About the Report
Base Year 2024Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Overview
- The Bahrain Spinal Muscular Atrophy (SMA) Treatment Market is valued at approximately USD 185 million, based on a five-year historical analysis. This growth is primarily driven by advancements in genetic therapies, increased awareness of SMA, and the rising prevalence of the disease in the region. The introduction of innovative treatments has significantly improved patient outcomes, leading to a higher demand for effective therapies. The global SMA treatment market continues to expand with strong momentum, reflecting the increasing adoption of disease-modifying therapies across developed and emerging markets.
- Key players in this market include Bahrain, Saudi Arabia, and the United Arab Emirates. These countries dominate the market due to their advanced healthcare infrastructure, significant investments in biotechnology, and a growing number of specialized clinics for SMA treatment. The presence of leading pharmaceutical companies and research institutions further enhances their market position. The GCC region has emerged as a focal point for rare disease management, with healthcare systems increasingly prioritizing access to breakthrough therapies and specialized medical expertise.
- Bahrain's Ministry of Health has implemented comprehensive healthcare policies aligned with regional standards for rare disease management. The country's healthcare system has adopted streamlined approval pathways for orphan drugs and gene therapies, facilitating faster market entry for innovative SMA treatments. This regulatory environment, combined with government support for rare disease awareness and patient access programs, ensures that cutting-edge therapeutic options are available and accessible to the patient population requiring specialized care.
 Treatment Market.png)
Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Segmentation
By Treatment Type:The treatment type segmentation includes various therapeutic approaches for SMA, focusing on innovative solutions to manage the disease effectively.
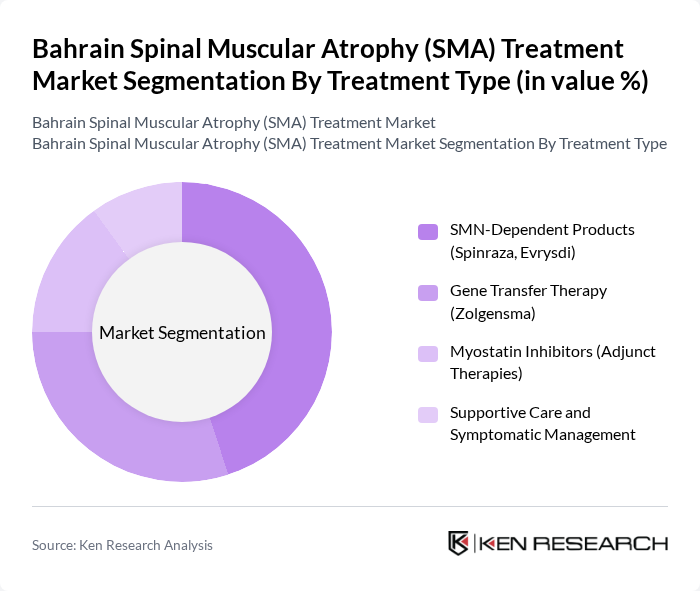
The SMN-Dependent Products segment, which includes Spinraza and Evrysdi, is currently dominating the market due to their proven efficacy in increasing survival and improving motor function in patients with SMA. The high demand for these therapies is driven by their ability to address the underlying genetic cause of the disease, leading to better patient outcomes. Additionally, the increasing awareness among healthcare providers and patients about the benefits of early intervention has further propelled the growth of this segment. The availability of these products in Bahrain has significantly improved treatment accessibility, making them the preferred choice for many healthcare professionals.
By Route of Administration:This segmentation focuses on the various methods through which SMA treatments are administered to patients.
