About the Report
Base Year 2024Japan External Counterpulsation Devices Market Overview
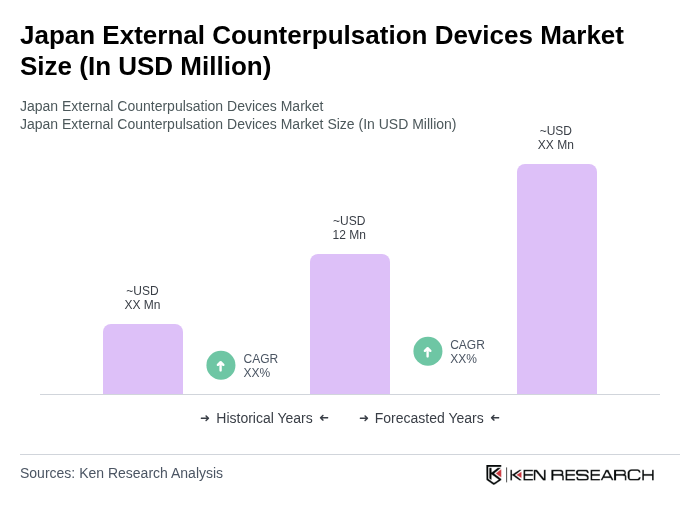
- The Japan External Counterpulsation Devices Market is valued at USD 12 million, based on a five-year historical analysis and Japan’s share within the global external counterpulsation and EECP devices market. This growth is primarily driven by the increasing prevalence of cardiovascular diseases, rising geriatric population, and advancements in medical technology. Japan has one of the highest proportions of older adults globally, and cardiovascular diseases remain a leading cause of morbidity, supporting demand for non-invasive therapies such as EECP. The demand for non-invasive treatment options has also been reinforced by wider clinical evidence for EECP in refractory angina and heart failure management, contributing to greater adoption of external counterpulsation devices in clinical and specialty cardiac rehabilitation settings.
- Key cities such as Tokyo, Osaka, and Yokohama dominate the market due to their advanced healthcare infrastructure, high concentration of hospitals and cardiac clinics, and significant investment in medical technology. Japan’s large cardiovascular care ecosystem is concentrated in major metropolitan areas, where university hospitals and high-volume cardiac centers drive utilization of advanced diagnostic and therapeutic technologies, including EECP systems, for complex coronary artery disease and heart failure cases.
- In 2023, the Japanese government implemented regulations to enhance the safety and efficacy of medical devices, including external counterpulsation systems. Under the Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices (PMD Act) enforced by the Ministry of Health, Labour and Welfare (MHLW) and the Pharmaceuticals and Medical Devices Agency (PMDA), external counterpulsation devices are regulated as therapeutic medical devices and must obtain marketing approval with evidence from clinical data, quality management (QMS) conformity, and post?marketing safety measures. In addition, the 2021 revision of the Pharmaceuticals and Medical Devices Act and related enforcement ordinances strengthened requirements for post?market surveillance, periodic safety reports, and field safety corrective actions for higher?risk device classes, which apply to cardiovascular and circulatory support systems. These frameworks mandate rigorous clinical evaluation, ongoing vigilance, and quality control, supporting high safety standards, clinician confidence, and broader institutional adoption of such technologies.
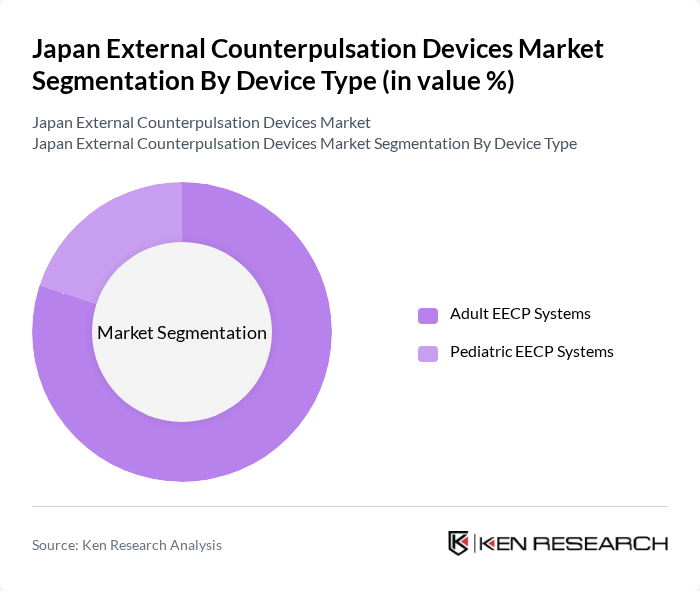
Japan External Counterpulsation Devices Market Segmentation
By Device Type:The market is segmented into Adult EECP Systems and Pediatric EECP Systems. Adult EECP Systems dominate the market due to the higher prevalence of ischemic heart disease, chronic stable angina, and heart failure among adults and older adults, leading to increased demand for non-invasive adjunctive treatment options, especially for patients who are not ideal candidates for revascularization. Pediatric EECP Systems, while important, cater to a much smaller patient population because coronary artery disease and chronic refractory angina are far less common in children, thus holding a lesser market share.
