About the Report
Base Year 2024Bahrain Acute External Ventricular Drain Market Overview
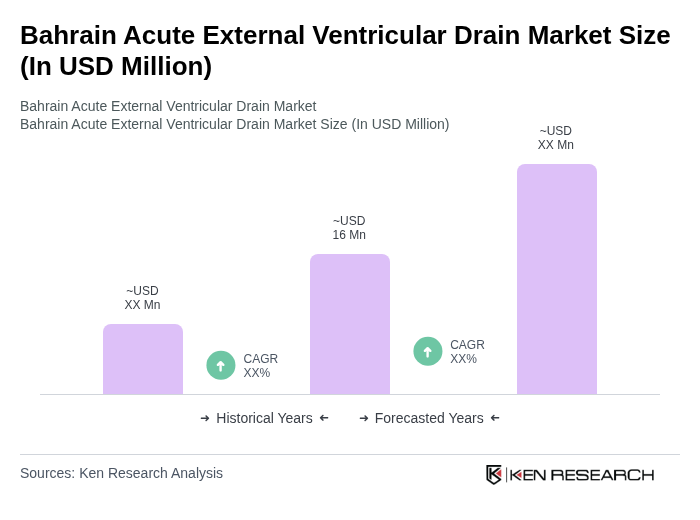
- The Bahrain Acute External Ventricular Drain market is valued at USD 16 million, based on a five-year historical analysis and extrapolation from global and Middle Eastern benchmarks in the external ventricular drain sector. This growth is primarily driven by the increasing prevalence of neurological disorders, advancements in medical technology, the expansion of tertiary care neurosurgical capacity, and the rising number of neurosurgical procedures performed in the country. The demand for effective drainage solutions in critical care settings, including management of traumatic brain injury, subarachnoid hemorrhage, and hydrocephalus, has significantly contributed to the market's expansion.
- Key cities such as Manama and Muharraq dominate the market due to their advanced healthcare infrastructure and concentration of tertiary care hospitals. These urban centers host the major government and private hospitals in the Capital and Muharraq Governorates, which are equipped with specialized neurosurgery centers that cater to a higher volume of patients requiring acute external ventricular drainage, thus driving the demand for these medical devices.
- External ventricular drains and EVD catheters used in Bahrain are regulated under the medical device framework of the National Health Regulatory Authority (NHRA), including requirements for registration, import licensing, and compliance with safety and performance standards as set out in instruments such as the “NHRA Medical Devices Regulation, 2015” issued by the National Health Regulatory Authority. These rules govern classification, conformity assessment, post?market surveillance, and infection?control related performance for invasive neurosurgical devices, and hospitals increasingly adopt antimicrobial and antibiotic-impregnated EVD catheters—aligned with global infection-prevention guidelines—to reduce catheter?associated ventriculitis and improve neurosurgical outcomes.
Bahrain Acute External Ventricular Drain Market Segmentation
By Product:The product segmentation includes various types of external ventricular drain systems that cater to different clinical needs. The subsegments are External Ventricular Drain Sets, EVD Catheters (including antimicrobial/antibiotic-impregnated), EVD Accessories and Consumables (bags, connectors, stopcocks), and Integrated ICP Monitoring EVD Systems. Each of these products plays a crucial role in the management of intracranial pressure and cerebrospinal fluid drainage, with antimicrobial/antibiotic-impregnated catheters and integrated ICP monitoring systems increasingly used to lower infection risk and enable more precise neurocritical care.