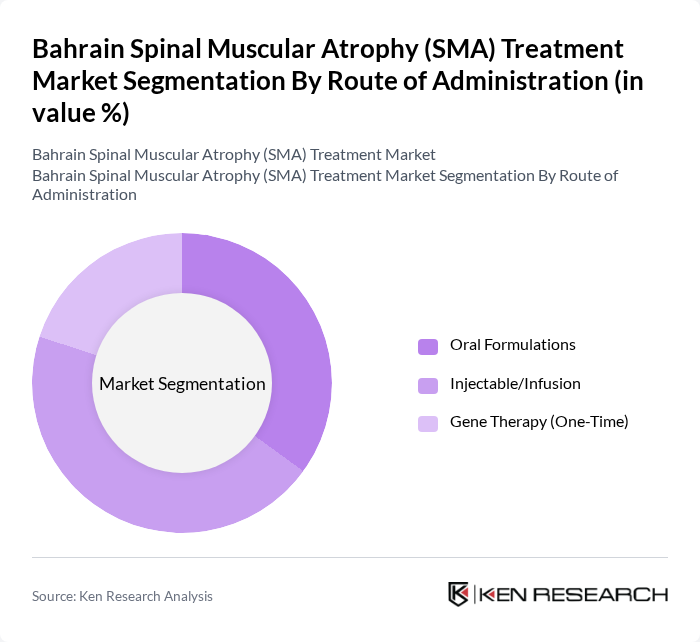
The Injectable/Infusion route of administration remains a significant segment in the market, primarily due to the nature of established therapies available for SMA, which often require direct delivery into the bloodstream for optimal efficacy. Treatments like Spinraza and Zolgensma are administered via injection, allowing for rapid absorption and action within the body. However, oral formulations have gained substantial market traction with the increasing adoption of agents like Evrysdi, which provides patient convenience and reduces the burden of frequent clinical visits. This method is preferred by healthcare providers as it ensures that the medication reaches the target areas effectively. The growing trend towards personalized medicine and the development of new oral and injectable therapies are expected to sustain balanced market dynamics across both segments.
Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Competitive Landscape
The Bahrain Spinal Muscular Atrophy (SMA) Treatment Market is characterized by a dynamic mix of regional and international players. Leading participants such as Novartis AG (Zolgensma Manufacturer), Roche Holding AG (Evrysdi Developer), Biogen Inc. (Spinraza Developer), Pfizer Inc., Scholar Rock Holding Corporation (Apitegromab Developer), Sarepta Therapeutics, Inc., PTC Therapeutics, Inc., Biohaven Pharmaceutical (Talditercept Alfa Developer), Sanofi S.A., Amgen Inc., GlaxoSmithKline PLC, Eli Lilly and Company, Merck & Co., Inc., Ionis Pharmaceuticals (ION306 Developer) contribute to innovation, geographic expansion, and service delivery in this space.
Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Industry Analysis
Growth Drivers
- Increasing Prevalence of SMA:The prevalence of Spinal Muscular Atrophy (SMA) in Bahrain is estimated at approximately 1 in 10,000 live births, translating to around 35 new cases annually. This rising incidence is driving demand for effective treatments. The World Health Organization (WHO) reported that the number of diagnosed SMA cases has increased by 20% over the past five years, highlighting the urgent need for innovative therapies and healthcare solutions tailored to this population.
- Advancements in Gene Therapy:Recent advancements in gene therapy have revolutionized SMA treatment options, with therapies like Zolgensma showing efficacy in clinical trials. The global market for gene therapies is projected to reach $110 billion by 2024, indicating a significant investment in research and development. In Bahrain, the Ministry of Health has allocated $6 million for gene therapy initiatives, enhancing access to cutting-edge treatments for SMA patients and fostering local clinical trials.
- Government Support and Funding:The Bahraini government has increased funding for rare diseases, including SMA, with a budget allocation of $3 million in 2024 specifically for SMA research and treatment programs. This support is crucial for developing new therapies and improving patient access to existing treatments. Additionally, the establishment of a national registry for rare diseases aims to enhance data collection and facilitate better healthcare planning and resource allocation.
Market Challenges
- High Cost of Innovative Treatments:Innovative SMA treatments, such as gene therapies, can cost upwards of $2.5 million per patient, creating significant financial barriers for families and healthcare systems. The high cost limits accessibility, with only 15% of patients currently receiving these therapies in Bahrain. This financial strain is compounded by limited insurance coverage for rare diseases, necessitating urgent policy reforms to improve affordability and access to life-saving treatments.
- Limited Access to Specialized Healthcare Facilities:Bahrain has only three specialized centers for SMA treatment, which are often overwhelmed by patient demand. With a population of approximately 1.8 million, the limited number of facilities results in long wait times and inadequate care for SMA patients. The World Bank reports that healthcare access in Bahrain is hindered by geographical disparities, necessitating investments in expanding specialized healthcare infrastructure to meet the growing needs of SMA patients.
Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Future Outlook
The future of the SMA treatment market in Bahrain appears promising, driven by ongoing advancements in gene therapy and increased government funding. As the healthcare landscape evolves, the integration of telemedicine and digital health solutions is expected to enhance patient access to specialized care. Furthermore, collaborations with international research institutions will likely accelerate the development of innovative therapies, ultimately improving patient outcomes and quality of life for those affected by SMA in Bahrain.