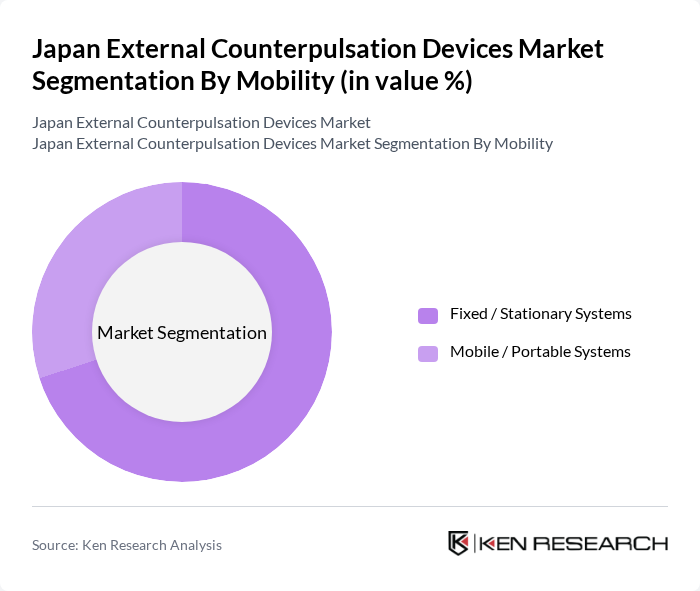
By Mobility:The market is divided into Fixed / Stationary Systems and Mobile / Portable Systems. Fixed / Stationary Systems are the leading segment, primarily due to their widespread use in hospitals, cardiovascular centers, and dedicated EECP clinics where patients receive treatment in a controlled environment with continuous monitoring. Mobile / Portable Systems are gaining traction, especially in outpatient and rehabilitation settings, home?oriented cardiac wellness programs, and smaller facilities seeking flexible deployment, but still represent a smaller portion of the market because most reimbursed EECP sessions are delivered within institutional care settings.
Japan External Counterpulsation Devices Market Competitive Landscape
The Japan External Counterpulsation Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as VasoMedical Inc., Vasomedical Global Corp. (EECP®), PSK-Health (Neo_cardial EECP), Vasogenics Japan Co., Ltd., Omay Medical Technology Co., Ltd., Guangzhou Yidian Medical Equipment Co., Ltd., ScottCare Corporation, Haobro Medical, Hongjiang Medical, VSK Medical, Major Japanese Medtech OEM Partners (Terumo, Nihon Kohden, Fukuda Denshi), Select Cardiac Rehabilitation Network Providers in Japan, Emerging Domestic EECP Start-ups, Regional Distributors and Service Partners, Contract Manufacturers & Component Suppliers contribute to innovation, geographic expansion, and service delivery in this space.
Japan External Counterpulsation Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiovascular Diseases:Cardiovascular diseases (CVDs) are a leading cause of mortality in Japan, with approximately 1.0 million deaths annually attributed to heart-related issues. The World Health Organization reported that CVDs account for 27% of all deaths in the country. This alarming statistic drives the demand for effective treatment options, including external counterpulsation devices, as healthcare providers seek innovative solutions to manage and treat these conditions effectively.
- Rising Awareness About Non-Invasive Treatment Options:The Japanese healthcare system is increasingly focusing on non-invasive treatment methods, with a reported 45% of patients preferring such options over surgical interventions. This shift is supported by educational campaigns and healthcare provider initiatives that highlight the benefits of external counterpulsation devices, which offer effective treatment for angina and heart failure without the risks associated with invasive procedures, thus expanding their market presence.
- Technological Advancements in Medical Devices:Japan is at the forefront of medical technology innovation, with the country investing approximately $50 billion in healthcare R&D in future. This investment has led to significant advancements in external counterpulsation devices, enhancing their efficacy and usability. The integration of smart technologies, such as real-time monitoring and data analytics, is expected to further boost the adoption of these devices, making them more appealing to healthcare providers and patients alike.
Market Challenges
- High Cost of Devices:The average cost of external counterpulsation devices in Japan ranges from $35,000 to $55,000, which poses a significant barrier to widespread adoption. Many healthcare facilities, especially smaller clinics, struggle to justify such high expenditures, particularly in a market where budget constraints are prevalent. This financial challenge limits access to these advanced treatment options for patients who could benefit from them.