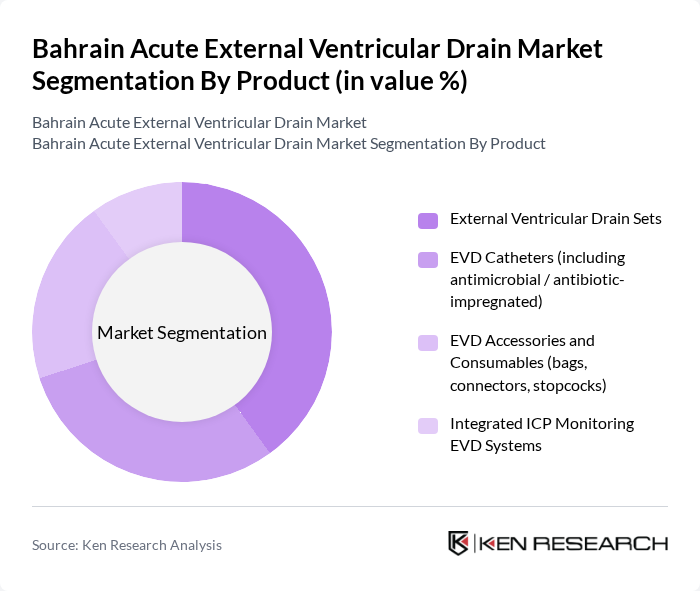
The External Ventricular Drain Sets segment is currently dominating the market due to their essential role in neurosurgical procedures. These sets are widely used in hospitals for managing patients with elevated intracranial pressure, particularly in critical care and emergency settings for conditions such as traumatic brain injury, subarachnoid hemorrhage, and acute hydrocephalus. The increasing incidence of traumatic brain injuries and other neurological conditions, supported by global data showing a high burden of road-traffic injuries and neurovascular events in the Middle East and Africa, has led to a higher demand for these drain sets. Additionally, advancements in technology—such as closed drainage systems, more accurate pressure transducers, and infection?control features—have improved the efficacy and safety of these products, further driving their adoption in healthcare facilities.
By Indication / Application:The application segmentation includes various medical conditions that necessitate the use of external ventricular drainage. The subsegments are Traumatic Brain Injury (TBI), Subarachnoid Hemorrhage, Intracerebral / Intraventricular Hemorrhage, and Acute Hydrocephalus and Other CSF Disorders. Each application represents a critical area where EVD systems are utilized to manage cerebrospinal fluid effectively and to monitor and control intracranial pressure in neurocritical care patients.
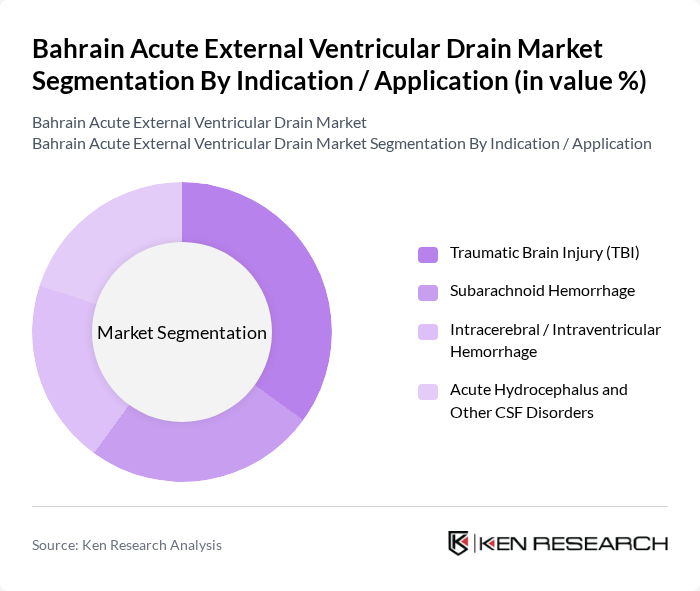
The Traumatic Brain Injury (TBI) segment is the leading application for external ventricular drainage systems, in line with global practice where EVDs are a standard of care for intracranial pressure monitoring and cerebrospinal fluid diversion in severe TBI. The rising incidence of TBIs due to road-traffic accidents and falls in the broader Middle East and Africa region has significantly increased the demand for effective drainage solutions. Hospitals are increasingly adopting EVD systems to manage intracranial pressure in TBI patients, which has led to advancements in product design and functionality, including integration with multimodal neuromonitoring platforms and improved infection?control features. The focus on improving patient outcomes in critical care settings, supported by investments in intensive care and neurosurgical capacity in Gulf Cooperation Council countries, further solidifies the dominance of this application in the market.