Market Opportunities
- Expansion of Telemedicine Services:The growth of telemedicine services presents a significant opportunity to improve access to SMA care in Bahrain. With an estimated 70% of the population using smartphones, telehealth can facilitate remote consultations and follow-ups, reducing travel burdens for patients and enhancing treatment adherence. This shift could lead to better management of SMA and increased patient satisfaction.
- Development of Patient Support Programs:Establishing comprehensive patient support programs can significantly enhance the quality of life for SMA patients and their families. These programs, which could include counseling, educational resources, and financial assistance, are essential for navigating the complexities of SMA treatment. With an estimated 250 SMA patients in Bahrain, targeted support initiatives can foster community engagement and improve treatment outcomes.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Treatment Type | SMN-Dependent Products (Spinraza, Evrysdi) Gene Transfer Therapy (Zolgensma) Myostatin Inhibitors (Adjunct Therapies) Supportive Care and Symptomatic Management |
| By Route of Administration | Oral Formulations Injectable/Infusion Gene Therapy (One-Time) |
| By End-User | Hospitals and Tertiary Care Centers Specialized SMA Clinics Home Care Settings Primary Healthcare Centers |
| By Patient Age Group | Pediatric (0-18 years) Adult (18-60 years) Senior (60+ years) |
| By Treatment Setting | Inpatient/Hospital-Based Outpatient Home-Based Care with Monitoring |
| By SMA Type | Type I (Infantile-Onset) Type II (Intermediate) Type III (Late-Onset/Kugelberg-Welander) Type IV (Adult-Onset) |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Bahrain Ministry of Health)
Pharmaceutical Manufacturers
Biotechnology Companies
Healthcare Providers and Hospitals
Patient Advocacy Groups
Health Insurance Companies
Medical Device Manufacturers
Players Mentioned in the Report:
Novartis AG (Zolgensma Manufacturer)
Roche Holding AG (Evrysdi Developer)
Biogen Inc. (Spinraza Developer)
Pfizer Inc.
Scholar Rock Holding Corporation (Apitegromab Developer)
Sarepta Therapeutics, Inc.
PTC Therapeutics, Inc.
Biohaven Pharmaceutical (Talditercept Alfa Developer)
Sanofi S.A.
Amgen Inc.
GlaxoSmithKline PLC
Eli Lilly and Company
Merck & Co., Inc.
Ionis Pharmaceuticals (ION306 Developer)
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of SMA in Bahrain
3.1.2 Advancements in gene therapy and treatment options
3.1.3 Government support and funding for rare diseases
3.1.4 Rising awareness and diagnosis rates
3.2 Market Challenges
3.2.1 High cost of innovative treatments
3.2.2 Limited access to specialized healthcare facilities
3.2.3 Regulatory hurdles for new therapies
3.2.4 Shortage of trained healthcare professionals
3.3 Market Opportunities
3.3.1 Expansion of telemedicine services
3.3.2 Collaborations with international research institutions
3.3.3 Development of patient support programs
3.3.4 Investment in local manufacturing of treatments
3.4 Market Trends
3.4.1 Shift towards personalized medicine
3.4.2 Increased focus on patient-centric care
3.4.3 Growth of digital health solutions
3.4.4 Rising interest in clinical trials for SMA therapies
3.5 Government Regulation
3.5.1 Approval processes for orphan drugs
3.5.2 Pricing regulations for new therapies
3.5.3 Policies supporting rare disease research
3.5.4 Guidelines for clinical trial conduct
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Segmentation
8.1 By Treatment Type
8.1.1 SMN-Dependent Products (Spinraza, Evrysdi)
8.1.2 Gene Transfer Therapy (Zolgensma)
8.1.3 Myostatin Inhibitors (Adjunct Therapies)
8.1.4 Supportive Care and Symptomatic Management
8.2 By Route of Administration
8.2.1 Oral Formulations
8.2.2 Injectable/Infusion
8.2.3 Gene Therapy (One-Time)
8.3 By End-User
8.3.1 Hospitals and Tertiary Care Centers
8.3.2 Specialized SMA Clinics
8.3.3 Home Care Settings
8.3.4 Primary Healthcare Centers
8.4 By Patient Age Group
8.4.1 Pediatric (0-18 years)
8.4.2 Adult (18-60 years)
8.4.3 Senior (60+ years)
8.5 By Treatment Setting
8.5.1 Inpatient/Hospital-Based
8.5.2 Outpatient
8.5.3 Home-Based Care with Monitoring
8.6 By SMA Type
8.6.1 Type I (Infantile-Onset)
8.6.2 Type II (Intermediate)
8.6.3 Type III (Late-Onset/Kugelberg-Welander)
8.6.4 Type IV (Adult-Onset)
9. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Based on Total Revenue)
9.2.3 SMA Product Portfolio Revenue (USD Million)
9.2.4 Year-over-Year Revenue Growth Rate (%)
9.2.5 Market Penetration in Bahrain/GCC Region (%)
9.2.6 Number of SMA-Specific Therapies in Portfolio
9.2.7 Pipeline Products (Late-Stage Clinical Development)
9.2.8 R&D Spending as % of Revenue
9.2.9 Healthcare Partnerships and Distribution Networks in GCC
9.2.10 Regulatory Approvals/Certifications in Bahrain/GFDA
9.2.11 Patient Assistance Program Availability
9.2.12 Average Pricing per Treatment Course (USD)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Novartis AG (Zolgensma Manufacturer)
