About the Report
Base Year 2024Australia External Counterpulsation Devices Market Overview
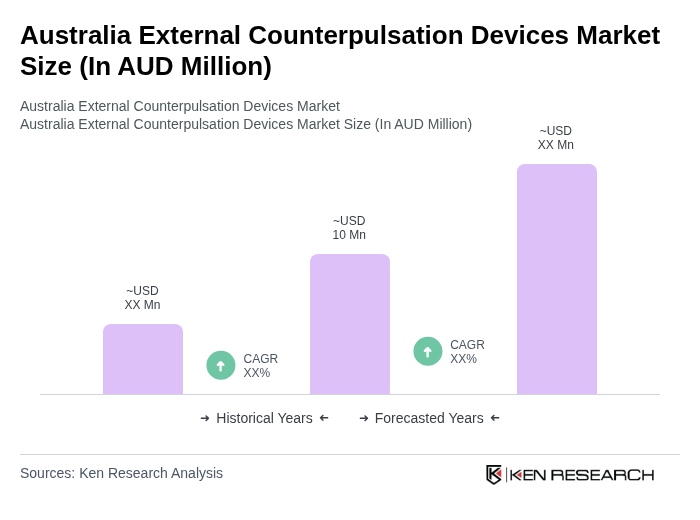
- The Australia External Counterpulsation Devices Market is valued at AUD 10 million, based on a five-year historical analysis and by benchmarking against the global external counterpulsation / EECP devices market and Australia’s share of the broader cardiovascular devices market. This growth is primarily driven by the increasing prevalence of cardiovascular diseases in Australia, rising awareness about non?invasive treatment options such as EECP for refractory angina and heart failure symptoms, and ongoing advancements in medical technology that enhance the efficacy and clinical adoption of external counterpulsation therapy.
- Key cities such as Sydney, Melbourne, and Brisbane dominate the market due to their advanced healthcare infrastructure, high concentration of tertiary cardiac care facilities, and a growing population of elderly individuals who are more susceptible to cardiovascular conditions. These factors contribute to a robust demand for external counterpulsation devices in these regions, supported by the presence of leading hospitals, specialist cardiology practices, and cardiac rehabilitation networks that are more likely to integrate EECP into multimodal management pathways for chronic coronary syndromes and heart failure patients.
- In 2023, the Australian government continued to strengthen regulations to ensure the safety and efficacy of medical devices, including external counterpulsation systems, under the Therapeutic Goods (Medical Devices) Regulations 2002 issued by the Australian Government and administered by the Therapeutic Goods Administration (TGA). This regulatory framework mandates that all medical devices must comply with the Essential Principles, undergo appropriate conformity assessment procedures, and be included in the Australian Register of Therapeutic Goods (ARTG) before being supplied for clinical use, thereby enhancing patient safety and promoting the adoption of high?quality medical technologies such as EECP systems.
Australia External Counterpulsation Devices Market Segmentation
By Product:The product segmentation includes EECP Systems and Accessories & Consumables (cuffs, hoses, ECG leads, others). The EECP systems are the primary focus due to their effectiveness in treating various cardiovascular conditions such as chronic stable angina and symptoms related to heart failure and left ventricular dysfunction, while accessories and consumables support the functionality, patient comfort, and ongoing usability of these systems during repeated treatment sessions.
The EECP Systems segment dominates the market due to their proven effectiveness in treating refractory angina and improving overall cardiovascular health, with EECP recognized as a non?invasive, FDA?approved therapy for chronic stable angina and used internationally as an adjunctive option in patients not suitable for revascularisation. The increasing adoption of these systems in hospitals and outpatient clinics, coupled with a growing body of clinical and real?world evidence supporting their use for symptom relief, functional capacity improvement and quality?of?life gains, has led to a significant rise in demand. Additionally, the trend towards non?invasive and cost?effective treatment options in cardiovascular care, including interest in EECP for indications such as microvascular angina and post?COVID?19 cardiovascular symptom management, has further solidified the position of EECP systems as a preferred choice among healthcare providers looking to expand therapeutic options beyond pharmacologic and interventional procedures.
