Region:Asia
Author(s):Geetanshi
Product Code:KRAC8369
Pages:82
Published On:November 2025
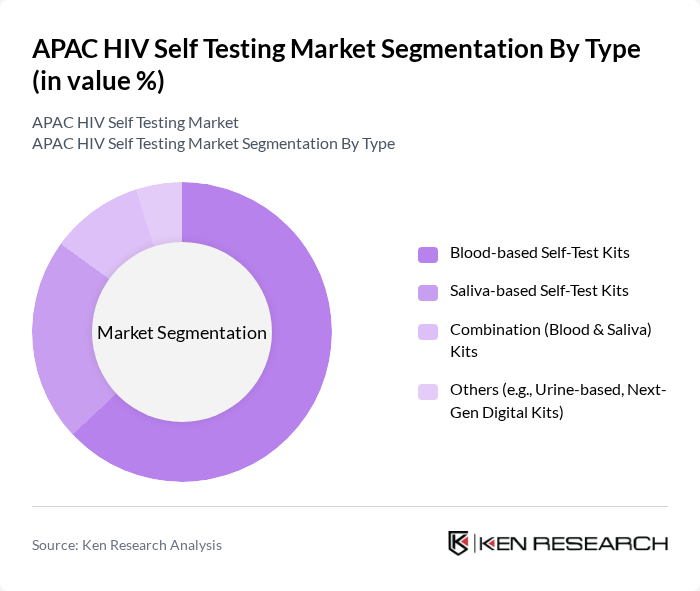
By Type:The market is segmented into various types of self-test kits, including blood-based, saliva-based, combination kits, and others. Blood-based self-test kits are currently the most popular due to their high accuracy and clinical recommendation, accounting for the majority of sales. Saliva-based kits are gaining traction for their ease of use and non-invasiveness, especially among first-time testers and younger populations. Combination kits, which offer both blood and saliva testing, appeal to consumers seeking comprehensive results and flexibility. The "Others" category includes urine-based and next-generation digital kits, which are emerging as innovative solutions for niche and tech-savvy segments .
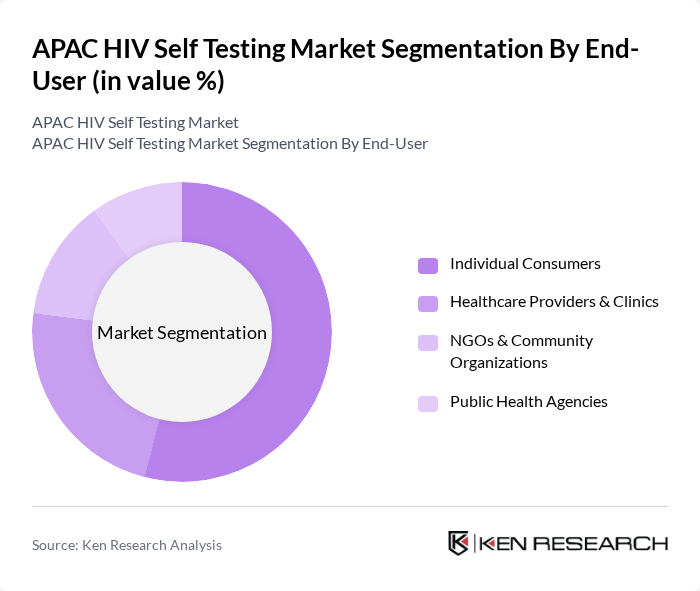
By End-User:The end-user segmentation includes individual consumers, healthcare providers and clinics, NGOs and community organizations, and public health agencies. Individual consumers represent the largest segment, driven by the growing trend of self-testing for privacy, convenience, and stigma reduction. Healthcare providers and clinics play a significant role in distributing self-testing kits and supporting linkage to care. NGOs and community organizations are crucial in reaching high-risk and underserved populations, while public health agencies support testing initiatives through funding, policy, and awareness campaigns .
The APAC HIV Self Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics, OraSure Technologies, Inc., BioSure (UK) Ltd, Mylan N.V. (Viatris Inc.), Cepheid, Hologic, Inc., Siemens Healthineers AG, Alere Inc. (now part of Abbott), Sedia Biosciences Corporation, MedMira Inc., ACON Laboratories, Inc., Chembio Diagnostics, Inc., Atomo Diagnostics, and Wondfo Biotech Co., Ltd. contribute to innovation, geographic expansion, and service delivery in this space .
The APAC HIV self-testing market is poised for significant transformation, driven by technological advancements and a shift towards preventive healthcare. As telehealth services expand, more individuals will have access to remote consultations, facilitating the use of self-testing kits. Additionally, the integration of mobile technology in testing processes is expected to enhance user experience and engagement. With increasing consumer preference for privacy, the market is likely to see innovative solutions that cater to these evolving needs, fostering a more proactive approach to HIV management.
| Segment | Sub-Segments |
|---|---|
| By Type | Blood-based Self-Test Kits Saliva-based Self-Test Kits Combination (Blood & Saliva) Kits Others (e.g., Urine-based, Next-Gen Digital Kits) |
| By End-User | Individual Consumers Healthcare Providers & Clinics NGOs & Community Organizations Public Health Agencies |
| By Country/Region | China India Japan Australia Southeast Asia (e.g., Thailand, Vietnam, Indonesia) Rest of APAC |
| By Distribution Channel | Online Pharmacies & E-commerce Brick-and-Mortar Pharmacies Hospitals & Clinics Community-Based Distribution |
| By Consumer Demographics | Age Group (18-24, 25-34, 35-44, 45+) Gender Socioeconomic Status Key Populations (MSM, Sex Workers, etc.) |
| By Testing Frequency | Regular Testing Occasional Testing One-time Testing Others |
| By Product Features | Ease of Use Accuracy/Sensitivity Speed of Results Digital Connectivity/Companion App |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Healthcare Providers | 120 | Doctors, Nurses, Public Health Officials |
| At-risk Individuals | 85 | Individuals aged 18-50, LGBTQ+ community members |
| Community Organizations | 65 | NGO Representatives, Outreach Coordinators |
| Pharmaceutical Distributors | 55 | Sales Managers, Product Managers |
| Regulatory Bodies | 45 | Policy Makers, Health Administrators |
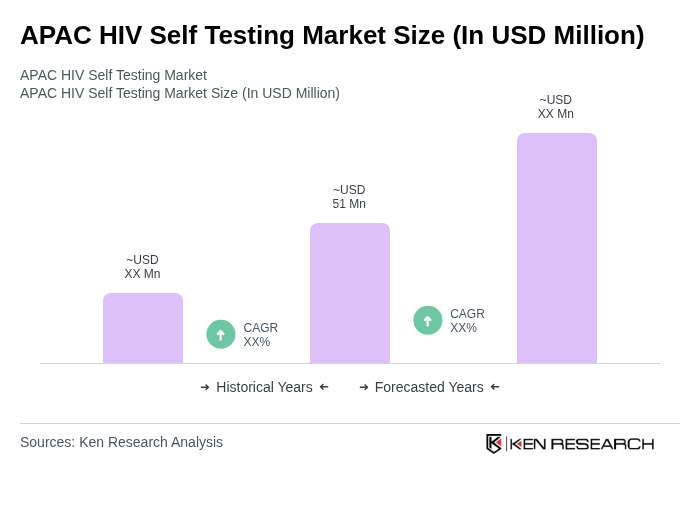
The APAC HIV Self Testing Market is valued at approximately USD 51 million, reflecting a significant growth driven by increased awareness of HIV status, government initiatives promoting self-testing, and the rising prevalence of HIV in the region.