About the Report
Base Year 2024Bahrain External Counterpulsation Devices Market Overview
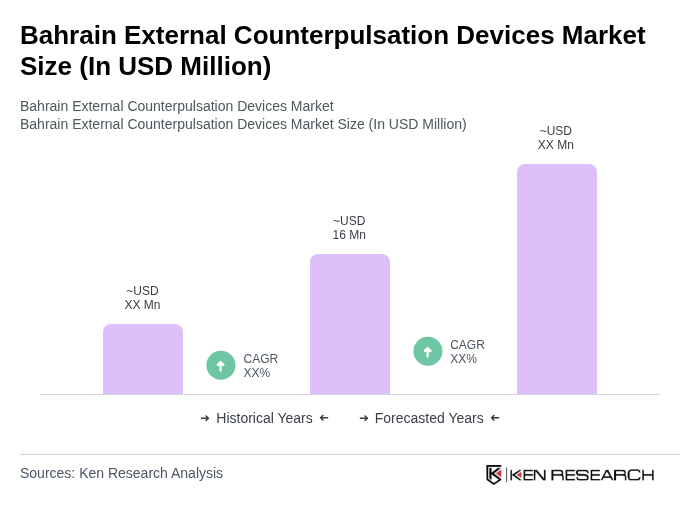
- The Bahrain External Counterpulsation Devices Market is valued at USD 16 million, based on a five-year historical analysis and triangulation with regional interventional cardiology and cardiovascular device spending trends. This growth is primarily driven by the increasing prevalence of cardiovascular diseases, which remain the leading cause of death in Bahrain and account for a substantial share of total mortality, alongside high levels of hypertension, obesity and diabetes in the population. The rising demand for non-invasive treatment options, together with advancements in hemodynamic monitoring, imaging, and cardiac rehabilitation technologies, and growing awareness among healthcare professionals regarding the role of external counterpulsation therapy as an adjunct for chronic coronary syndromes and heart failure, further support market expansion.
- Key demand clusters in this market include the Capital Governorate, which concentrates a significant number of tertiary hospitals and specialized cardiac centers, such as Salmaniya Medical Complex and several large private hospitals with comprehensive cardiology services. The region's advanced healthcare infrastructure, higher density of interventional cardiologists, and sustained investment in medical technologies and cardiac rehabilitation programs contribute to its dominance. Additionally, the Southern Governorate is emerging as a relevant growth area as new hospitals and specialty clinics are added to serve expanding residential developments and an increasing patient population with cardiovascular risk factors.
- In 2023, Bahrain's government implemented regulations to enhance the quality and safety of medical devices, including external counterpulsation devices. Under the Medical Devices Interim Regulation issued by the National Health Regulatory Authority (NHRA) in 2023, all medical devices must comply with NHRA classification rules, undergo technical and safety evaluation, and obtain NHRA registration and market authorization before being imported, distributed or used in healthcare facilities, with requirements that include conformity to recognized international standards, provision of clinical evaluation data and post?market vigilance obligations. These provisions ensure that only safe and effective products are made available to patients through licensed providers.
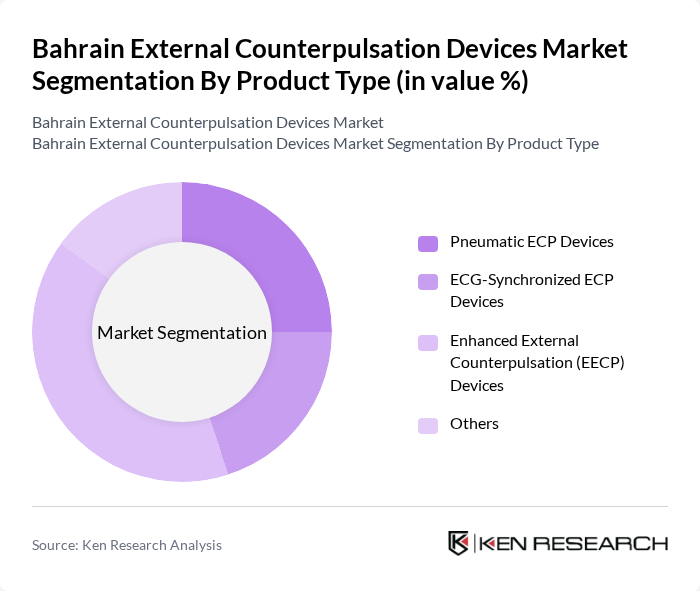
Bahrain External Counterpulsation Devices Market Segmentation
By Product Type:The product type segmentation includes Pneumatic ECP Devices, ECG-Synchronized ECP Devices, Enhanced External Counterpulsation (EECP) Devices, and Others. Among these, Enhanced External Counterpulsation (EECP) Devices are leading the market due to their effectiveness in treating chronic stable angina and heart failure symptoms in patients who are not ideal candidates for further revascularization, as documented in multiple clinical studies and guidelines discussing EECP as an adjunctive therapy. The growing preference for non-invasive treatment options, shorter recovery times, and outpatient-based therapies, together with the increasing number of international clinical studies supporting EECP therapy for improving exercise tolerance and quality of life, contribute to its dominance.