By Mobility:The mobility segmentation includes Fixed Systems and Mobile / Portable Systems. Fixed systems are typically used in hospitals and cardiac centers, where they are integrated into dedicated EECP suites or cardiac rehabilitation units, while mobile systems offer flexibility for outpatient settings, satellite clinics, and potentially supervised home?based programs in selected patients.
Fixed Systems lead the market due to their widespread use in hospitals and cardiac centers, where they are integrated into comprehensive cardiac care programs, including multidisciplinary management of complex angina and heart failure patients. The reliability, higher throughput capacity, and advanced monitoring and safety features of fixed systems make them the preferred choice for healthcare providers handling higher patient volumes. However, the growing trend towards home care, ambulatory cardiac rehabilitation, and decentralized outpatient services is driving the demand for Mobile / Portable Systems, which offer convenience, improved access in regional areas, and scheduling flexibility for patients undergoing EECP treatment outside traditional tertiary care settings.
Australia External Counterpulsation Devices Market Competitive Landscape
The Australia External Counterpulsation Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Vaso Corporation, Renew Group Private Limited, ScottCare Cardiovascular Solutions, Vasomedical Inc., ACS Diagnostics Inc., Shenzhen Deyilong Technology Co., Ltd., Shanghai Goodmed Medical Device Co., Ltd., Medtronic plc (Australia Operations), Abbott Laboratories (Australia Operations), Boston Scientific Corporation (Australia Operations), GE HealthCare (Australia & New Zealand), Siemens Healthineers (Australia & New Zealand), Canon Medical Systems Corporation (Oceania), Local Cardiac Rehabilitation & EECP Clinics Network, Emerging Domestic EECP Technology Start?ups contribute to innovation, geographic expansion, and service delivery in this space.
Australia External Counterpulsation Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiovascular Diseases:The Australian Bureau of Statistics reported that cardiovascular diseases affect approximately 1.2 million Australians, accounting for around one in four deaths in the country. This alarming statistic drives the demand for effective treatment options, including external counterpulsation devices. With the healthcare expenditure projected to continue rising in future, the focus on innovative therapies to manage cardiovascular conditions is expected to intensify, further propelling market growth.
- Rising Awareness About Non-Invasive Treatment Options:A survey by the Heart Foundation indicated that 70% of Australians are now aware of non-invasive treatments for heart diseases. This growing awareness is crucial as it encourages patients to seek alternatives to traditional surgical procedures. The Australian healthcare system is increasingly promoting non-invasive therapies, which are expected to see a rise in adoption, thereby enhancing the market for external counterpulsation devices significantly.
- Technological Advancements in Medical Devices:The Australian medical device market is projected to grow to AUD 12 billion by 2024, driven by innovations in technology. Recent advancements in external counterpulsation devices, such as improved efficacy and user-friendly designs, are attracting healthcare providers. The integration of smart technologies and data analytics into these devices is enhancing patient outcomes, making them more appealing to both practitioners and patients, thus driving market growth.
Market Challenges
- High Cost of Devices:The average cost of external counterpulsation devices in Australia ranges from AUD 30,000 to AUD 50,000, which poses a significant barrier to adoption. Many healthcare facilities, especially smaller clinics, struggle to justify such high capital expenditures. This financial constraint limits the availability of these devices, hindering market penetration and growth, particularly in rural areas where healthcare budgets are tighter.
- Limited Reimbursement Policies:According to the Australian Government Department of Health, reimbursement for external counterpulsation therapy is limited, with only a few private health insurers covering these treatments. This lack of comprehensive reimbursement policies discourages healthcare providers from investing in these devices, as patients may be unable to afford out-of-pocket expenses. Consequently, this challenge restricts market growth and accessibility for patients in need of such therapies.
Australia External Counterpulsation Devices Market Future Outlook
The future of the external counterpulsation devices market in Australia appears promising, driven by an increasing focus on preventive healthcare and personalized medicine. As the healthcare landscape evolves, the integration of artificial intelligence and telemedicine is expected to enhance patient monitoring and treatment efficacy. Furthermore, the expansion of healthcare infrastructure will facilitate greater access to these devices, ensuring that more patients can benefit from non-invasive cardiovascular treatments in the coming years.
