About the Report
Base Year 2024Global AI Enabled Medical Devices Market Overview
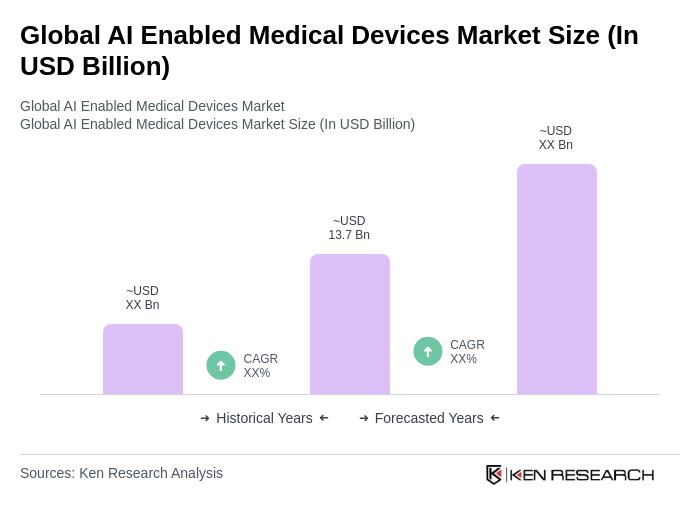
- The Global AI Enabled Medical Devices Market is valued at USD 13.7 billion, based on a five-year historical analysis. This growth is primarily driven by advancements in artificial intelligence technology, increasing demand for personalized and precision healthcare solutions, and the rising prevalence of chronic diseases. The integration of AI in medical devices enhances diagnostic accuracy, automates clinical workflows, improves treatment efficiency, and enables continuous patient monitoring, thereby propelling market expansion. Additional growth drivers include the rapid adoption of AI-powered imaging, wearable monitoring devices, and telehealth platforms, as well as supportive regulatory and reimbursement environments.
- Key players in this market include the United States, Germany, and China. The United States leads due to its robust healthcare infrastructure, significant investments in research and development, and a high adoption rate of innovative technologies. Germany benefits from its strong manufacturing base and proactive regulatory support, while China is rapidly expanding its healthcare sector, focusing on AI integration to improve patient outcomes and address the needs of an aging population.
- In 2023, the U.S. government implemented the 21st Century Cures Act, issued by the U.S. Congress in 2016 and enforced by the Food and Drug Administration (FDA), which aims to accelerate the development and approval of innovative medical devices, including AI-enabled technologies. This regulation streamlines the FDA approval process for new devices, establishes standards for clinical validation, and facilitates faster access to market while ensuring safety and efficacy, thereby fostering growth in the AI-enabled medical devices sector.
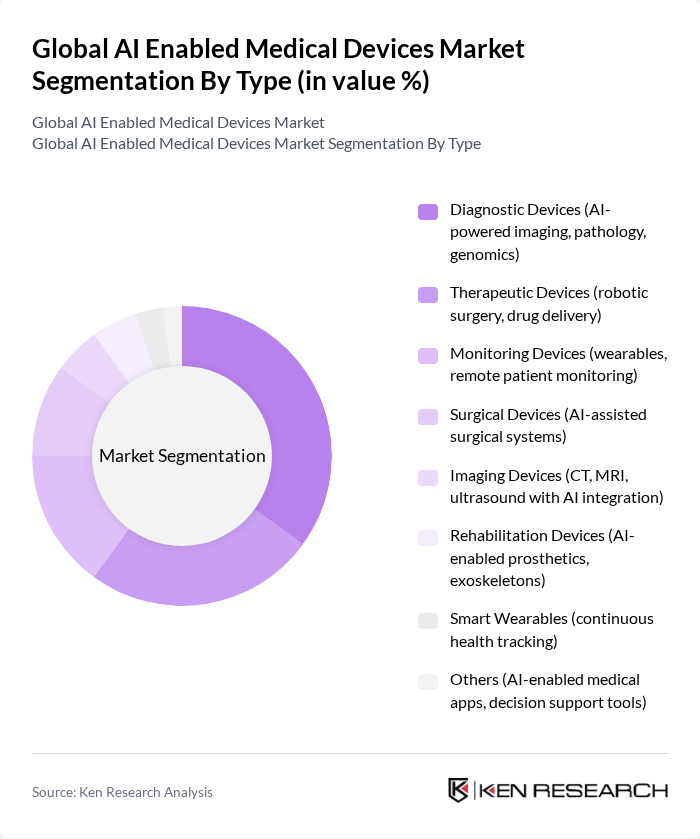
Global AI Enabled Medical Devices Market Segmentation
By Type:The market is segmented into various types of devices, including diagnostic devices, therapeutic devices, monitoring devices, surgical devices, imaging devices, rehabilitation devices, smart wearables, and others. Among these, diagnostic devices are currently leading the market due to their critical role in early disease detection and management. The increasing demand for AI-powered imaging, pathology, and genomics solutions is driving this segment's growth, as healthcare providers seek to enhance diagnostic accuracy, automate image analysis, and improve clinical decision-making.