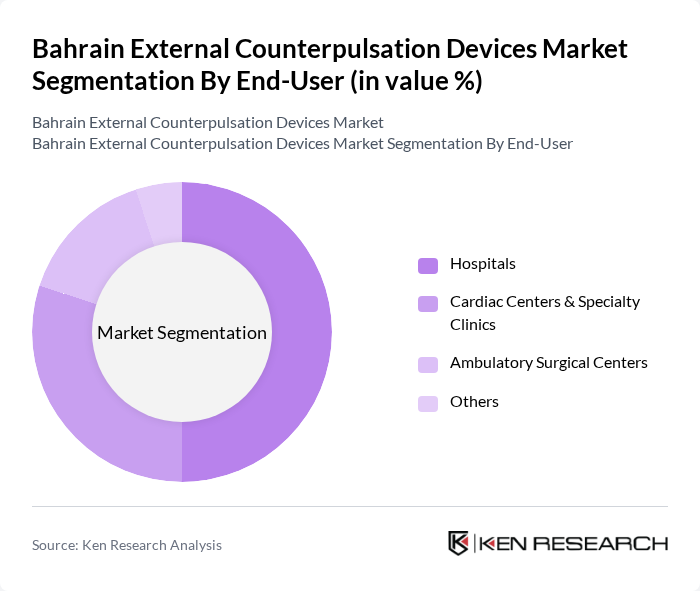
By End-User:The end-user segmentation includes Hospitals, Cardiac Centers & Specialty Clinics, Ambulatory Surgical Centers, and Others. Hospitals are the dominant end-user segment, primarily due to their comprehensive facilities and resources for treating cardiovascular diseases, including coronary care units, catheterization laboratories, and multidisciplinary cardiac teams. The increasing number of patients seeking treatment in hospitals for acute coronary syndromes, heart failure and post?intervention rehabilitation, along with the availability of advanced diagnostic and monitoring technologies, drives the demand for external counterpulsation devices in this segment. Cardiac centers and specialty clinics complement this demand by offering outpatient EECP programs, follow?up care and long?term disease management pathways.
Bahrain External Counterpulsation Devices Market Competitive Landscape
The Bahrain External Counterpulsation Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Vaso Corporation, Cardiomedics Inc., Scottcare Corporation, ACS Diagnostics, BTL Industries, Vasomedical Inc., PSK-Health Medical Equipment, Medtronic, Philips Healthcare, GE Healthcare, Siemens Healthineers, Abbott Laboratories, Boston Scientific, Edwards Lifesciences, Terumo Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Bahrain External Counterpulsation Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiovascular Diseases:The World Health Organization reported that cardiovascular diseases (CVDs) account for approximately 37% of all deaths in Bahrain, with over 3,200 fatalities annually. This alarming statistic underscores the urgent need for effective treatment options, including external counterpulsation devices. As the population ages, the number of individuals suffering from CVDs is projected to rise, driving demand for innovative, non-invasive therapies that can improve patient outcomes and reduce healthcare costs.
- Rising Awareness About Non-Invasive Treatment Options:A recent survey indicated that 65% of Bahraini healthcare professionals are increasingly aware of non-invasive treatment modalities for cardiovascular conditions. This growing awareness is crucial as it encourages patients to seek alternative therapies, such as external counterpulsation devices, which are associated with fewer complications and shorter recovery times. The shift towards non-invasive treatments is expected to significantly enhance the adoption of these devices in clinical settings across Bahrain.
- Technological Advancements in Counterpulsation Devices:The Bahrain healthcare sector is witnessing rapid technological advancements, with investments in medical device innovation reaching approximately $60 million in future. These advancements include improved device efficacy, user-friendly interfaces, and enhanced patient monitoring capabilities. As healthcare providers increasingly adopt these state-of-the-art devices, the market for external counterpulsation devices is expected to expand, offering better treatment options for patients with cardiovascular diseases.
Market Challenges
- High Initial Costs of Devices:The average cost of external counterpulsation devices in Bahrain ranges from $20,000 to $30,000, which poses a significant barrier to adoption, especially for smaller healthcare facilities. This high initial investment can deter hospitals from purchasing these devices, limiting patient access to advanced treatment options. As a result, many healthcare providers may opt for less expensive alternatives, hindering the growth of the counterpulsation device market in the region.