Bahrain Acute External Ventricular Drain Market Competitive Landscape
The Bahrain Acute External Ventricular Drain Market is characterized by a dynamic mix of regional and international players. Leading participants such as Medtronic plc, Johnson & Johnson MedTech (Codman neurosurgery legacy portfolio), B. Braun Melsungen AG, Integra LifeSciences Holdings Corporation, Sophysa SA, Möller Medical GmbH, Neuromedex GmbH, Natus Medical Incorporated, Medline Industries, LP, Stryker Corporation, Terumo Corporation, Fresenius Kabi AG, Local & Regional Distributors (Bahrain & GCC-based medical suppliers), Other Emerging and Niche EVD Manufacturers, Contract Manufacturers and OEM Partners contribute to innovation, geographic expansion, and service delivery in this space.
Bahrain Acute External Ventricular Drain Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Neurological Disorders:The World Health Organization reported that neurological disorders affect approximately 1 in 6 people globally, with a significant rise in conditions such as stroke and traumatic brain injuries. In Bahrain, the number of reported neurological cases has increased by 10% over the past five years, leading to a higher demand for acute external ventricular drains. This growing patient population necessitates advanced medical interventions, driving market growth in the region.
- Advancements in Medical Technology:The Bahrain healthcare sector has seen a 15% increase in investment in medical technology over the last three years, fostering innovation in devices like external ventricular drains. Enhanced imaging techniques and minimally invasive surgical methods have improved patient outcomes, making these devices more effective. As hospitals adopt cutting-edge technologies, the demand for advanced external ventricular drains is expected to rise, further propelling market growth.
- Rising Healthcare Expenditure:Bahrain's healthcare expenditure is projected to reach approximately $2.1 billion in future, reflecting a 5% increase from the previous period. This rise is driven by government initiatives to improve healthcare services and infrastructure. Increased funding allows for the procurement of advanced medical devices, including external ventricular drains, thereby enhancing treatment options for patients with acute neurological conditions and supporting market expansion.
Market Challenges
- High Cost of Advanced Medical Devices:The cost of advanced external ventricular drains can exceed $1,200 per unit, which poses a significant barrier for many healthcare facilities in Bahrain. Budget constraints and limited financial resources often lead to prioritization of essential services over advanced technologies. This challenge restricts the widespread adoption of these devices, hindering market growth despite the increasing need for effective neurological treatments.
- Limited Access to Specialized Healthcare:In Bahrain, only 25% of healthcare facilities are equipped with specialized neurological departments, limiting access to necessary treatments. This lack of infrastructure results in delayed diagnoses and treatments for patients requiring external ventricular drains. Consequently, the market faces challenges in reaching its full potential, as many patients are unable to receive timely and appropriate care for their neurological conditions.
Bahrain Acute External Ventricular Drain Market Future Outlook
The Bahrain Acute External Ventricular Drain market is poised for significant growth, driven by technological advancements and increasing healthcare investments. As the government continues to enhance healthcare infrastructure, the integration of innovative medical devices will likely improve patient outcomes. Additionally, the rising prevalence of neurological disorders will necessitate more effective treatment options. The market is expected to adapt to these trends, focusing on patient-centric care and the incorporation of telemedicine solutions to address access challenges.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Bahraini government plans to invest $800 million in healthcare infrastructure in future, which will enhance access to specialized services. This investment will create opportunities for the adoption of advanced medical devices, including external ventricular drains, thereby improving treatment options for patients with neurological disorders.
