Region:Middle East
Author(s):Rebecca
Product Code:KRAC4627
Pages:91
Published On:October 2025
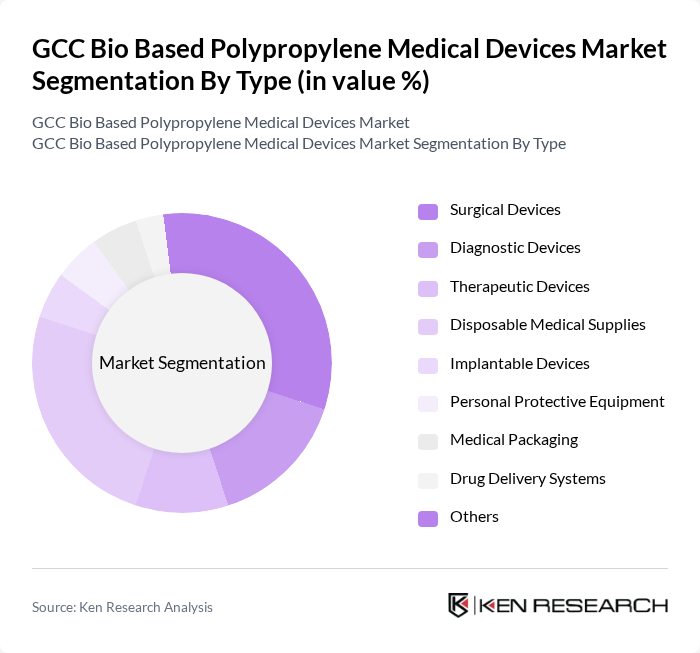
By Type:The market is segmented into various types of medical devices, including surgical devices, diagnostic devices, therapeutic devices, disposable medical supplies, implantable devices, personal protective equipment, medical packaging, drug delivery systems, and others. Among these, surgical devices and disposable medical supplies are particularly prominent due to their high demand in hospitals and clinics. The increasing focus on infection control and patient safety has led to a surge in the use of disposable medical supplies, while surgical devices are essential for various medical procedures. The adoption of bio-based polypropylene is especially notable in single-use and high-volume applications, reflecting the region's emphasis on sustainability and infection control .
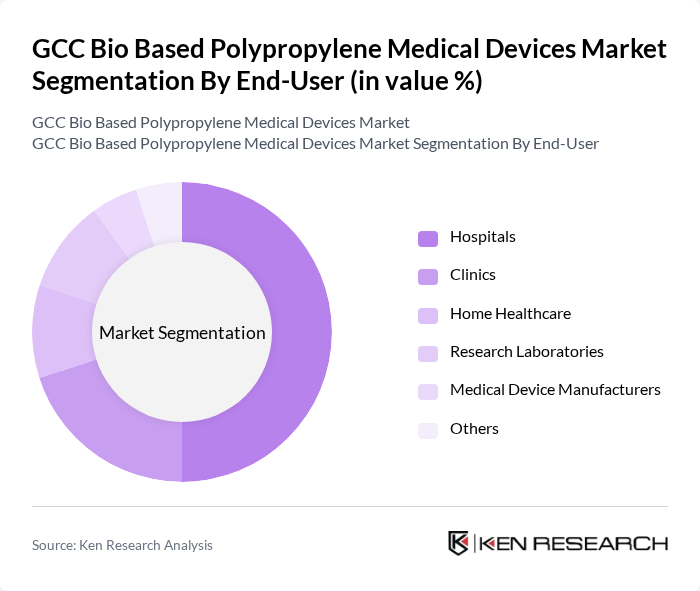
By End-User:The end-user segmentation includes hospitals, clinics, home healthcare, research laboratories, medical device manufacturers, and others. Hospitals are the leading end-users due to their extensive use of medical devices for patient care and treatment. The increasing number of surgical procedures and the growing emphasis on patient safety and hygiene have driven the demand for bio-based medical devices in hospitals, making them a significant contributor to market growth. The adoption of sustainable materials is further supported by institutional procurement policies and regional healthcare sustainability initiatives .
The GCC Bio Based Polypropylene Medical Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as SABIC, BASF SE, Braskem S.A., Borealis AG, Mitsui Chemicals, Inc., Avient Corporation, FKuR Kunststoff GmbH, Danimer Scientific, Reliance Industries Ltd., Arkema S.A., DOW, TotalEnergies Corbion, Green Dot Bioplastics, Solvay S.A., Eastman Chemical Company contribute to innovation, geographic expansion, and service delivery in this space .
The future of the GCC bio-based polypropylene medical devices market appears promising, driven by increasing consumer demand for sustainable healthcare solutions and supportive government policies. As technological advancements continue to lower production costs and improve material performance, the market is likely to see a rise in innovative applications. Additionally, the growing emphasis on environmental sustainability will further propel the adoption of bio-based materials, positioning the GCC as a leader in eco-friendly medical device manufacturing in future.
| Segment | Sub-Segments |
|---|---|
| By Type | Surgical Devices Diagnostic Devices Therapeutic Devices Disposable Medical Supplies Implantable Devices Personal Protective Equipment Medical Packaging Drug Delivery Systems Others |
| By End-User | Hospitals Clinics Home Healthcare Research Laboratories Medical Device Manufacturers Others |
| By Application | Cardiovascular Orthopedic Neurology Wound Care Surgical Sutures Medical Mesh Others |
| By Distribution Channel | Direct Sales Online Retail Medical Supply Distributors Pharmacies Others |
| By Material Type | Bio Based Polypropylene Other Biopolymers (e.g., PLA, PHA) Composites Others |
| By Price Range | Economy Mid-Range Premium |
| By Region | GCC Countries (Saudi Arabia, UAE, Qatar, Kuwait, Oman, Bahrain) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Bio-based Polypropylene Medical Devices | 120 | Product Managers, R&D Directors |
| Healthcare Procurement Strategies | 90 | Procurement Managers, Supply Chain Analysts |
| Regulatory Compliance in Medical Devices | 60 | Regulatory Affairs Specialists, Quality Assurance Managers |
| Market Trends in Sustainable Medical Solutions | 50 | Market Analysts, Sustainability Officers |
| End-user Feedback on Medical Devices | 70 | Healthcare Professionals, Clinical Staff |
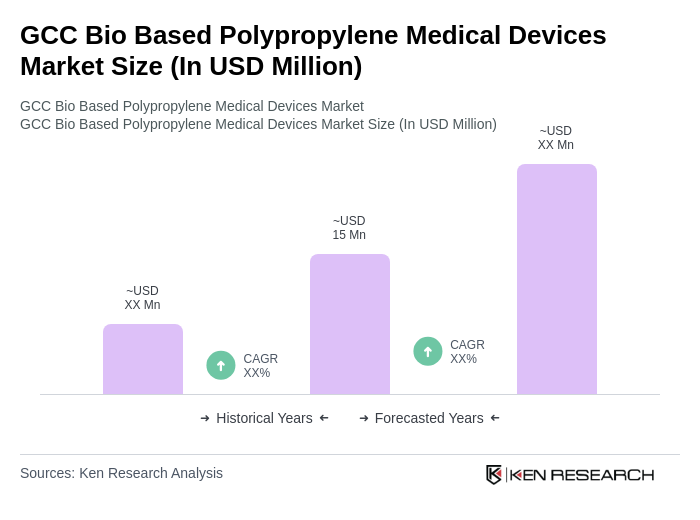
The GCC Bio Based Polypropylene Medical Devices Market is valued at approximately USD 15 million, reflecting a growing trend towards sustainable medical solutions and advancements in biopolymer technology within the region.