Region:Middle East
Author(s):Geetanshi
Product Code:KRAC3159
Pages:86
Published On:October 2025
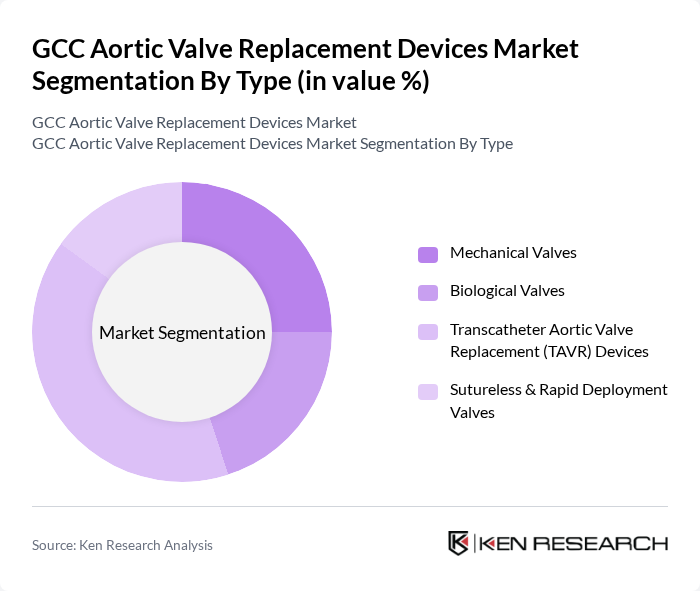
By Type:The market is segmented into various types of aortic valve replacement devices, including Mechanical Valves, Biological Valves, Transcatheter Aortic Valve Replacement (TAVR) Devices, and Sutureless & Rapid Deployment Valves. Among these,TAVR devicesare gaining significant traction due to their minimally invasive nature, which reduces recovery time and hospital stays. The increasing preference for less invasive surgical options, coupled with ongoing technological innovation and favorable reimbursement policies, is driving the demand for TAVR devices, making them a leading sub-segment in the market.
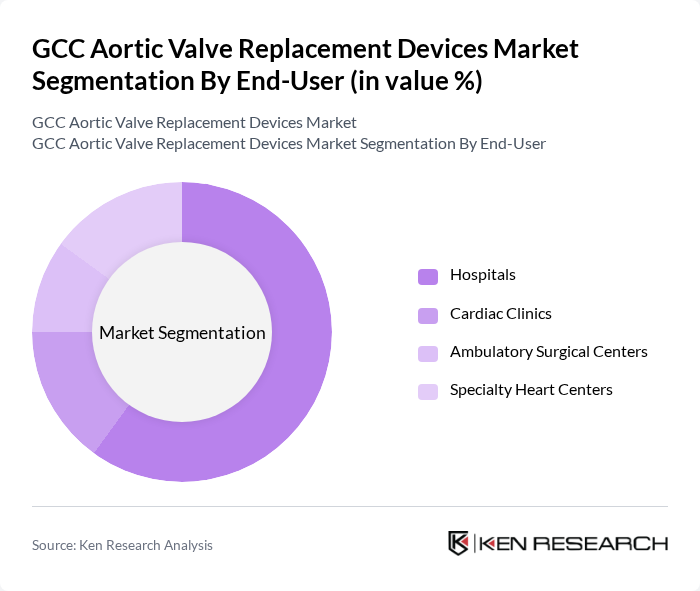
By End-User:The end-user segmentation includes Hospitals, Cardiac Clinics, Ambulatory Surgical Centers, and Specialty Heart Centers.Hospitalsare the dominant end-user segment, primarily due to their capacity to provide comprehensive cardiac care and advanced surgical facilities. The increasing number of cardiac surgeries performed in hospitals, along with the availability of specialized medical teams and integrated care pathways, contributes to their leading position in the market.
The GCC Aortic Valve Replacement Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Edwards Lifesciences Corporation, Medtronic plc, Boston Scientific Corporation, Abbott Laboratories, St. Jude Medical, LLC (now part of Abbott), JenaValve Technology, Inc., LivaNova PLC, CryoLife, Inc. (now Artivion, Inc.), Sorin Group (now part of LivaNova PLC), Terumo Corporation, Micro Interventional Devices, Inc., Neovasc Inc. (now part of Shockwave Medical, Inc.), Tendyne Holdings, Inc. (an Abbott company), Meril Life Sciences Pvt. Ltd., and Braile Biomédica contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC aortic valve replacement devices market appears promising, driven by ongoing technological advancements and increasing healthcare investments. As healthcare infrastructure expands, more facilities will adopt innovative surgical techniques, enhancing patient outcomes. Additionally, the integration of digital health technologies is expected to streamline patient management and improve surgical precision. These trends indicate a robust growth trajectory for the market, with a focus on improving accessibility and affordability of aortic valve replacement procedures.
| Segment | Sub-Segments |
|---|---|
| By Type | Mechanical Valves Biological Valves Transcatheter Aortic Valve Replacement (TAVR) Devices Sutureless & Rapid Deployment Valves |
| By End-User | Hospitals Cardiac Clinics Ambulatory Surgical Centers Specialty Heart Centers |
| By Distribution Channel | Direct Sales Distributors Online Sales |
| By Region | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Patient Demographics | Age Group (Adults, Seniors) Gender (Male, Female) |
| By Surgical Approach | Open-heart Surgery Minimally Invasive Surgery (Including TAVR) |
| By Price Range | Premium Mid-range Economy |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiologists in GCC Hospitals | 100 | Interventional Cardiologists, Electrophysiologists |
| Cardiac Surgeons in GCC Clinics | 60 | Cardiac Surgeons, Thoracic Surgeons |
| Hospital Procurement Managers | 50 | Procurement Officers, Supply Chain Managers |
| Patients Post-Aortic Valve Replacement | 40 | Post-operative Patients, Caregivers |
| Healthcare Policy Makers | 40 | Health Economists, Policy Advisors |
The GCC Aortic Valve Replacement Devices Market is valued at approximately USD 950 million, driven by the rising prevalence of aortic stenosis and advancements in minimally invasive procedures like transcatheter aortic valve replacement (TAVR).