Region:Middle East
Author(s):Dev
Product Code:KRAA3945
Pages:88
Published On:January 2026
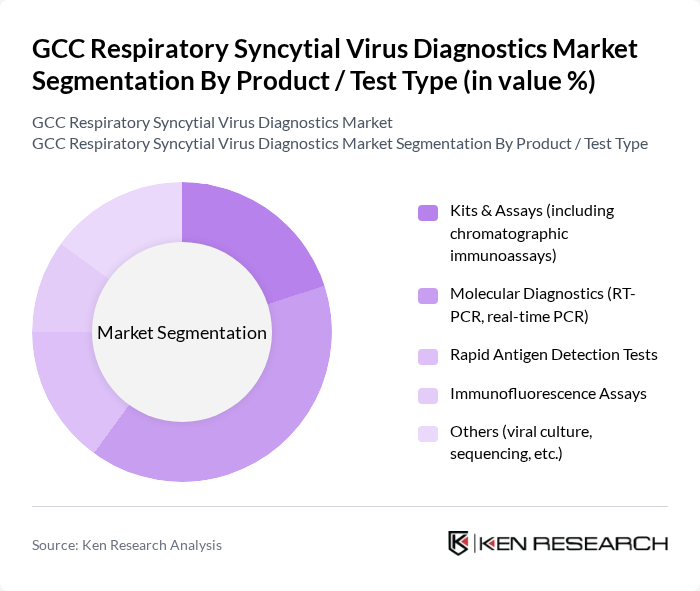
By Product / Test Type:The market is segmented into various product types, including Kits & Assays, Molecular Diagnostics, Rapid Antigen Detection Tests, Immunofluorescence Assays, and Others. Molecular Diagnostics, particularly RT-PCR and real-time PCR, are leading the market due to their high sensitivity and specificity in detecting RSV, in line with global trends where molecular tests account for more than half of RSV diagnostics revenue. The increasing adoption of these advanced diagnostic methods in hospitals and laboratories, often through multiplex respiratory panels that simultaneously detect RSV, influenza, and other viruses, is driving their dominance as they provide rapid, reliable results essential for effective patient management and infection control.
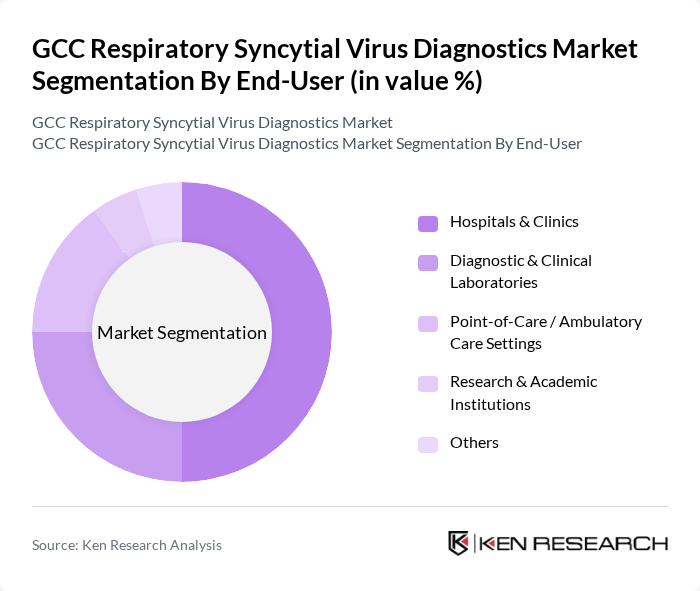
By End-User:The end-user segmentation includes Hospitals & Clinics, Diagnostic & Clinical Laboratories, Point-of-Care / Ambulatory Care Settings, Research & Academic Institutions, and Others. Hospitals & Clinics are the leading end-users, consistent with global patterns where institutional settings account for the majority of RSV testing, driven by the increasing number of RSV cases requiring clinical evaluation and the need for immediate diagnosis and treatment. The growing trend of point-of-care testing, including rapid antigen and cartridge-based molecular platforms used in emergency departments and outpatient clinics, is also contributing to the market, as it allows for rapid diagnosis in ambulatory settings, supports early triage decisions, and enhances patient care.
