Region:Middle East
Author(s):Geetanshi
Product Code:KRAC8302
Pages:88
Published On:November 2025
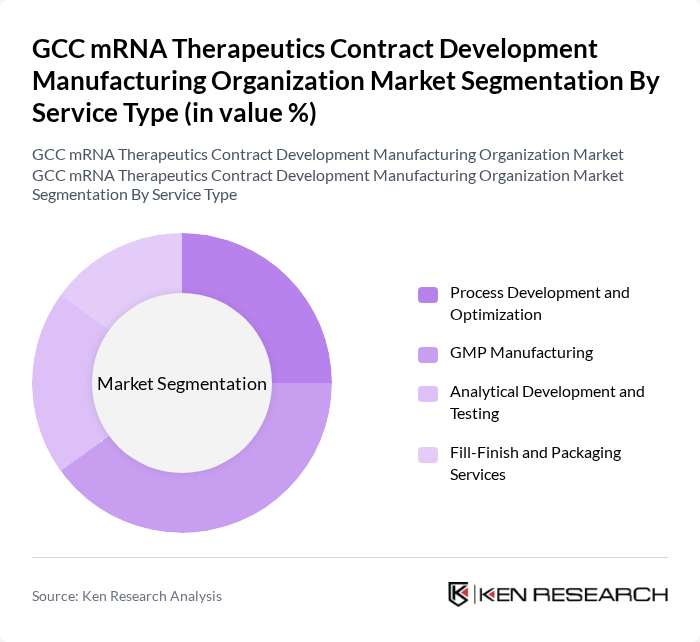
By Service Type:The service type segmentation includes essential services supporting the development and manufacturing of mRNA therapeutics: Process Development and Optimization, GMP Manufacturing, Analytical Development and Testing, and Fill-Finish and Packaging Services. GMP Manufacturing remains the leading subsegment, reflecting the growing demand for compliant production processes that meet stringent regulatory standards. The focus on GMP is driven by the need for high-quality, scalable, and safe manufacturing to support clinical and commercial supply .
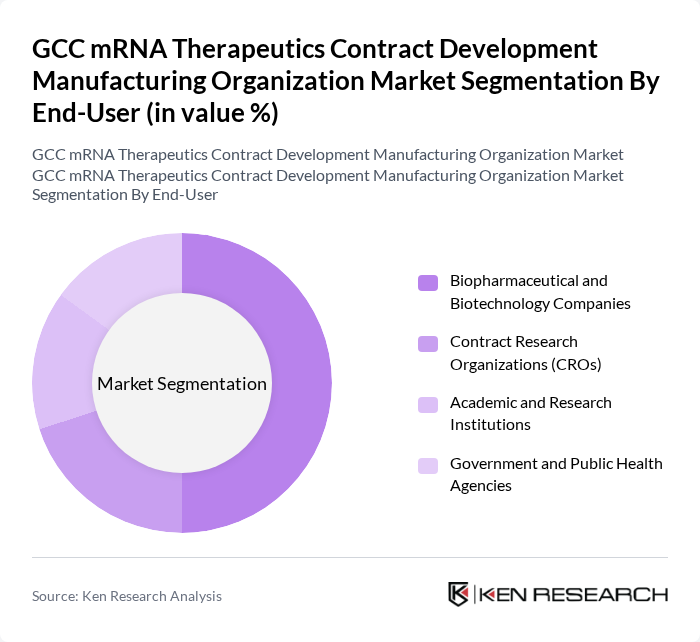
By End-User:The end-user segmentation includes Biopharmaceutical and Biotechnology Companies, Contract Research Organizations (CROs), Academic and Research Institutions, and Government and Public Health Agencies. Biopharmaceutical and Biotechnology Companies dominate this segment, driven by their pursuit of innovative therapies and significant investments in mRNA technology. These companies account for the majority of CDMO service utilization, followed by CROs and academic institutions, which are increasingly partnering with CDMOs for research and clinical development .
