GCC Bispecific Antibodies Market Overview
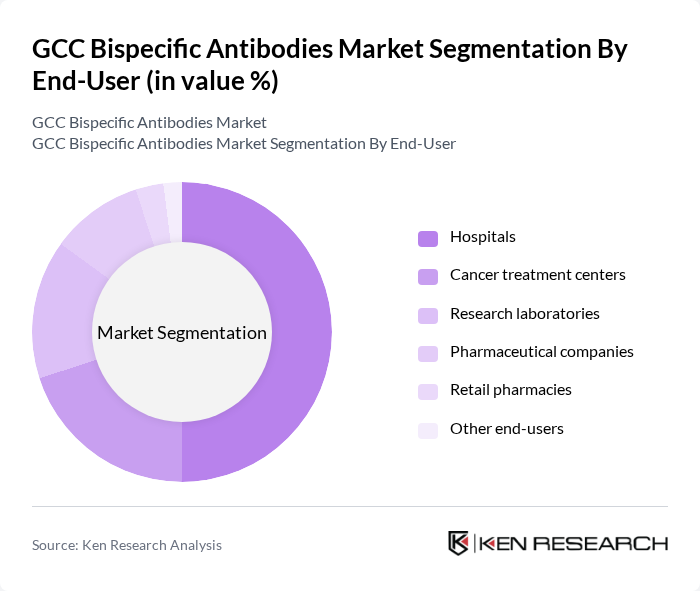
- The GCC Bispecific Antibodies Market is valued at USD 230 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cancer and autoimmune diseases, alongside advancements in biotechnology that enhance the efficacy of bispecific antibodies. The rising demand for targeted therapies and personalized medicine has further propelled market expansion, as healthcare providers seek innovative solutions to improve patient outcomes. Recent trends include the introduction of next-generation bispecific formats, accelerated clinical trial activity, and the integration of artificial intelligence in antibody design, which are further catalyzing market growth in the region .
- Key players in this market include Saudi Arabia and the United Arab Emirates, which dominate due to their robust healthcare infrastructure and significant investments in research and development. The presence of leading pharmaceutical companies and research institutions in these countries fosters innovation and accelerates the development of bispecific antibodies, making them pivotal players in the GCC region. Strategic partnerships with multinational biopharma companies and the establishment of regional clinical trial hubs have further strengthened their market leadership .
- In 2023, the Saudi Food and Drug Authority (SFDA) implemented the “Guideline on the Regulatory Requirements for Registration of Biological Products (including Bispecific Antibodies), 2023,” issued by the Saudi Food and Drug Authority. This regulation mandates that all bispecific antibody products undergo a centralized review process, including submission of comprehensive clinical and quality data, local Good Manufacturing Practice (GMP) certification, and post-marketing surveillance. The guideline aims to expedite clinical trial approvals and regulatory reviews, thereby enhancing patient access to innovative therapies while ensuring safety and efficacy in the GCC region.
GCC Bispecific Antibodies Market Segmentation
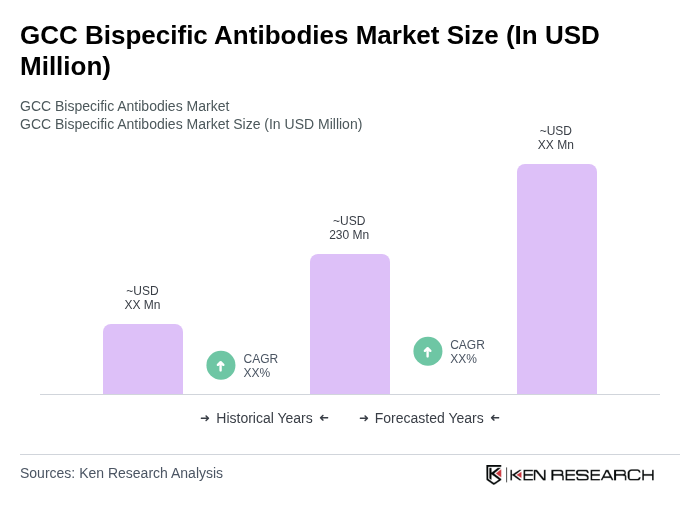
By Type:The market is segmented into various types of bispecific antibodies, including IgG-based, Non-IgG-based, Fragment-based, Full-length, and Other types. Among these, IgG-based bispecific antibodies are leading the market due to their established efficacy and safety profiles, making them the preferred choice for oncological applications. The increasing focus on targeted therapies and the clinical success of IgG-based formats in hematological malignancies and solid tumors have further solidified their dominance in the market .
