About the Report
Base Year 2024Listen to the audio summary
Germany Biotech Vaccines & Therapeutics Market Overview
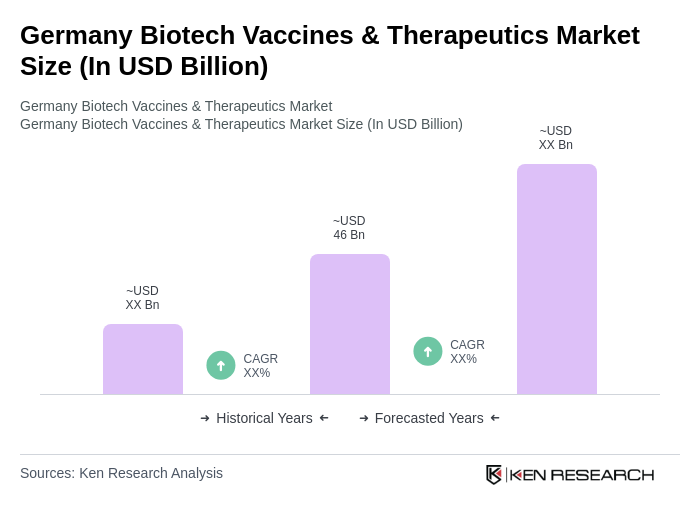
- The Germany Biotech Vaccines & Therapeutics Market is valued at USD 46 billion, based on a five-year historical analysis. This growth is primarily driven by increasing investments in research and development, a rising prevalence of chronic and infectious diseases, and a growing demand for personalized medicine. The market has seen significant advancements in vaccine technology, gene therapies, and biopharmaceutical solutions, which have contributed to its expansion .
- Key cities such asBerlin, Munich, and Frankfurtdominate the market due to their robust healthcare infrastructure, presence of leading biotech firms, and strong academic institutions. These cities serve as hubs for innovation and collaboration, attracting talent and investment, which further enhances their market position .
- The "Digital Healthcare Act" (Digitale-Versorgung-Gesetz, DVG), issued by the Federal Ministry of Health in 2019, aims to promote digital health solutions and streamline the approval process for innovative therapies and vaccines. This regulation mandates the integration of digital health applications (DiGA) into statutory health insurance, enhances data interoperability, and supports faster patient access to advanced treatments .
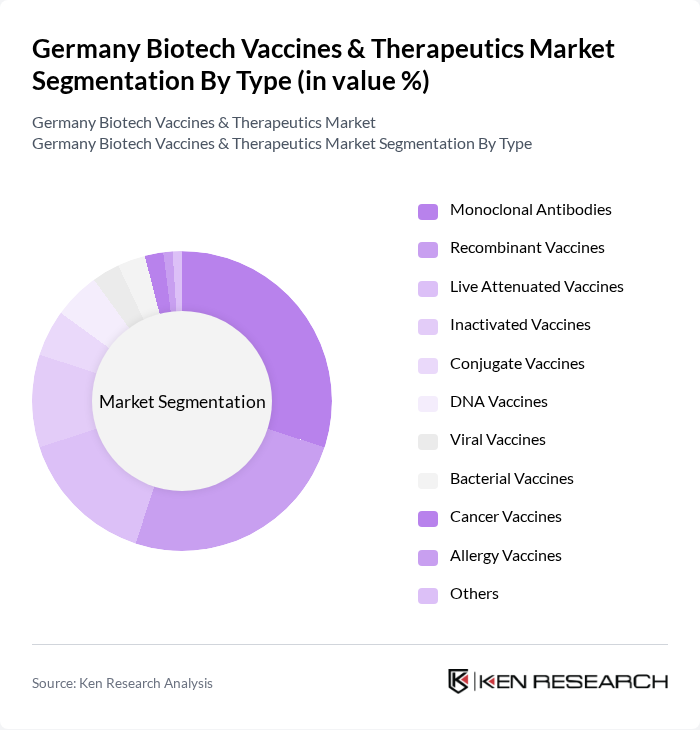
Germany Biotech Vaccines & Therapeutics Market Segmentation
By Type:The market is segmented into various types of vaccines and therapeutics, including monoclonal antibodies, recombinant vaccines, live attenuated vaccines, inactivated vaccines, conjugate vaccines, DNA vaccines, viral vaccines, bacterial vaccines, cancer vaccines, allergy vaccines, and others. Among these,monoclonal antibodies and recombinant vaccinesare leading the market due to their effectiveness and widespread application in treating cancer, autoimmune, and infectious diseases .