The GCC Respiratory Syncytial Virus Diagnostics Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, F. Hoffmann-La Roche AG (Roche Diagnostics), Thermo Fisher Scientific Inc., bioMérieux SA, Cepheid (Danaher Corporation), Hologic, Inc., QuidelOrtho Corporation, Becton, Dickinson and Company, Siemens Healthineers AG, GenMark Diagnostics, Inc. (Roche), PerkinElmer, Inc. (Revvity), Agilent Technologies, Inc., Luminex Corporation (DiaSorin S.p.A.), Meridian Bioscience, Inc., DiaSorin S.p.A. contribute to innovation, geographic expansion, and service delivery in this space, particularly through rapid antigen tests, molecular RSV assays, and integrated respiratory panels that are increasingly deployed across hospitals and laboratories in the GCC.
The future of the GCC Respiratory Syncytial Virus diagnostics market appears promising, driven by technological advancements and increased healthcare investments. The integration of artificial intelligence in diagnostic processes is expected to enhance accuracy and efficiency, while the expansion of telemedicine services will improve access to care. Furthermore, the growing emphasis on preventive healthcare measures will likely lead to increased demand for rapid diagnostic tests, positioning the market for significant growth in future.
| Segment | Sub-Segments |
|---|---|
| By Product / Test Type | Kits & Assays (including chromatographic immunoassays) Molecular Diagnostics (RT-PCR, real-time PCR) Rapid Antigen Detection Tests Immunofluorescence Assays Others (viral culture, sequencing, etc.) |
| By End-User | Hospitals & Clinics Diagnostic & Clinical Laboratories Point-of-Care / Ambulatory Care Settings Research & Academic Institutions Others |
| By Sample Type | Nasopharyngeal Swabs / Aspirates Oropharyngeal (Throat) Swabs Nasal Swabs Blood and Serum Samples Others (e.g., bronchoalveolar lavage) |
| By Setting / Testing Location | Centralized Laboratory Testing Point-of-Care Testing Home / At-Home Testing Others |
| By Geography | Saudi Arabia UAE Qatar Kuwait Oman Bahrain |
| By Patient Age Group | Neonates and Infants (0–2 years) Children (3–17 years) Adults (18–64 years) Geriatric (65+ years) Immunocompromised Patients (all ages) |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pediatric Hospitals and Clinics | 120 | Pediatricians, Hospital Administrators |
| Diagnostic Laboratories | 90 | Laboratory Managers, Technicians |
| Public Health Agencies | 60 | Public Health Officials, Epidemiologists |
| Healthcare Policy Makers | 50 | Health Policy Analysts, Government Officials |
| Pharmaceutical Companies | 70 | Product Managers, Market Access Specialists |
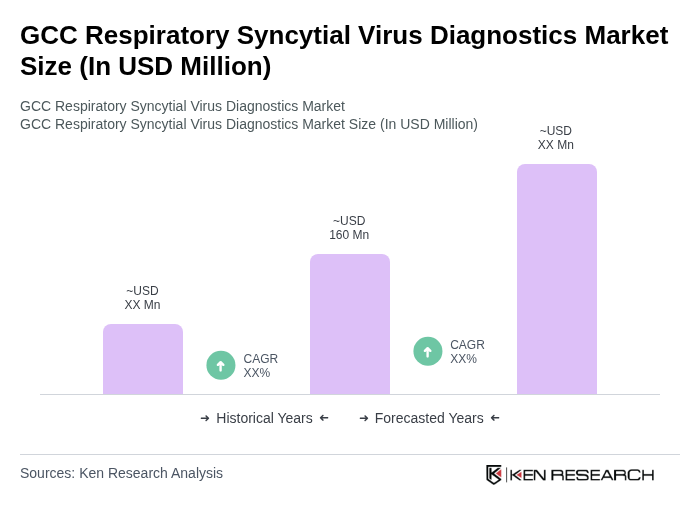
The GCC Respiratory Syncytial Virus Diagnostics Market is valued at approximately USD 160 million, reflecting its significant share within the global RSV diagnostics market, driven by increasing respiratory infections and advancements in diagnostic technologies.