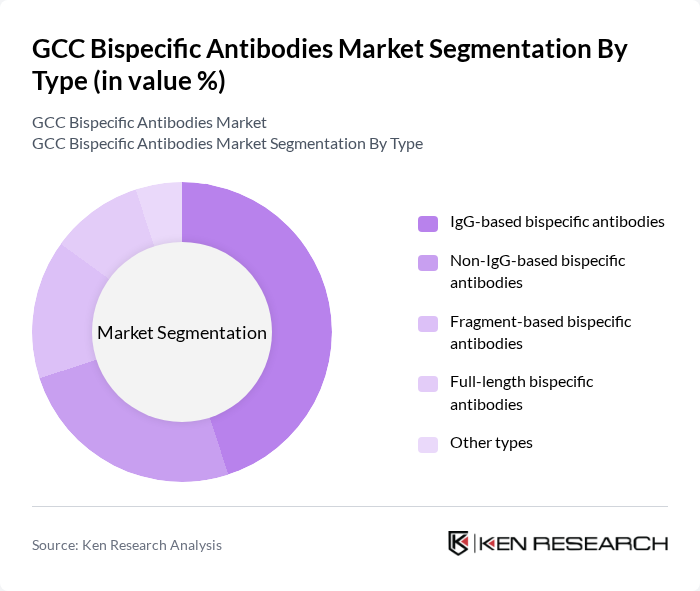
By End-User:The end-user segmentation includes Hospitals, Cancer treatment centers, Research laboratories, Pharmaceutical companies, Retail pharmacies, and Other end-users. Hospitals are the leading end-users, driven by the increasing number of cancer patients requiring advanced treatment options. The growing adoption of bispecific antibodies in clinical settings is enhancing treatment efficacy and patient outcomes, thereby solidifying their position in the healthcare landscape. Cancer treatment centers and research laboratories are also significant contributors due to their focus on clinical trials and translational research .
GCC Bispecific Antibodies Market Competitive Landscape
The GCC Bispecific Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Genentech, a member of the Roche Group, AbbVie Inc., Regeneron Pharmaceuticals, Inc., Merck & Co., Inc., Pfizer Inc., Novartis AG, Sanofi S.A., Takeda Pharmaceutical Company Limited, GSK plc, Eli Lilly and Company, Bayer AG, Bristol-Myers Squibb Company, Johnson & Johnson, AstraZeneca PLC, WuXi Biologics, Innovent Biologics, MacroGenics, Inc., Janssen Biotech, Inc. (Johnson & Johnson), Zymeworks Inc. contribute to innovation, geographic expansion, and service delivery in this space.
GCC Bispecific Antibodies Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cancer and Autoimmune Diseases:The GCC region has witnessed a significant rise in cancer cases, with the World Health Organization reporting over 250,000 new cancer cases annually. Additionally, autoimmune diseases are affecting approximately 7% of the population, leading to a growing demand for innovative treatment options. This increasing prevalence drives the need for bispecific antibodies, which can target multiple pathways, enhancing therapeutic efficacy and improving patient outcomes.
- Advancements in Biotechnology and Drug Development:The GCC has invested heavily in biotechnology, with funding exceeding $1.5 billion for research and development. This investment has led to breakthroughs in bispecific antibody technologies, enabling the development of more effective therapies. The establishment of biotechnology hubs in countries like Saudi Arabia and the UAE fosters innovation, attracting global partnerships and enhancing the region's capabilities in drug development.
- Rising Demand for Targeted Therapies:The global shift towards personalized medicine is reflected in the GCC, where targeted therapies are increasingly preferred. In future, the market for targeted therapies in the region was valued at approximately $4 billion, with bispecific antibodies gaining traction due to their ability to simultaneously engage multiple targets. This trend is supported by healthcare providers' growing emphasis on precision medicine, which aligns with patient needs and improves treatment outcomes.
Market Challenges
- High Development Costs and Complex Manufacturing Processes:The development of bispecific antibodies involves intricate manufacturing processes, leading to costs that can exceed $600 million per product. This financial burden poses a significant challenge for companies, particularly smaller firms, limiting their ability to invest in research and development. The complexity of these processes also results in longer timelines for product development, further straining resources and delaying market entry.
- Regulatory Hurdles and Lengthy Approval Timelines:The regulatory landscape for bispecific antibodies is complex, with approval processes often taking over 12 years. The GCC's regulatory bodies require extensive clinical trial data, which can delay product launches. In future, only 25% of submitted bispecific antibody applications received timely approvals, highlighting the challenges companies face in navigating regulatory requirements while ensuring compliance with safety and efficacy standards.
GCC Bispecific Antibodies Market Future Outlook
The future of the bispecific antibodies market in the GCC appears promising, driven by ongoing advancements in biotechnology and a growing focus on personalized medicine. As healthcare systems increasingly adopt targeted therapies, the demand for innovative treatment options is expected to rise. Furthermore, collaborations between pharmaceutical companies and research institutions are likely to accelerate the development of novel bispecific formats, enhancing therapeutic options for patients and addressing unmet medical needs in the region.
Market Opportunities
- Expansion into Emerging Markets:The GCC region presents significant opportunities for bispecific antibodies, particularly in emerging markets where healthcare infrastructure is improving. With a projected increase in healthcare spending to $120 billion in future, companies can capitalize on this growth by introducing innovative therapies tailored to local needs, thereby expanding their market reach and enhancing patient access to advanced treatments.
- Collaborations and Partnerships with Research Institutions:Strategic partnerships with research institutions can facilitate the development of cutting-edge bispecific antibodies. In future, over 50 collaborations were reported in the GCC, focusing on innovative therapies. These partnerships can leverage shared expertise and resources, accelerating the research process and enhancing the potential for successful product development, ultimately benefiting both parties involved.