Region:Middle East
Author(s):Rebecca
Product Code:KRAD7395
Pages:100
Published On:December 2025
By Test Type:The testing market is segmented into various types, including Extractables and Leachables Testing, Bioburden and Sterility Testing, Endotoxin and Pyrogen Testing, Particulate and Visual Inspection Testing, Physicochemical and Material Characterization Testing, Functional Performance / Integrity Testing, and Others. Each of these subsegments plays a vital role in ensuring the safety and efficacy of biopharmaceutical products, with extractables and leachables, sterility/bioburden, endotoxin, and integrity testing reflecting the core analytical and quality attributes emphasized in global biopharmaceutical consumables testing practice.

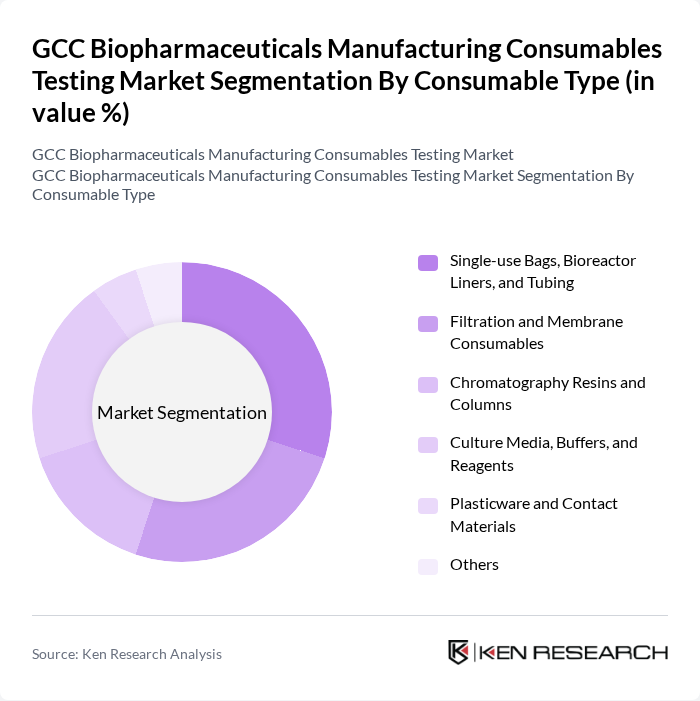
By Consumable Type:The consumable types include Single-use Bags, Bioreactor Liners, and Tubing, Filtration and Membrane Consumables, Chromatography Resins and Columns, Culture Media, Buffers, and Reagents, Plasticware and Contact Materials, and Others. These consumables are essential for the biopharmaceutical manufacturing process, ensuring efficiency, flexibility of single-use bioprocessing, contamination control, and compliance with regulatory standards, in line with global trends where media bags/containers, filtration systems, and chromatography materials are among the most critical consumable categories in bioprocessing.
The GCC Biopharmaceuticals Manufacturing Consumables Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Merck KGaA (MilliporeSigma), Danaher Corporation (Cytiva, Pall Corporation), Sartorius AG, Charles River Laboratories International, Inc., SGS SA, Eurofins Scientific SE, WuXi Biologics (Cayman) Inc., Lonza Group Ltd., Bio-Rad Laboratories, Inc., Agilent Technologies, Inc., TÜV SÜD AG, Saudi Food & Drug Authority (SFDA) – National Laboratories, G42 Healthcare (M42 Group), United Arab Emirates, Qatar Biobank / Qatar Biomedical Research Institute (QBRI) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC biopharmaceuticals manufacturing consumables testing market appears promising, driven by ongoing technological advancements and increased healthcare investments. As the region continues to prioritize biopharmaceutical innovation, the integration of automation and AI in testing processes will enhance efficiency and accuracy. Furthermore, the expansion of manufacturing facilities will create a robust ecosystem, fostering collaboration between industry players and research institutions, ultimately leading to improved patient outcomes and market growth.
| Segment | Sub-Segments |
|---|---|
| By Test Type | Extractables and Leachables Testing Bioburden and Sterility Testing Endotoxin and Pyrogen Testing Particulate and Visual Inspection Testing Physicochemical and Material Characterization Testing Functional Performance / Integrity Testing (e.g., filter integrity, container closure) Others |
| By Consumable Type | Single-use Bags, Bioreactor Liners, and Tubing Filtration and Membrane Consumables Chromatography Resins and Columns Culture Media, Buffers, and Reagents Plasticware and Contact Materials (vials, stoppers, connectors) Others |
| By Stage of Bioprocess | Upstream Processing (cell culture, media preparation) Downstream Processing (purification, formulation) Fill–Finish and Packaging Others |
| By Application | Quality Control and Release Testing In-process Monitoring Raw Material and Supplier Qualification Validation and Qualification Studies Others |
| By End-User | Biopharmaceutical Manufacturers Contract Development and Manufacturing Organizations (CDMOs/CMOs) Contract Research and Testing Organizations (CROs/CTOs) Academic and Research Institutes Others |
| By Geography | Saudi Arabia United Arab Emirates Qatar Kuwait Oman Bahrain |
| By Compliance and Regulatory Category | GMP-certified Testing Services GLP / Research-use-only Testing Regulatory Submission-support Testing (e.g., for EMA/FDA/SFDA) Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Biopharmaceutical Manufacturing Facilities | 120 | Quality Assurance Managers, Production Supervisors |
| Consumables Suppliers | 80 | Sales Directors, Product Managers |
| Regulatory Compliance Experts | 60 | Regulatory Affairs Managers, Compliance Officers |
| Research and Development Departments | 70 | R&D Managers, Laboratory Technicians |
| Quality Control Laboratories | 90 | Laboratory Managers, Quality Control Analysts |
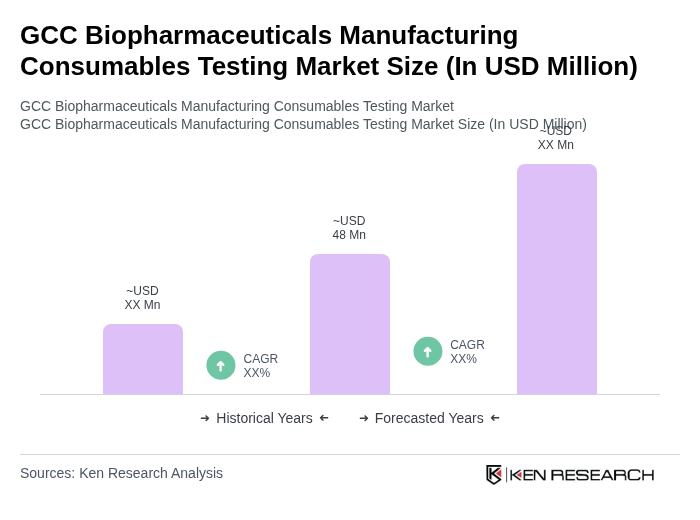
The GCC Biopharmaceuticals Manufacturing Consumables Testing Market is valued at approximately USD 48 million, based on historical analysis and its share of the global biopharmaceuticals manufacturing consumables testing market, which is estimated to be around USD 710720 million.