- Growth in Telemedicine and Remote Monitoring:The telemedicine market in Bahrain is expected to grow by 20% in future, driven by increased demand for remote healthcare services. This trend presents opportunities for integrating remote monitoring solutions with external ventricular drains, allowing for better patient management and follow-up care, ultimately enhancing treatment efficacy and patient satisfaction.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product | External Ventricular Drain Sets EVD Catheters (including antimicrobial / antibiotic-impregnated) EVD Accessories and Consumables (bags, connectors, stopcocks) Integrated ICP Monitoring EVD Systems |
| By Indication / Application | Traumatic Brain Injury (TBI) Subarachnoid Hemorrhage Intracerebral / Intraventricular Hemorrhage Acute Hydrocephalus and Other CSF Disorders |
| By End-User Facility | Tertiary Care Hospitals and Neurosurgery Centers Government / Public Hospitals Private Hospitals and Clinics Military and Specialized Emergency Care Facilities |
| By Care Setting | Intensive Care Units (ICU) Operating Rooms (OR) Emergency Departments Neurocritical Care Units |
| By Patient Type | Pediatric Patients Adult Patients Geriatric Patients Others |
| By Distribution Channel | Direct Tenders to Hospitals (Government & Private) Local Medical Device Distributors / Importers Group Purchasing Organizations (GPOs) & Regional Procurement Hubs Online / E-procurement Platforms |
| By Region (Governorate) | Northern Governorate Southern Governorate Capital Governorate Muharraq Governorate |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Bahrain National Health Regulatory Authority)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Importers and Exporters
Industry Associations and Societies
Health Insurance Companies
Players Mentioned in the Report:
Medtronic plc
Johnson & Johnson MedTech (Codman neurosurgery legacy portfolio)
B. Braun Melsungen AG
Integra LifeSciences Holdings Corporation
Sophysa SA
Moller Medical GmbH
Neuromedex GmbH
Natus Medical Incorporated
Medline Industries, LP
Stryker Corporation
Terumo Corporation
Fresenius Kabi AG
Local & Regional Distributors (Bahrain & GCC-based medical suppliers)
Other Emerging and Niche EVD Manufacturers
Contract Manufacturers and OEM Partners
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain Acute External Ventricular Drain Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain Acute External Ventricular Drain Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain Acute External Ventricular Drain Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of neurological disorders
3.1.2 Advancements in medical technology
3.1.3 Rising healthcare expenditure
3.1.4 Growing awareness of acute medical conditions
3.2 Market Challenges
3.2.1 High cost of advanced medical devices
3.2.2 Limited access to specialized healthcare
3.2.3 Regulatory hurdles in product approval
3.2.4 Shortage of trained medical professionals
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing demand for minimally invasive procedures
3.3.3 Collaborations with international medical firms
3.3.4 Growth in telemedicine and remote monitoring
3.4 Market Trends
3.4.1 Shift towards patient-centric care
3.4.2 Integration of AI in medical devices
3.4.3 Rise in outpatient surgical procedures
3.4.4 Focus on sustainable healthcare practices
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced safety standards for medical devices
3.5.3 Incentives for local manufacturing
3.5.4 Guidelines for clinical trials and approvals
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain Acute External Ventricular Drain Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain Acute External Ventricular Drain Market Segmentation
8.1 By Product
8.1.1 External Ventricular Drain Sets
8.1.2 EVD Catheters (including antimicrobial / antibiotic-impregnated)
8.1.3 EVD Accessories and Consumables (bags, connectors, stopcocks)
8.1.4 Integrated ICP Monitoring EVD Systems
8.2 By Indication / Application
8.2.1 Traumatic Brain Injury (TBI)
8.2.2 Subarachnoid Hemorrhage
8.2.3 Intracerebral / Intraventricular Hemorrhage
8.2.4 Acute Hydrocephalus and Other CSF Disorders
8.3 By End-User Facility
8.3.1 Tertiary Care Hospitals and Neurosurgery Centers
8.3.2 Government / Public Hospitals
8.3.3 Private Hospitals and Clinics
8.3.4 Military and Specialized Emergency Care Facilities
8.4 By Care Setting
8.4.1 Intensive Care Units (ICU)
8.4.2 Operating Rooms (OR)
8.4.3 Emergency Departments
8.4.4 Neurocritical Care Units
8.5 By Patient Type
8.5.1 Pediatric Patients
8.5.2 Adult Patients
8.5.3 Geriatric Patients
8.5.4 Others
8.6 By Distribution Channel
8.6.1 Direct Tenders to Hospitals (Government & Private)
8.6.2 Local Medical Device Distributors / Importers
8.6.3 Group Purchasing Organizations (GPOs) & Regional Procurement Hubs
8.6.4 Online / E-procurement Platforms
8.7 By Region (Governorate)
8.7.1 Northern Governorate
8.7.2 Southern Governorate
8.7.3 Capital Governorate
8.7.4 Muharraq Governorate