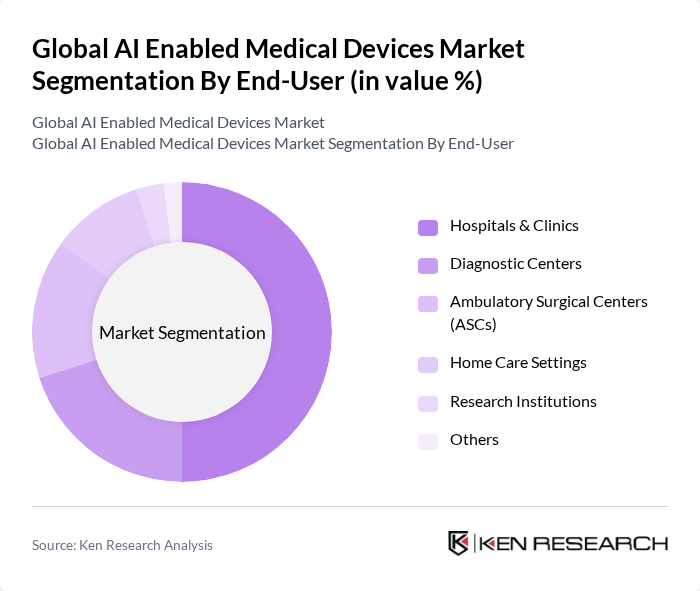
By End-User:The market is segmented by end-users, including hospitals & clinics, diagnostic centers, ambulatory surgical centers (ASCs), home care settings, research institutions, and others. Hospitals and clinics dominate this segment, driven by the increasing adoption of AI technologies to improve patient care, operational efficiency, and clinical outcomes. The growing need for advanced diagnostic and therapeutic solutions in these settings is propelling the demand for AI-enabled medical devices, with hospitals leading in revenue share and device deployment.
Global AI Enabled Medical Devices Market Competitive Landscape
The Global AI Enabled Medical Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Siemens Healthineers, Philips Healthcare, GE Healthcare, Medtronic, IBM Watson Health, Johnson & Johnson (Ethicon, DePuy Synthes), Boston Scientific, Abbott Laboratories, Stryker Corporation, Canon Medical Systems, Nuance Communications (Microsoft Health AI), Zebra Medical Vision, Aidoc, Tempus Labs, PathAI, Butterfly Network, HeartFlow, Viz.ai, Cleerly, Enlitic contribute to innovation, geographic expansion, and service delivery in this space.
Global AI Enabled Medical Devices Market Industry Analysis
Growth Drivers
- Increasing Demand for Personalized Medicine:The global personalized medicine market is projected to reach $2.5 trillion in future, driven by advancements in genomics and biotechnology. This surge is prompting healthcare providers to adopt AI-enabled medical devices that can analyze patient data for tailored treatments. The World Health Organization reported that personalized medicine can improve patient outcomes by 30%, further fueling demand for innovative AI solutions in healthcare settings.
- Advancements in Machine Learning Algorithms:The machine learning market in healthcare is expected to grow to $20 billion in future, with AI algorithms enhancing diagnostic accuracy and treatment efficacy. These advancements enable devices to process vast datasets, leading to improved patient care. According to a report by Frost & Sullivan, AI-driven diagnostics can reduce diagnostic errors by up to 50%, significantly impacting healthcare delivery and operational efficiency.
- Rising Healthcare Expenditure:Global healthcare spending is projected to reach $10 trillion in future, driven by aging populations and increased chronic disease prevalence. This rise in expenditure is facilitating investments in AI-enabled medical devices, which are essential for improving healthcare delivery. The Centers for Medicare & Medicaid Services reported that healthcare spending in the U.S. alone is expected to grow at an annual rate of approximately 5%, highlighting the financial commitment to advanced medical technologies.