Market Opportunities
- Expansion of Healthcare Infrastructure:The Australian government plans to invest AUD 5 billion in healthcare infrastructure in future, which will enhance access to advanced medical technologies. This investment is likely to create opportunities for external counterpulsation devices, as more facilities will be equipped to offer these innovative treatments, ultimately benefiting patient care and market growth.
- Increasing Investment in R&D:The Australian medical research sector is projected to receive AUD 1.5 billion in funding in future, focusing on innovative healthcare solutions. This influx of investment will likely spur advancements in external counterpulsation technology, leading to improved device efficacy and patient outcomes. As research progresses, new applications for these devices may emerge, further expanding their market potential.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Product | EECP Systems Accessories & Consumables (cuffs, hoses, ECG leads, others) |
| By Mobility | Fixed Systems Mobile / Portable Systems |
| By Indication | Refractory Angina / Stable Angina Chronic Heart Failure Acute Myocardial Infarction Recovery Other Cardiovascular Indications (e.g., ischemic cardiomyopathy, left ventricular dysfunction) |
| By Care Setting | Hospitals & Cardiac Centers Outpatient Clinics & Specialized EECP Centers Home Care Settings |
| By Region | New South Wales Victoria Queensland Western Australia South Australia Rest of Australia |
| By Patient Profile | Age Group (Middle-aged Adults, Elderly) Risk Profile (High-risk CVD, Post?event Rehabilitation, Others) |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Therapeutic Goods Administration, Australian Competition and Consumer Commission)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Wholesalers
Industry Associations (e.g., Medical Technology Association of Australia)
Health Insurance Companies
Players Mentioned in the Report:
Vaso Corporation
Renew Group Private Limited
ScottCare Cardiovascular Solutions
Vasomedical Inc.
ACS Diagnostics Inc.
Shenzhen Deyilong Technology Co., Ltd.
Shanghai Goodmed Medical Device Co., Ltd.
Medtronic plc (Australia Operations)
Abbott Laboratories (Australia Operations)
Boston Scientific Corporation (Australia Operations)
GE HealthCare (Australia & New Zealand)
Siemens Healthineers (Australia & New Zealand)
Canon Medical Systems Corporation (Oceania)
Local Cardiac Rehabilitation & EECP Clinics Network
Emerging Domestic EECP Technology Startups
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Australia External Counterpulsation Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Australia External Counterpulsation Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Australia External Counterpulsation Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiovascular diseases
3.1.2 Rising awareness about non-invasive treatment options
3.1.3 Technological advancements in medical devices
3.1.4 Growing geriatric population
3.2 Market Challenges
3.2.1 High cost of devices
3.2.2 Limited reimbursement policies
3.2.3 Lack of trained professionals
3.2.4 Competition from alternative therapies
3.3 Market Opportunities
3.3.1 Expansion of healthcare infrastructure
3.3.2 Increasing investment in R&D
3.3.3 Collaborations with healthcare providers
3.3.4 Rising demand for home healthcare solutions
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Integration of AI in medical devices
3.4.3 Focus on preventive healthcare
3.4.4 Growth of telemedicine
3.5 Government Regulation
3.5.1 Therapeutic Goods Administration (TGA) guidelines
3.5.2 Medical Device Regulations
3.5.3 Compliance with international standards
3.5.4 Reporting adverse events
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Australia External Counterpulsation Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Australia External Counterpulsation Devices Market Segmentation
8.1 By Product
8.1.1 EECP Systems
8.1.2 Accessories & Consumables (cuffs, hoses, ECG leads, others)
8.2 By Mobility
8.2.1 Fixed Systems
8.2.2 Mobile / Portable Systems
8.3 By Indication
8.3.1 Refractory Angina / Stable Angina
8.3.2 Chronic Heart Failure
8.3.3 Acute Myocardial Infarction Recovery
8.3.4 Other Cardiovascular Indications (e.g., ischemic cardiomyopathy, left ventricular dysfunction)
8.4 By Care Setting
8.4.1 Hospitals & Cardiac Centers
8.4.2 Outpatient Clinics & Specialized EECP Centers
8.4.3 Home Care Settings
8.5 By Region
8.5.1 New South Wales
8.5.2 Victoria
8.5.3 Queensland
8.5.4 Western Australia
8.5.5 South Australia
8.5.6 Rest of Australia
8.6 By Patient Profile
8.6.1 Age Group (Middle-aged Adults, Elderly)
8.6.2 Risk Profile (High-risk CVD, Post?event Rehabilitation, Others)
9. Australia External Counterpulsation Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Global revenue band: Large, Mid, Emerging)