- Limited Awareness Among Healthcare Professionals:Despite the rising awareness among patients, a significant gap remains in the knowledge of external counterpulsation devices among healthcare professionals. A study revealed that only 40% of cardiologists in Bahrain are familiar with the benefits and applications of these devices. This lack of awareness can lead to underutilization and missed opportunities for patients who could benefit from non-invasive treatments, ultimately stunting market growth.
Bahrain External Counterpulsation Devices Market Future Outlook
The future of the Bahrain external counterpulsation devices market appears promising, driven by increasing healthcare investments and a growing emphasis on preventive care. As the government continues to enhance healthcare infrastructure, the adoption of innovative medical technologies is expected to rise. Additionally, the integration of telemedicine and remote monitoring solutions will likely facilitate better patient management, further propelling the demand for counterpulsation devices. This evolving landscape presents significant opportunities for market players to innovate and expand their offerings.
Market Opportunities
- Expansion of Healthcare Infrastructure:Bahrain's government is investing approximately $1 billion in healthcare infrastructure improvements by 2025. This investment will enhance access to advanced medical technologies, including external counterpulsation devices, thereby increasing their adoption in hospitals and clinics across the country.
- Growth in Telemedicine and Remote Monitoring:The telemedicine market in Bahrain is projected to reach $60 million in future, driven by increased demand for remote healthcare services. This growth presents an opportunity for integrating external counterpulsation devices with telehealth platforms, enabling better patient monitoring and follow-up care, ultimately improving treatment outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product Type | Pneumatic ECP Devices ECG?Synchronized ECP Devices Enhanced External Counterpulsation (EECP) Devices Others |
| By End-User | Hospitals Cardiac Centers & Specialty Clinics Ambulatory Surgical Centers Others |
| By Clinical Indication | Chronic Stable Angina Refractory Angina Chronic Heart Failure Others (e.g., Myocardial Ischemia, Cardiogenic Shock Adjunct) |
| By Distribution Channel | Direct Tenders (Public Hospitals & Institutions) Distributors & Local Agents Online / E?Procurement Platforms Others |
| By Region | Northern Governorate Southern Governorate Capital Governorate Muharraq Governorate |
| By Patient Demographics | Age Group (Adults, Seniors) Gender (Male, Female) Socioeconomic Status (Low, Middle, High) Others |
| By Technology Adoption | Early Adopters Mainstream Users Late Adopters Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Bahrain National Health Regulatory Authority)
Manufacturers and Producers of Medical Devices
Distributors and Retailers of Medical Equipment
Healthcare Providers and Hospitals
Health Insurance Companies
Industry Associations and Medical Societies
Financial Institutions and Investment Banks
Players Mentioned in the Report:
Vaso Corporation
Cardiomedics Inc.
Scottcare Corporation
ACS Diagnostics
BTL Industries
Vasomedical Inc.
PSK-Health Medical Equipment
Medtronic
Philips Healthcare
GE Healthcare
Siemens Healthineers
Abbott Laboratories
Boston Scientific
Edwards Lifesciences
Terumo Corporation
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain External Counterpulsation Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain External Counterpulsation Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain External Counterpulsation Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiovascular diseases
3.1.2 Rising awareness about non-invasive treatment options
3.1.3 Technological advancements in counterpulsation devices
3.1.4 Supportive government healthcare initiatives
3.2 Market Challenges
3.2.1 High initial costs of devices
3.2.2 Limited awareness among healthcare professionals
3.2.3 Regulatory hurdles in device approval
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing geriatric population
3.3.3 Potential for international collaborations
3.3.4 Growth in telemedicine and remote monitoring
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Integration of AI in medical devices
3.4.3 Focus on patient-centric healthcare solutions
3.4.4 Rise in outpatient treatment options
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced clinical trial requirements
3.5.3 Guidelines for device usage in hospitals
3.5.4 Incentives for local manufacturing
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain External Counterpulsation Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain External Counterpulsation Devices Market Segmentation
8.1 By Product Type
8.1.1 Pneumatic ECP Devices
8.1.2 ECG?Synchronized ECP Devices
8.1.3 Enhanced External Counterpulsation (EECP) Devices
8.1.4 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Cardiac Centers & Specialty Clinics
8.2.3 Ambulatory Surgical Centers
8.2.4 Others
8.3 By Clinical Indication
8.3.1 Chronic Stable Angina
8.3.2 Refractory Angina
8.3.3 Chronic Heart Failure
8.3.4 Others (e.g., Myocardial Ischemia, Cardiogenic Shock Adjunct)