Market Challenges
- High Development Costs:The development of AI-enabled medical devices often requires significant financial investment, with costs averaging around $1 billion for bringing a new device to market. This high barrier to entry can deter smaller companies from innovating. According to the National Institutes of Health, only 1 in 10 medical devices successfully navigates the development process, underscoring the financial risks involved in this sector.
- Regulatory Hurdles:Navigating the regulatory landscape for AI-enabled medical devices can be complex and time-consuming. The FDA has issued over 100 guidance documents for AI technologies, which can delay product launches. A report from the Regulatory Affairs Professionals Society indicates that the average time for regulatory approval can exceed 18 months, posing a significant challenge for companies aiming to introduce innovative solutions quickly.
Global AI Enabled Medical Devices Market Future Outlook
The future of AI-enabled medical devices is poised for transformative growth, driven by technological advancements and increasing healthcare demands. As telemedicine and remote patient monitoring become mainstream, the integration of AI will enhance patient engagement and care efficiency. Furthermore, the focus on cybersecurity will ensure that patient data remains secure, fostering trust in AI technologies. The collaboration between healthcare providers and tech companies will likely accelerate innovation, leading to more sophisticated medical devices that improve patient outcomes and operational efficiencies.
Market Opportunities
- Expansion in Emerging Markets:Emerging markets, particularly in Asia and Africa, are experiencing rapid healthcare infrastructure development. The World Bank estimates that healthcare spending in these regions will increase by approximately 10% annually, creating significant opportunities for AI-enabled medical devices. This growth can enhance access to advanced healthcare solutions, addressing unmet medical needs in these regions.
- Development of Wearable Devices:The wearable medical device market is projected to reach $60 billion in future, driven by consumer demand for health monitoring solutions. AI integration in wearables can provide real-time health insights, improving chronic disease management. According to a report by Grand View Research, the increasing prevalence of lifestyle-related diseases will further boost the demand for AI-enabled wearables, presenting a lucrative opportunity for manufacturers.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Diagnostic Devices (AI-powered imaging, pathology, genomics) Therapeutic Devices (robotic surgery, drug delivery) Monitoring Devices (wearables, remote patient monitoring) Surgical Devices (AI-assisted surgical systems) Imaging Devices (CT, MRI, ultrasound with AI integration) Rehabilitation Devices (AI-enabled prosthetics, exoskeletons) Smart Wearables (continuous health tracking) Others (AI-enabled medical apps, decision support tools) |
| By End-User | Hospitals & Clinics Diagnostic Centers Ambulatory Surgical Centers (ASCs) Home Care Settings Research Institutions Others |
| By Application | Cardiology Neurology Radiology Oncology Mental and Behavioral Health Ophthalmology Orthopedics Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | North America (U.S., Canada, Mexico) Europe (Germany, UK, France, Italy, Spain, Nordics) Asia-Pacific (China, Japan, India, South Korea, Australia, Thailand) Latin America (Brazil, Argentina) Middle East & Africa (South Africa, Saudi Arabia, UAE, Kuwait) |
| By Component | Hardware Software (AI algorithms, SaMD) Services (integration, support) |
| By Price Range | Low Price Mid Price High Price |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., FDA, EMA)
Manufacturers and Producers
Healthcare Providers and Hospitals
Medical Device Distributors
Health Insurance Companies
Technology Providers and Software Developers
Industry Associations and Advocacy Groups
Players Mentioned in the Report:
Siemens Healthineers
Philips Healthcare
GE Healthcare
Medtronic
IBM Watson Health
Johnson & Johnson (Ethicon, DePuy Synthes)
Boston Scientific
Abbott Laboratories
Stryker Corporation
Canon Medical Systems
Nuance Communications (Microsoft Health AI)
Zebra Medical Vision
Aidoc
Tempus Labs
PathAI
Butterfly Network
HeartFlow
Viz.ai
Cleerly
Enlitic