- Limited Reimbursement Policies:The reimbursement landscape for external counterpulsation devices in Japan is restrictive, with only 30% of healthcare providers receiving adequate coverage for these treatments. This limitation discourages hospitals from investing in such technologies, as they face financial risks associated with unguaranteed reimbursements. Consequently, this challenge hampers the growth potential of the external counterpulsation devices market in the region.
Japan External Counterpulsation Devices Market Future Outlook
The future of the external counterpulsation devices market in Japan appears promising, driven by ongoing technological advancements and a growing emphasis on preventive healthcare. As the healthcare system increasingly prioritizes non-invasive treatment options, the demand for these devices is expected to rise. Additionally, the integration of artificial intelligence and telemedicine services will likely enhance patient outcomes and streamline treatment processes, further solidifying the role of external counterpulsation devices in cardiovascular care.
Market Opportunities
- Expansion into Rural Healthcare Facilities:There is a significant opportunity to introduce external counterpulsation devices in rural healthcare settings, where access to advanced cardiovascular treatments is limited. By targeting these underserved areas, manufacturers can tap into a new patient base, potentially increasing device adoption and improving health outcomes for rural populations.
- Collaborations with Healthcare Providers:Forming strategic partnerships with hospitals and clinics can enhance the market presence of external counterpulsation devices. Collaborations can facilitate training programs for healthcare professionals, ensuring they are well-equipped to utilize these devices effectively, thereby increasing their acceptance and usage in clinical settings.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Device Type | Adult EECP Systems Pediatric EECP Systems |
| By Mobility | Fixed / Stationary Systems Mobile / Portable Systems |
| By Application / Indication | Refractory Angina Chronic Heart Failure Myocardial Ischemia & Other CAD Cardiac Rehabilitation & Secondary Prevention |
| By End-User | Hospitals Cardiac Clinics & Specialty Centers Rehabilitation Centers Other Healthcare Facilities |
| By Component | Consoles & Controllers Cuffs & Inflation Systems Monitoring & Software Modules Consumables & Accessories |
| By Distribution Channel | Direct Tender / Institutional Sales Distributors & Dealers Online & Other Channels |
| By Region (Japan) | Kanto Kansai Chubu Hokkaido & Tohoku Chugoku & Shikoku Kyushu & Okinawa |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Ministry of Health, Labour and Welfare)
Manufacturers and Producers
Distributors and Retailers
Healthcare Providers and Hospitals
Medical Device Regulatory Agencies (e.g., Pharmaceuticals and Medical Devices Agency)
Industry Associations (e.g., Japan Medical Device Industry Association)
Health Insurance Companies
Players Mentioned in the Report:
VasoMedical Inc.
Vasomedical Global Corp. (EECP)
PSK-Health (Neo_cardial EECP)
Vasogenics Japan Co., Ltd.
Omay Medical Technology Co., Ltd.
Guangzhou Yidian Medical Equipment Co., Ltd.
ScottCare Corporation
Haobro Medical
Hongjiang Medical
VSK Medical
Major Japanese Medtech OEM Partners (Terumo, Nihon Kohden, Fukuda Denshi)
Select Cardiac Rehabilitation Network Providers in Japan
Emerging Domestic EECP Start-ups
Regional Distributors and Service Partners
Contract Manufacturers & Component Suppliers
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Japan External Counterpulsation Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Japan External Counterpulsation Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Japan External Counterpulsation Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiovascular diseases
3.1.2 Rising awareness about non-invasive treatment options
3.1.3 Technological advancements in medical devices
3.1.4 Growing geriatric population in Japan
3.2.1 High cost of devices
3.2.2 Limited reimbursement policies
3.2.3 Lack of trained professionals
3.2.4 Regulatory hurdles
3.3 Market Opportunities
3.3.1 Expansion into rural healthcare facilities
3.3.2 Collaborations with healthcare providers
3.3.3 Development of portable devices
3.3.4 Increasing investment in healthcare infrastructure
3.4 Market Trends
3.4.1 Shift towards preventive healthcare
3.4.2 Integration of AI in medical devices
3.4.3 Focus on patient-centric care
3.4.4 Rise in telemedicine services
3.5 Government Regulation
3.5.1 Stricter quality control measures
3.5.2 Enhanced approval processes for new devices
3.5.3 Incentives for local manufacturing
3.5.4 Guidelines for clinical trials
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Japan External Counterpulsation Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Japan External Counterpulsation Devices Market Segmentation
8.1 By Device Type
8.1.1 Adult EECP Systems