9.2.3 Australia EECP Revenue (AUD, latest year)
9.2.4 3?Year Australia EECP Revenue CAGR
9.2.5 Installed Base in Australia (number of EECP units)
9.2.6 New Installations per Year in Australia
9.2.7 Market Share in Australia EECP Devices (%)
9.2.8 EBITDA Margin – EECP Segment (%)
9.2.9 R&D Intensity (% of revenue)
9.2.10 Average Selling Price Band (per system, AUD)
9.2.11 Service & Maintenance Revenue Share (%)
9.2.12 Key Reference Sites / KOL Adoptions in Australia
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Vaso Corporation
9.5.2 Renew Group Private Limited
9.5.3 ScottCare Cardiovascular Solutions
9.5.4 Vasomedical Inc.
9.5.5 ACS Diagnostics Inc.
9.5.6 Shenzhen Deyilong Technology Co., Ltd.
9.5.7 Shanghai Goodmed Medical Device Co., Ltd.
9.5.8 Medtronic plc (Australia Operations)
9.5.9 Abbott Laboratories (Australia Operations)
9.5.10 Boston Scientific Corporation (Australia Operations)
9.5.11 GE HealthCare (Australia & New Zealand)
9.5.12 Siemens Healthineers (Australia & New Zealand)
9.5.13 Canon Medical Systems Corporation (Oceania)
9.5.14 Local Cardiac Rehabilitation & EECP Clinics Network
9.5.15 Emerging Domestic EECP Technology Start?ups
10. Australia External Counterpulsation Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Vendor Selection Criteria
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Budget Prioritization
10.2.3 Procurement Strategies
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Common Challenges Faced
10.3.2 Device Performance Issues
10.3.3 Cost-Effectiveness Concerns
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Requirements
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 ROI Measurement Techniques
10.5.2 Use Case Success Stories
10.5.3 Future Expansion Opportunities
10.5.4 Others
11. Australia External Counterpulsation Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
12. Control vs Risk Trade-Off
12.1 Ownership Considerations
12.2 Partnerships Evaluation
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from Australian health organizations and medical device associations
- Review of published market studies and white papers on external counterpulsation devices
- Examination of government health statistics and regulatory frameworks related to cardiovascular treatments
Primary Research
- Interviews with cardiologists and healthcare professionals specializing in non-invasive cardiac therapies
- Surveys with hospital procurement managers regarding purchasing trends and device preferences
- Field interviews with medical device distributors and suppliers in the Australian market
Validation & Triangulation
- Cross-validation of findings through multiple data sources, including clinical studies and market reports
- Triangulation of insights from primary interviews with secondary data trends
- Sanity checks conducted through expert panel reviews comprising industry veterans
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and cardiovascular disease prevalence
- Segmentation of the market by end-user categories, including hospitals and outpatient clinics
- Incorporation of government health initiatives promoting non-invasive treatment options
Bottom-up Modeling
- Volume estimates derived from sales data of leading external counterpulsation device manufacturers
- Operational cost analysis based on pricing models and reimbursement rates for treatments
- Calculation of market size based on device usage frequency and patient demographics
Forecasting & Scenario Analysis
- Multi-factor regression analysis incorporating trends in cardiovascular health awareness and technology adoption
- Scenario modeling based on potential changes in healthcare policies and reimbursement frameworks
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Clinics | 60 | Cardiologists, Clinic Managers |
| Hospitals with Cardiac Units | 80 | Procurement Officers, Medical Directors |
| Outpatient Rehabilitation Centers | 50 | Rehabilitation Specialists, Facility Administrators |
| Medical Device Distributors | 40 | Sales Managers, Product Specialists |
| Health Insurance Providers | 40 | Policy Analysts, Claims Managers |
Frequently Asked Questions
What is the current value of the Australia External Counterpulsation Devices Market?
The Australia External Counterpulsation Devices Market is valued at approximately AUD 10 million, reflecting a historical analysis and benchmarking against the global market for external counterpulsation devices and Australias share of the broader cardiovascular devices market.