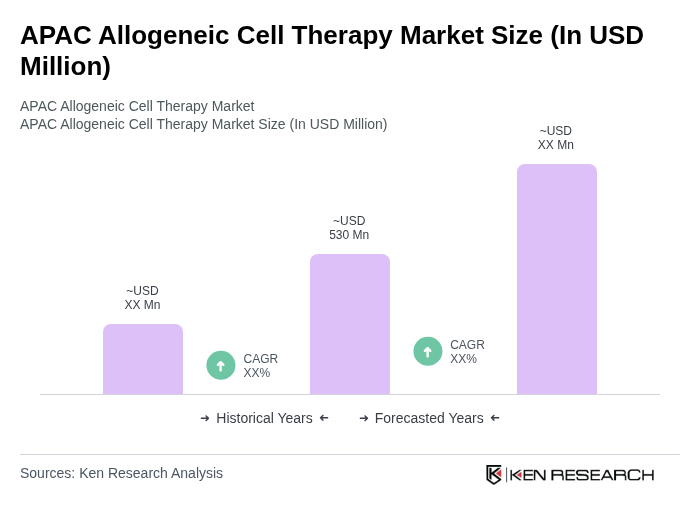
The APAC Allogeneic Cell Therapy Market is valued at approximately USD 530 million, driven by advancements in biotechnology, increased prevalence of chronic diseases, and rising investments in research and development.