8.1.2 Pediatric EECP Systems
8.2 By Mobility
8.2.1 Fixed / Stationary Systems
8.2.2 Mobile / Portable Systems
8.3 By Application / Indication
8.3.1 Refractory Angina
8.3.2 Chronic Heart Failure
8.3.3 Myocardial Ischemia & Other CAD
8.3.4 Cardiac Rehabilitation & Secondary Prevention
8.4 By End-User
8.4.1 Hospitals
8.4.2 Cardiac Clinics & Specialty Centers
8.4.3 Rehabilitation Centers
8.4.4 Other Healthcare Facilities
8.5 By Component
8.5.1 Consoles & Controllers
8.5.2 Cuffs & Inflation Systems
8.5.3 Monitoring & Software Modules
8.5.4 Consumables & Accessories
8.6 By Distribution Channel
8.6.1 Direct Tender / Institutional Sales
8.6.2 Distributors & Dealers
8.6.3 Online & Other Channels
8.7 By Region (Japan)
8.7.1 Kanto
8.7.2 Kansai
8.7.3 Chubu
8.7.4 Hokkaido & Tohoku
8.7.5 Chugoku & Shikoku
8.7.6 Kyushu & Okinawa
9. Japan External Counterpulsation Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Japan EECP Revenue (US$ Mn)
9.2.4 3-year CAGR in Japan EECP Revenue (%)
9.2.5 Installed Base in Japan (Number of Systems)
9.2.6 Annual Procedure Volume (Number of Patients Treated)
9.2.7 Market Share in Japan EECP Market (%)
9.2.8 R&D Intensity (% of Revenue Spent on R&D)
9.2.9 Average Selling Price Band (US$ per System)
9.2.10 Gross Margin from EECP Portfolio (%)
9.2.11 Sales Coverage (Number of Prefectures Covered)
9.2.12 Key Hospital Accounts (Number of Tier-1 / University Hospitals)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 VasoMedical Inc.
9.5.2 Vasomedical Global Corp. (EECP®)
9.5.3 PSK-Health (Neo_cardial EECP)
9.5.4 Vasogenics Japan Co., Ltd.
9.5.5 Omay Medical Technology Co., Ltd.
9.5.6 Guangzhou Yidian Medical Equipment Co., Ltd.
9.5.7 ScottCare Corporation
9.5.8 Haobro Medical
9.5.9 Hongjiang Medical
9.5.10 VSK Medical
9.5.11 Major Japanese Medtech OEM Partners (Terumo, Nihon Kohden, Fukuda Denshi)
9.5.12 Select Cardiac Rehabilitation Network Providers in Japan
9.5.13 Emerging Domestic EECP Start-ups
9.5.14 Regional Distributors and Service Partners
9.5.15 Contract Manufacturers & Component Suppliers
10. Japan External Counterpulsation Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Suppliers
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Budget Prioritization
10.2.3 Procurement Strategies
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Device Availability
10.3.2 Training and Support
10.3.3 Cost Concerns
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Infrastructure Readiness
10.4.3 Financial Preparedness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 User Feedback
10.5.3 Expansion Opportunities
10.5.4 Others
11. Japan External Counterpulsation Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare associations and industry publications
- Review of government health statistics and regulatory frameworks related to medical devices
- Examination of academic journals and clinical studies focusing on external counterpulsation therapy
Primary Research
- Interviews with cardiologists and medical professionals specializing in heart disease treatment
- Surveys with hospital procurement managers regarding device purchasing decisions
- Field interviews with technicians and operators of counterpulsation devices in clinical settings
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and focus groups
- Triangulation of data from clinical outcomes, market trends, and regulatory insights
- Sanity checks through feedback from a panel of industry experts and stakeholders
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and cardiology device spending
- Segmentation by end-user categories such as hospitals, clinics, and rehabilitation centers
- Incorporation of demographic trends and prevalence rates of cardiovascular diseases
Bottom-up Modeling
- Collection of sales data from leading manufacturers of counterpulsation devices
- Estimation of device usage rates based on patient treatment protocols and clinical guidelines
- Volume x average selling price calculations to derive revenue projections
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors such as aging population and healthcare reforms
- Scenario modeling based on potential changes in reimbursement policies and technological advancements
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiology Departments in Hospitals | 120 | Cardiologists, Department Heads |
| Private Clinics Offering Cardiac Rehabilitation | 90 | Clinic Owners, Rehabilitation Specialists |
| Medical Device Distributors | 75 | Sales Managers, Product Specialists |
| Health Insurance Providers | 70 | Policy Analysts, Claims Managers |
| Research Institutions Focused on Cardiovascular Health | 60 | Researchers, Clinical Trial Coordinators |
Frequently Asked Questions
What is the current market value of the Japan External Counterpulsation Devices Market?
The Japan External Counterpulsation Devices Market is valued at approximately USD 12 million, driven by the increasing prevalence of cardiovascular diseases and advancements in medical technology, particularly in non-invasive treatment options.