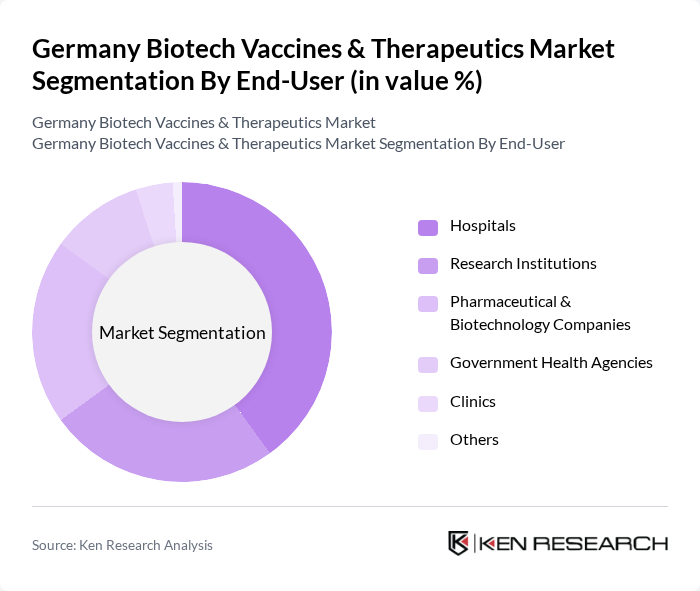
By End-User:The end-user segmentation includes hospitals, research institutions, pharmaceutical and biotechnology companies, government health agencies, clinics, and others.Hospitals and pharmaceutical companiesare the primary consumers of biotech vaccines and therapeutics, driven by the increasing need for advanced treatment options, the growing patient population, and the integration of innovative therapies into clinical practice .
Germany Biotech Vaccines & Therapeutics Market Competitive Landscape
The Germany Biotech Vaccines & Therapeutics Market is characterized by a dynamic mix of regional and international players. Leading participants such as BioNTech SE, CureVac AG, Merck KGaA, Boehringer Ingelheim GmbH, Bayer AG, Pfizer Deutschland GmbH, Sanofi-Aventis Deutschland GmbH, Novartis Pharma GmbH, Roche Deutschland Holding GmbH, GSK GmbH, AbbVie Deutschland GmbH, Johnson & Johnson GmbH, Bavarian Nordic GmbH, Evotec SE, Biotest AG contribute to innovation, geographic expansion, and service delivery in this space .
Germany Biotech Vaccines & Therapeutics Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Infectious Diseases:The rise in infectious diseases, such as COVID-19 and influenza, has significantly impacted the demand for biotech vaccines in Germany. In future, the incidence of infectious diseases is projected to reach approximately 1.6 million cases, according to the Robert Koch Institute. This surge necessitates innovative vaccine solutions, driving investments in biotech research and development. The urgency for effective vaccines is further underscored by the estimated healthcare costs of €32 billion associated with infectious diseases in Germany.
- Rising Investment in R&D:Germany's biotech sector is witnessing a substantial increase in research and development funding, with investments reaching €3.8 billion in future. This funding is crucial for developing advanced vaccine technologies and therapeutics. The German government, alongside private investors, is prioritizing biotech innovations, which is evident from the 16% year-on-year growth in R&D spending. This financial commitment fosters an environment conducive to breakthroughs in vaccine efficacy and safety, enhancing the overall market landscape.
- Government Support for Biotech Innovations:The German government has implemented various initiatives to bolster the biotech sector, including the Biotech Strategy 2025, which allocates €1.2 billion for biotech research. This support is vital for fostering innovation and streamlining the development process for vaccines and therapeutics. Additionally, the establishment of public-private partnerships has led to a 22% increase in collaborative projects, enhancing the capacity for rapid vaccine development and deployment in response to health crises.
Market Challenges
- High Development Costs:The development of biotech vaccines is associated with significant financial burdens, often exceeding €1.2 billion per product. These costs encompass extensive clinical trials, regulatory compliance, and manufacturing processes. In future, the average cost of bringing a new vaccine to market is projected to remain high, posing a barrier for smaller biotech firms. This financial strain can limit innovation and slow the introduction of new therapies, impacting overall market growth.
- Stringent Regulatory Requirements:The regulatory landscape for biotech vaccines in Germany is complex, with stringent requirements set by the European Medicines Agency (EMA) and the German Medicines Act (AMG). In future, the average time for vaccine approval is estimated at 11-13 years, which can hinder timely market entry. These rigorous regulations, while ensuring safety and efficacy, create challenges for companies seeking to navigate the approval process efficiently, potentially stifling innovation.