9. Bahrain Acute External Ventricular Drain Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Global and Regional EVD Revenues (USD Million)
9.2.3 5-year Revenue CAGR in EVD Portfolio
9.2.4 Bahrain EVD Market Share (%)
9.2.5 Install Base in Bahrain (Number of EVD Systems / Sets Sold Annually)
9.2.6 Average Selling Price (ASP) per EVD Set / Catheter
9.2.7 EBITDA Margin for EVD Segment (%)
9.2.8 R&D Intensity (% of Revenue Spent on EVD R&D)
9.2.9 New Product Launches in Last 3 Years (Count)
9.2.10 Tender Win Rate in Government / Public Hospitals (%)
9.2.11 Distribution Coverage (Number of Hospitals / Facilities Served in Bahrain)
9.2.12 Average Contract Duration and Renewal Rate
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Medtronic plc
9.5.2 Johnson & Johnson MedTech (Codman neurosurgery legacy portfolio)
9.5.3 B. Braun Melsungen AG
9.5.4 Integra LifeSciences Holdings Corporation
9.5.5 Sophysa SA
9.5.6 Möller Medical GmbH
9.5.7 Neuromedex GmbH
9.5.8 Natus Medical Incorporated
9.5.9 Medline Industries, LP
9.5.10 Stryker Corporation
9.5.11 Terumo Corporation
9.5.12 Fresenius Kabi AG
9.5.13 Local & Regional Distributors (Bahrain & GCC-based medical suppliers)
9.5.14 Other Emerging and Niche EVD Manufacturers
9.5.15 Contract Manufacturers and OEM Partners
10. Bahrain Acute External Ventricular Drain Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Supplier Selection Criteria
10.1.4 Contract Management Practices
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Priorities
10.2.2 Spending Patterns
10.2.3 Funding Sources
10.2.4 Future Projections
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges Faced by Hospitals
10.3.2 Issues in Specialty Clinics
10.3.3 Concerns in Rehabilitation Centers
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Case Studies of Successful Implementations
10.5.3 Future Use Cases
10.5.4 Others
11. Bahrain Acute External Ventricular Drain Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Strategy
9.1.3 Packaging Solutions
9.2 Export Entry Strategy
9.2.1 Target Countries Identification
9.2.2 Compliance Roadmap Development
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model Evaluation
11. Capital and Timeline Estimation
11.1 Capital Requirements Analysis
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability Strategies
14. Potential Partner List
14.1 Distributors Identification
14.2 Joint Ventures Opportunities
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup Activities
15.1.2 Market Entry Strategies
15.1.3 Growth Acceleration Plans
15.1.4 Scale & Stabilize Initiatives
15.2 Key Activities and Milestones
15.2.1 Milestone Tracking
15.2.2 Activity Scheduling
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of healthcare expenditure reports from the Bahrain Ministry of Health
- Review of published studies and white papers on external ventricular drain usage in neurosurgery
- Examination of market reports from regional healthcare associations and medical device regulatory bodies
Primary Research
- Interviews with neurosurgeons and critical care specialists in Bahrain
- Surveys with hospital procurement managers regarding purchasing patterns
- Field interviews with medical device distributors and suppliers in the region
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of market data with clinical usage statistics and hospital admission rates
- Sanity checks through feedback from a panel of healthcare professionals
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of total healthcare spending in Bahrain and allocation to neurosurgical procedures
- Analysis of the prevalence of neurological disorders requiring external ventricular drainage
- Incorporation of government healthcare initiatives and funding for neurosurgery
Bottom-up Modeling
- Volume estimates based on the number of surgeries performed annually in Bahraini hospitals
- Cost analysis of external ventricular drains and associated surgical procedures
- Assessment of market share held by various suppliers and manufacturers in the region
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering population growth and aging demographics
- Scenario modeling based on potential changes in healthcare policies and funding
- Baseline, optimistic, and pessimistic projections for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Neurosurgeons in Public Hospitals | 45 | Consultant Neurosurgeons, Surgical Residents |
| Neurosurgeons in Private Clinics | 40 | Private Practice Neurosurgeons, Clinic Managers |
| Hospital Procurement Managers | 35 | Procurement Officers, Supply Chain Managers |
| Medical Device Distributors | 30 | Sales Representatives, Regional Managers |
| Healthcare Policy Makers | 25 | Health Ministry Officials, Policy Analysts |
Frequently Asked Questions
What is the current value of the Bahrain Acute External Ventricular Drain market?
The Bahrain Acute External Ventricular Drain market is valued at approximately USD 16 million, reflecting a five-year historical analysis and extrapolation from global and Middle Eastern benchmarks in the external ventricular drain sector.