9.5.2 Roche Holding AG (Evrysdi Developer)
9.5.3 Biogen Inc. (Spinraza Developer)
9.5.4 Pfizer Inc.
9.5.5 Scholar Rock Holding Corporation (Apitegromab Developer)
9.5.6 Sarepta Therapeutics, Inc.
9.5.7 PTC Therapeutics, Inc.
9.5.8 Biohaven Pharmaceutical (Talditercept Alfa Developer)
9.5.9 Sanofi S.A.
9.5.10 Amgen Inc.
9.5.11 GlaxoSmithKline PLC
9.5.12 Eli Lilly and Company
9.5.13 Merck & Co., Inc.
9.5.14 Ionis Pharmaceuticals (ION306 Developer)
10. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation for Rare Diseases
10.1.2 Decision-Making Processes
10.1.3 Collaboration with Healthcare Providers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Healthcare Facilities
10.2.2 Funding for Research and Development
10.3 Pain Point Analysis by End-User Category
10.3.1 Access to Treatment
10.3.2 Affordability of Therapies
10.3.3 Availability of Specialized Care
10.4 User Readiness for Adoption
10.4.1 Awareness of Treatment Options
10.4.2 Willingness to Pay for Advanced Therapies
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Long-Term Health Outcomes
10.5.2 Cost Savings from Early Intervention
11. Bahrain Spinal Muscular Atrophy (SMA) Treatment Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-Ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Activity Timeline
15.2.2 Milestone Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of existing literature on Spinal Muscular Atrophy (SMA) treatment options and market dynamics
- Review of government health reports and statistics from the Bahrain Ministry of Health
- Examination of clinical trial registries and published research articles on SMA therapies
Primary Research
- Interviews with healthcare professionals specializing in neuromuscular disorders
- Surveys conducted with caregivers of SMA patients to understand treatment preferences
- Focus group discussions with patient advocacy groups to gather insights on market needs
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and secondary data sources
- Triangulation of treatment adoption rates with demographic and epidemiological data
- Sanity checks through feedback from a panel of medical experts and market analysts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of the total addressable market based on the prevalence of SMA in Bahrain
- Analysis of healthcare expenditure trends related to SMA treatments
- Incorporation of government healthcare initiatives and funding for rare diseases
Bottom-up Modeling
- Collection of data on treatment costs from local hospitals and clinics
- Estimation of patient population growth based on demographic trends
- Volume x cost analysis for different treatment modalities available in Bahrain
Forecasting & Scenario Analysis
- Multi-factor regression analysis considering advancements in SMA therapies and healthcare policies
- Scenario modeling based on potential changes in treatment accessibility and insurance coverage
- Baseline, optimistic, and pessimistic projections for market growth through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatric Neurologists | 40 | Specialists treating SMA patients |
| Healthcare Administrators | 40 | Decision-makers in hospitals and clinics |
| Pharmaceutical Representatives | 40 | Sales and marketing professionals in SMA treatment |
| Patient Caregivers | 40 | Individuals caring for SMA patients |
| Health Policy Makers | 40 | Government officials involved in healthcare policy |
Frequently Asked Questions
What is the current value of the Bahrain Spinal Muscular Atrophy (SMA) Treatment Market?
The Bahrain Spinal Muscular Atrophy (SMA) Treatment Market is valued at approximately USD 185 million, reflecting significant growth driven by advancements in genetic therapies and increased awareness of SMA in the region.