8.4 By Distribution Channel
8.4.1 Direct Tenders (Public Hospitals & Institutions)
8.4.2 Distributors & Local Agents
8.4.3 Online / E?Procurement Platforms
8.4.4 Others
8.5 By Region
8.5.1 Northern Governorate
8.5.2 Southern Governorate
8.5.3 Capital Governorate
8.5.4 Muharraq Governorate
8.6 By Patient Demographics
8.6.1 Age Group (Adults, Seniors)
8.6.2 Gender (Male, Female)
8.6.3 Socioeconomic Status (Low, Middle, High)
8.6.4 Others
8.7 By Technology Adoption
8.7.1 Early Adopters
8.7.2 Mainstream Users
8.7.3 Late Adopters
8.7.4 Others
9. Bahrain External Counterpulsation Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Global Revenue Tier)
9.2.3 Bahrain ECP Revenue (USD, Latest Year)
9.2.4 3-Year CAGR in Bahrain ECP Revenue
9.2.5 Installed Base in Bahrain (Number of Devices)
9.2.6 Market Share in Bahrain (%)
9.2.7 Average Selling Price Band in Bahrain
9.2.8 EBITDA Margin (%) – ECP Segment
9.2.9 R&D Intensity (% of Revenue)
9.2.10 Product Registration Count with NHRA
9.2.11 Tender Win Rate in Public Sector (%)
9.2.12 Average Contract Tenure (Years)
9.2.13 Customer Retention Rate (%)
9.2.14 Net Promoter Score (NPS)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Vaso Corporation
9.5.2 Cardiomedics Inc.
9.5.3 Scottcare Corporation
9.5.4 ACS Diagnostics
9.5.5 BTL Industries
9.5.6 Vasomedical Inc.
9.5.7 PSK-Health Medical Equipment
9.5.8 Medtronic
9.5.9 Philips Healthcare
9.5.10 GE Healthcare
9.5.11 Siemens Healthineers
9.5.12 Abbott Laboratories
9.5.13 Boston Scientific
9.5.14 Edwards Lifesciences
9.5.15 Terumo Corporation
10. Bahrain External Counterpulsation Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Key Projects
10.2.3 Funding Sources
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Common Challenges Faced
10.3.2 Device Performance Issues
10.3.3 Cost-Effectiveness Concerns
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Acceptance Levels
10.4.3 Support Requirements
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Scenarios
10.5.3 Long-term Benefits
10.5.4 Others
11. Bahrain External Counterpulsation Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Components
1.3 Value Proposition Development
1.4 Revenue Streams
1.5 Cost Structure
1.6 Key Partnerships
1.7 Customer Segments
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Market Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
3.3 Online Distribution Channels
3.4 Logistics and Supply Chain Management
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments Analysis
5.3 Emerging Trends
5.4 Future Demand Projections
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
6.3 Customer Feedback Mechanisms
6.4 Relationship Management Strategies
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Customer-Centric Innovations
7.4 Competitive Advantages
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Management Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare associations and medical device regulatory bodies in Bahrain
- Review of published market studies and white papers on external counterpulsation devices
- Examination of demographic and health statistics from Bahrain's Ministry of Health
Primary Research
- Interviews with cardiologists and healthcare professionals specializing in non-invasive cardiac treatments
- Surveys with hospital procurement managers regarding purchasing trends and device preferences
- Focus groups with patients who have undergone treatment using external counterpulsation devices
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and stakeholder feedback
- Triangulation of data from healthcare providers, manufacturers, and regulatory agencies
- Sanity checks through comparative analysis with similar markets in the GCC region
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and cardiology service budgets
- Segmentation of the market by device type, application, and end-user demographics
- Incorporation of government health initiatives promoting non-invasive treatments
Bottom-up Modeling
- Collection of sales data from leading manufacturers of external counterpulsation devices operating in Bahrain
- Estimation of device usage rates based on patient population and treatment protocols
- Cost analysis based on pricing models and reimbursement rates from health insurance providers
Forecasting & Scenario Analysis
- Multi-variable forecasting using trends in cardiovascular disease prevalence and treatment advancements
- Scenario analysis based on potential regulatory changes and healthcare policy shifts
- Development of baseline, optimistic, and pessimistic market growth projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Clinics | 60 | Cardiologists, Clinic Managers |
| Hospitals with Cardiac Units | 60 | Procurement Officers, Department Heads |
| Health Insurance Providers | 40 | Policy Analysts, Claims Managers |
| Patient Advocacy Groups | 40 | Patient Representatives, Health Educators |
| Medical Device Distributors | 50 | Sales Managers, Product Specialists |
Frequently Asked Questions
What is the current value of the Bahrain External Counterpulsation Devices Market?
The Bahrain External Counterpulsation Devices Market is valued at approximately USD 16 million, reflecting a historical analysis and alignment with regional trends in interventional cardiology and cardiovascular device spending.