The GCC mRNA Therapeutics Contract Development Manufacturing Organization Market is characterized by a dynamic mix of regional and international players. Leading participants such as Lonza Group AG, Wacker Biotech GmbH, Aldevron (Danaher Company), Thermo Fisher Scientific (Patheon), Samsung Biologics, CordenPharma, Catalent Inc., BioNTech Manufacturing Services, Moderna Manufacturing Partnerships, Emergent BioSolutions, Evonik Industries AG, Rentschler Biopharma, WuXi Biologics, AGC Biologics, MilliporeSigma (Merck KGaA CDMO Division) contribute to innovation, geographic expansion, and service delivery in this space.
The future of the GCC mRNA therapeutics market appears promising, driven by ongoing technological advancements and increasing collaboration between public and private sectors. As governments prioritize healthcare innovation, the region is likely to see enhanced funding and support for mRNA research. Additionally, the growing focus on decentralized manufacturing models will facilitate quicker responses to emerging health threats, positioning the GCC as a key player in the global biopharmaceutical landscape.
| Segment | Sub-Segments |
|---|---|
| By Service Type | Process Development and Optimization GMP Manufacturing Analytical Development and Testing Fill-Finish and Packaging Services |
| By End-User | Biopharmaceutical and Biotechnology Companies Contract Research Organizations (CROs) Academic and Research Institutions Government and Public Health Agencies |
| By Therapeutic Application | Infectious Diseases and Vaccines Oncology and Cancer Immunotherapy Genetic Disorders and Rare Diseases Cardiovascular and Metabolic Diseases |
| By Manufacturing Technology | In Vitro Transcription (IVT) Lipid Nanoparticle (LNP) Formulation and Encapsulation Plasmid DNA Production Aseptic Fill-Finish and Cold Chain Management |
| By Region | Saudi Arabia United Arab Emirates Qatar Kuwait Bahrain Oman |
| By Scale of Production | Preclinical and Early-Stage Development Clinical Trial Manufacturing Commercial-Scale Production |
| By Regulatory Compliance Level | GMP-Certified Facilities ISO 13485 Certified Services FDA and EMA Compliant Operations |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| mRNA Therapeutics Development | 60 | R&D Directors, Biotech Executives |
| Contract Manufacturing Services | 50 | Operations Managers, Quality Assurance Managers |
| Regulatory Compliance in mRNA | 40 | Regulatory Affairs Specialists, Compliance Officers |
| Market Access Strategies for mRNA | 45 | Market Access Managers, Health Economists |
| Investment Trends in mRNA Technologies | 40 | Venture Capitalists, Investment Analysts |
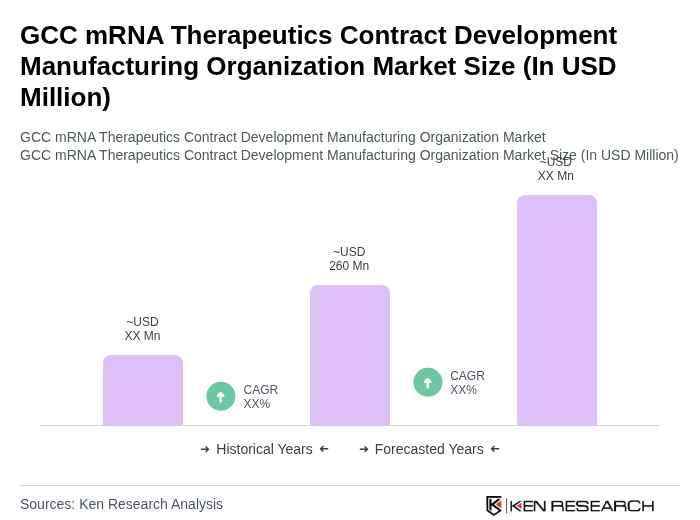
The GCC mRNA Therapeutics Contract Development Manufacturing Organization market is valued at approximately USD 260 million, reflecting significant growth driven by increased demand for mRNA-based therapeutics, particularly following the COVID-19 pandemic.