Germany Biotech Vaccines & Therapeutics Market Future Outlook
The future of the Germany biotech vaccines and therapeutics market appears promising, driven by technological advancements and increased collaboration between public and private sectors. The shift towards mRNA vaccine technology is expected to revolutionize vaccine development, enhancing speed and efficacy. Additionally, the integration of artificial intelligence in drug discovery is anticipated to streamline processes, reduce costs, and improve outcomes. As preventive healthcare gains traction, the focus on innovative vaccine solutions will likely expand, fostering a robust market environment.
Market Opportunities
- Expansion into Emerging Markets:There is a significant opportunity for German biotech firms to expand into emerging markets, where the demand for vaccines is rapidly increasing. With a projected market growth of €2.5 billion in regions like Southeast Asia and Africa, companies can leverage their expertise to address unmet healthcare needs, enhancing global health outcomes while driving revenue growth.
- Collaborations with Research Institutions:Collaborating with academic and research institutions presents a valuable opportunity for innovation in vaccine development. In future, partnerships are expected to increase by 30%, facilitating knowledge exchange and resource sharing. These collaborations can accelerate the development of next-generation vaccines, positioning companies at the forefront of the biotech industry and enhancing their competitive edge.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Monoclonal Antibodies Recombinant Vaccines Live Attenuated Vaccines Inactivated Vaccines Conjugate Vaccines DNA Vaccines Viral Vaccines Bacterial Vaccines Cancer Vaccines Allergy Vaccines Others |
| By End-User | Hospitals Research Institutions Pharmaceutical & Biotechnology Companies Government Health Agencies Clinics Others |
| By Application | Infectious Diseases Cancer Treatment Autoimmune Disorders Allergic Conditions Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Hospital Pharmacies Others |
| By Region | North Germany South Germany East Germany West Germany Central Germany Others |
| By Price Range | Low Price Mid Price High Price |
| By Regulatory Approval Status | Approved Under Review Not Approved |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Paul-Ehrlich-Institut, Bundesinstitut für Arzneimittel und Medizinprodukte)
Biotech and Pharmaceutical Companies
Healthcare Providers and Hospitals
Clinical Research Organizations
Biotechnology Industry Associations
Public Health Organizations
Insurance Companies and Payers
Players Mentioned in the Report:
BioNTech SE
CureVac AG
Merck KGaA
Boehringer Ingelheim GmbH
Bayer AG
Pfizer Deutschland GmbH
Sanofi-Aventis Deutschland GmbH
Novartis Pharma GmbH
Roche Deutschland Holding GmbH
GSK GmbH
AbbVie Deutschland GmbH
Johnson & Johnson GmbH
Bavarian Nordic GmbH
Evotec SE
Biotest AG
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Germany Biotech Vaccines & Therapeutics Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Germany Biotech Vaccines & Therapeutics Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Germany Biotech Vaccines & Therapeutics Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of infectious diseases
3.1.2 Rising investment in R&D
3.1.3 Government support for biotech innovations
3.1.4 Growing demand for personalized medicine
3.2 Market Challenges
3.2.1 High development costs
3.2.2 Stringent regulatory requirements
3.2.3 Market access barriers
3.2.4 Competition from traditional vaccines
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Collaborations with research institutions
3.3.3 Advancements in vaccine technology
3.3.4 Increased focus on preventive healthcare
3.4 Market Trends
3.4.1 Shift towards mRNA vaccines
3.4.2 Integration of AI in drug development
3.4.3 Rise of biosimilars
3.4.4 Emphasis on sustainability in production
3.5 Government Regulation
3.5.1 EU regulations on vaccine approval
3.5.2 German Medicines Act (AMG)
3.5.3 Biologics Control Act
3.5.4 Data protection regulations (GDPR)
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Germany Biotech Vaccines & Therapeutics Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Germany Biotech Vaccines & Therapeutics Market Segmentation
8.1 By Type
8.1.1 Monoclonal Antibodies
8.1.2 Recombinant Vaccines
8.1.3 Live Attenuated Vaccines
8.1.4 Inactivated Vaccines
8.1.5 Conjugate Vaccines
8.1.6 DNA Vaccines
8.1.7 Viral Vaccines
8.1.8 Bacterial Vaccines
8.1.9 Cancer Vaccines
8.1.10 Allergy Vaccines
8.1.11 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Research Institutions
8.2.3 Pharmaceutical & Biotechnology Companies
8.2.4 Government Health Agencies
8.2.5 Clinics
8.2.6 Others
8.3 By Application
8.3.1 Infectious Diseases
8.3.2 Cancer Treatment
8.3.3 Autoimmune Disorders
8.3.4 Allergic Conditions
