Region:Middle East
Author(s):Dev
Product Code:KRAC4105
Pages:98
Published On:October 2025
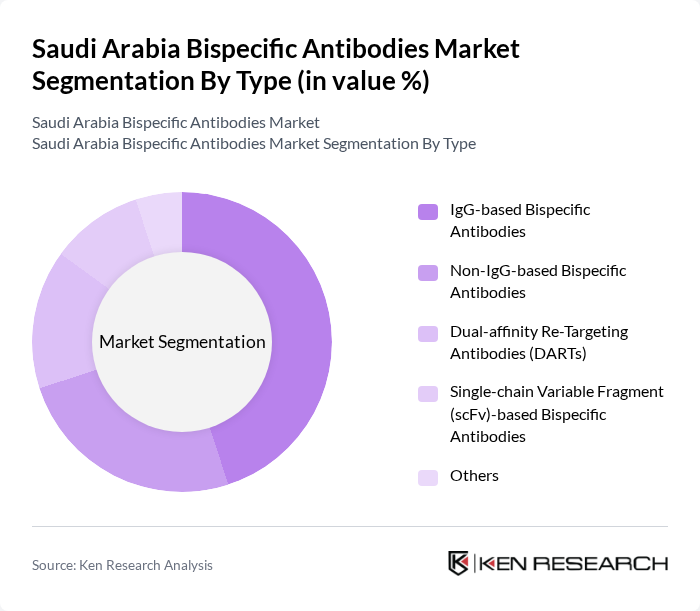
By Type:The market is segmented into various types of bispecific antibodies, including IgG-based bispecific antibodies, Non-IgG-based bispecific antibodies, Dual-affinity Re-Targeting Antibodies (DARTs), Single-chain Variable Fragment (scFv)-based bispecific antibodies, and others. Among these, IgG-based bispecific antibodies are leading the market due to their established efficacy and safety profiles in clinical applications .
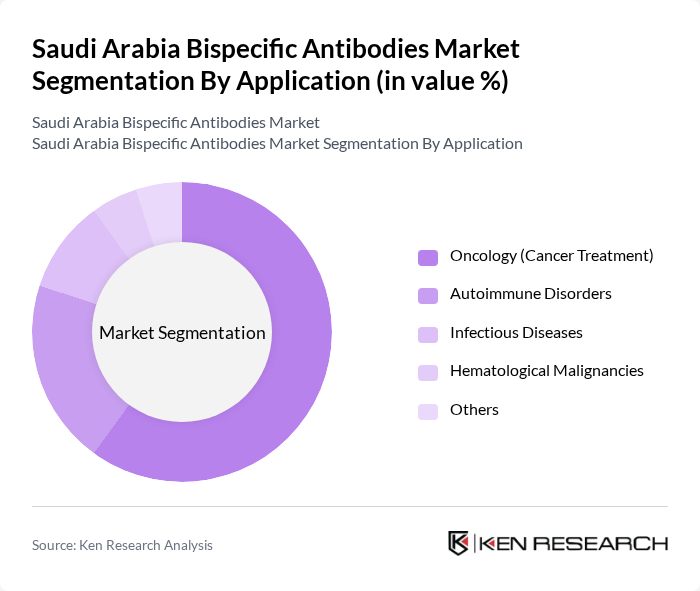
By Application:The applications of bispecific antibodies include oncology (cancer treatment), autoimmune disorders, infectious diseases, hematological malignancies, and others. The oncology segment is the most significant, driven by the increasing incidence of cancer and the demand for targeted therapies that improve patient outcomes .
The Saudi Arabia Bispecific Antibodies Market is characterized by a dynamic mix of regional and international players. Leading participants such as Amgen Inc., Genentech, Inc. (Roche Group), AbbVie Inc., Regeneron Pharmaceuticals, Inc., Bayer AG, Merck & Co., Inc., Pfizer Inc., Sanofi S.A., Takeda Pharmaceutical Company Limited, Novartis AG, GSK plc, Eli Lilly and Company, Bristol-Myers Squibb Company, AstraZeneca PLC, Johnson & Johnson, Immunocore Holdings plc, Janssen Pharmaceuticals (Johnson & Johnson), Akeso, Inc., Taisho Pharmaceutical Co., Ltd., Saudi Pharmaceutical Industries & Medical Appliances Corporation (SPIMACO), Tabuk Pharmaceuticals Manufacturing Co., Jamjoom Pharma, Hikma Pharmaceuticals plc contribute to innovation, geographic expansion, and service delivery in this space.
The future of the bispecific antibodies market in Saudi Arabia appears promising, driven by ongoing advancements in biotechnology and a supportive regulatory environment. As healthcare infrastructure continues to expand, the accessibility of innovative therapies will improve. Additionally, the increasing focus on personalized medicine is expected to drive demand for bispecific antibodies, as they offer tailored treatment options. Collaborations between pharmaceutical companies and research institutions will further enhance the development of these therapies, positioning Saudi Arabia as a key player in the biopharmaceutical landscape.
| Segment | Sub-Segments |
|---|---|
| By Type | IgG-based Bispecific Antibodies Non-IgG-based Bispecific Antibodies Dual-affinity Re-Targeting Antibodies (DARTs) Single-chain Variable Fragment (scFv)-based Bispecific Antibodies Others |
| By Application | Oncology (Cancer Treatment) Autoimmune Disorders Infectious Diseases Hematological Malignancies Others |
| By End-User | Hospitals Research Institutions Pharmaceutical & Biotechnology Companies Diagnostic Laboratories Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Hospital Pharmacies Others |
| By Region | Central Region Eastern Region Western Region Southern Region |
| By Pricing Strategy | Premium Pricing Competitive Pricing Value-based Pricing Others |
| By Research Phase | Preclinical Clinical Trials Marketed Products Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Treatment Centers | 60 | Oncologists, Clinical Researchers |
| Pharmaceutical Manufacturers | 50 | Product Managers, R&D Directors |
| Healthcare Policy Makers | 40 | Health Economists, Regulatory Affairs Specialists |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
| Clinical Trial Coordinators | 40 | Clinical Research Associates, Trial Managers |
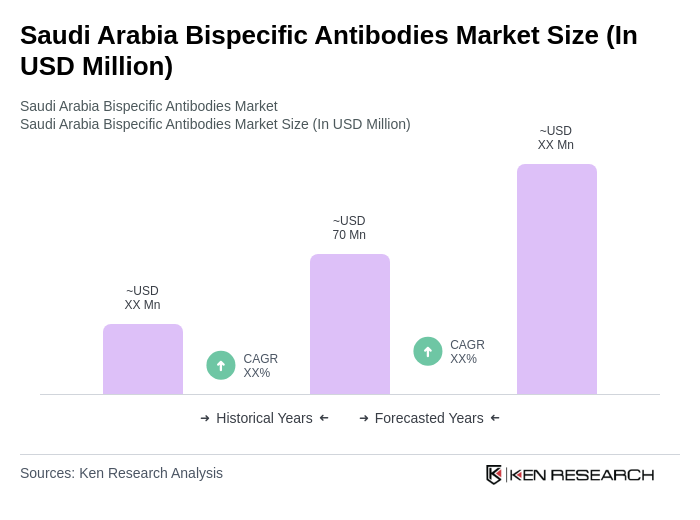
The Saudi Arabia Bispecific Antibodies Market is valued at approximately USD 70 million, reflecting significant growth driven by advancements in biotechnology and the increasing prevalence of chronic diseases, particularly cancer.