8.3.5 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Hospital Pharmacies
8.4.5 Others
8.5 By Region
8.5.1 North Germany
8.5.2 South Germany
8.5.3 East Germany
8.5.4 West Germany
8.5.5 Central Germany
8.5.6 Others
8.6 By Price Range
8.6.1 Low Price
8.6.2 Mid Price
8.6.3 High Price
8.7 By Regulatory Approval Status
8.7.1 Approved
8.7.2 Under Review
8.7.3 Not Approved
9. Germany Biotech Vaccines & Therapeutics Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, Small as per industry convention)
9.2.3 Revenue (Germany Biotech Vaccines & Therapeutics Segment)
9.2.4 Revenue Growth Rate (YoY %)
9.2.5 Market Share (%)
9.2.6 R&D Investment as % of Revenue
9.2.7 Number of Product Approvals (Annual)
9.2.8 Pipeline Strength (Number of Candidates in Clinical Trials)
9.2.9 Product Launch Frequency (Annual)
9.2.10 Distribution Network Coverage (Germany)
9.2.11 Strategic Partnerships/Collaborations (Annual)
9.2.12 Patent Portfolio Size (Relevant to Vaccines & Therapeutics)
9.2.13 Brand Recognition (Market Survey Score or Index)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 BioNTech SE
9.5.2 CureVac AG
9.5.3 Merck KGaA
9.5.4 Boehringer Ingelheim GmbH
9.5.5 Bayer AG
9.5.6 Pfizer Deutschland GmbH
9.5.7 Sanofi-Aventis Deutschland GmbH
9.5.8 Novartis Pharma GmbH
9.5.9 Roche Deutschland Holding GmbH
9.5.10 GSK GmbH
9.5.11 AbbVie Deutschland GmbH
9.5.12 Johnson & Johnson GmbH
9.5.13 Bavarian Nordic GmbH
9.5.14 Evotec SE
9.5.15 Biotest AG
10. Germany Biotech Vaccines & Therapeutics Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Health Ministry Procurement Trends
10.1.2 Budget Allocation for Vaccines
10.1.3 Collaboration with Biotech Firms
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in Biotech Facilities
10.2.2 Funding for Research Initiatives
10.2.3 Partnerships with Academic Institutions
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Vaccine Distribution
10.3.2 Access to Innovative Therapies
10.3.3 Budget Constraints
10.4 User Readiness for Adoption
10.4.1 Awareness of Biotech Solutions
10.4.2 Training and Support Needs
10.4.3 Adoption Barriers
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Evaluation of Vaccine Efficacy
10.5.2 Long-term Cost Savings
10.5.3 Opportunities for New Applications
11. Germany Biotech Vaccines & Therapeutics Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Efforts
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix Considerations
9.1.2 Pricing Band Analysis
9.1.3 Packaging Strategies
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Market Entry
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from German biotech associations and health organizations
- Review of scientific publications and clinical trial registries related to vaccines and therapeutics
- Examination of government policy documents and funding initiatives in the biotech sector
Primary Research
- Interviews with R&D heads at leading biotech firms in Germany
- Surveys with healthcare professionals involved in vaccine administration and therapeutic applications
- Focus groups with industry experts and regulatory bodies to gather insights on market trends
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert opinions
- Triangulation of quantitative data with qualitative insights from interviews
- Sanity checks through peer reviews and expert panel discussions
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on national healthcare expenditure and biotech funding
- Segmentation of the market by vaccine types and therapeutic categories
- Incorporation of demographic data and disease prevalence rates in Germany
Bottom-up Modeling
- Collection of sales data from major biotech companies and vaccine manufacturers
- Estimation of production capacities and operational costs for vaccine development
- Volume x pricing analysis for different therapeutic segments
Forecasting & Scenario Analysis
- Utilization of time-series analysis to project market growth based on historical data
- Scenario modeling based on potential regulatory changes and public health initiatives
- Development of baseline, optimistic, and pessimistic forecasts through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Vaccine Development Insights | 90 | Biotech R&D Directors, Clinical Trial Managers |
| Therapeutics Market Trends | 70 | Pharmaceutical Product Managers, Regulatory Affairs Specialists |
| Healthcare Provider Perspectives | 110 | Physicians, Pharmacists, Public Health Officials |
| Investment and Funding Analysis | 50 | Venture Capitalists, Biotech Investors |
| Regulatory Compliance Insights | 60 | Regulatory Affairs Managers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Germany Biotech Vaccines & Therapeutics Market?
The Germany Biotech Vaccines & Therapeutics Market is valued at approximately USD 46 billion, reflecting significant growth driven by increased investments in research and development, rising disease prevalence, and a demand for personalized medicine.