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global AI Enabled Medical Devices Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global AI Enabled Medical Devices Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global AI Enabled Medical Devices Market Analysis
3.1 Growth Drivers
3.1.1 Increasing Demand for Personalized Medicine
3.1.2 Advancements in Machine Learning Algorithms
3.1.3 Rising Healthcare Expenditure
3.1.4 Growing Adoption of Telemedicine
3.2 Market Challenges
3.2.1 High Development Costs
3.2.2 Regulatory Hurdles
3.2.3 Data Privacy Concerns
3.2.4 Integration with Existing Systems
3.3 Market Opportunities
3.3.1 Expansion in Emerging Markets
3.3.2 Collaborations with Tech Companies
3.3.3 Development of Wearable Devices
3.3.4 AI in Diagnostics and Imaging
3.4 Market Trends
3.4.1 Increased Investment in AI Research
3.4.2 Shift Towards Remote Patient Monitoring
3.4.3 Integration of AI with IoT Devices
3.4.4 Focus on Cybersecurity in Medical Devices
3.5 Government Regulation
3.5.1 FDA Guidelines for AI Devices
3.5.2 CE Marking for Medical Devices
3.5.3 HIPAA Compliance for Data Security
3.5.4 International Standards for AI in Healthcare
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global AI Enabled Medical Devices Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global AI Enabled Medical Devices Market Segmentation
8.1 By Type
8.1.1 Diagnostic Devices (AI-powered imaging, pathology, genomics)
8.1.2 Therapeutic Devices (robotic surgery, drug delivery)
8.1.3 Monitoring Devices (wearables, remote patient monitoring)
8.1.4 Surgical Devices (AI-assisted surgical systems)
8.1.5 Imaging Devices (CT, MRI, ultrasound with AI integration)
8.1.6 Rehabilitation Devices (AI-enabled prosthetics, exoskeletons)
8.1.7 Smart Wearables (continuous health tracking)
8.1.8 Others (AI-enabled medical apps, decision support tools)
8.2 By End-User
8.2.1 Hospitals & Clinics
8.2.2 Diagnostic Centers
8.2.3 Ambulatory Surgical Centers (ASCs)
8.2.4 Home Care Settings
8.2.5 Research Institutions
8.2.6 Others
8.3 By Application
8.3.1 Cardiology
8.3.2 Neurology
8.3.3 Radiology
8.3.4 Oncology
8.3.5 Mental and Behavioral Health
8.3.6 Ophthalmology
8.3.7 Orthopedics
8.3.8 Others
8.4 By Distribution Channel
8.4.1 Direct Sales
8.4.2 Distributors
8.4.3 Online Sales
8.4.4 Others
8.5 By Region
8.5.1 North America (U.S., Canada, Mexico)
8.5.2 Europe (Germany, UK, France, Italy, Spain, Nordics)
8.5.3 Asia-Pacific (China, Japan, India, South Korea, Australia, Thailand)
8.5.4 Latin America (Brazil, Argentina)
8.5.5 Middle East & Africa (South Africa, Saudi Arabia, UAE, Kuwait)
8.6 By Component
8.6.1 Hardware
8.6.2 Software (AI algorithms, SaMD)
8.6.3 Services (integration, support)
8.7 By Price Range
8.7.1 Low Price
8.7.2 Mid Price
8.7.3 High Price
9. Global AI Enabled Medical Devices Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Company Size (Large, Medium, Small)
9.2.3 Revenue Growth Rate (YoY %)
9.2.4 Market Penetration Rate (devices deployed, geographic reach)
9.2.5 Regulatory Approvals (number of FDA/CE-cleared AI devices)
9.2.6 R&D Investment (% of revenue)
9.2.7 Product Innovation Rate (new AI device launches/year)
9.2.8 Clinical Validation (number of peer-reviewed studies, trials)
9.2.9 Strategic Partnerships (number, type)
9.2.10 Customer Satisfaction Score (NPS, user ratings)
9.2.11 Pricing Strategy
9.2.12 Distribution Efficiency (channels, global coverage)
9.2.13 Brand Recognition (awards, rankings)
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Siemens Healthineers
9.5.2 Philips Healthcare
9.5.3 GE Healthcare
9.5.4 Medtronic
9.5.5 IBM Watson Health
9.5.6 Johnson & Johnson (Ethicon, DePuy Synthes)
9.5.7 Boston Scientific
9.5.8 Abbott Laboratories
9.5.9 Stryker Corporation
9.5.10 Canon Medical Systems
9.5.11 Nuance Communications (Microsoft Health AI)
9.5.12 Zebra Medical Vision
9.5.13 Aidoc
9.5.14 Tempus Labs
9.5.15 PathAI
9.5.16 Butterfly Network
9.5.17 HeartFlow
9.5.18 Viz.ai
9.5.19 Cleerly
9.5.20 Enlitic
10. Global AI Enabled Medical Devices Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Procurement Channels
10.1.4 Evaluation Criteria for Vendors
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment in AI Technologies
10.2.2 Budget for Medical Equipment
10.2.3 Spending on Training and Development
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Integration
10.3.2 Cost Constraints
10.3.3 Need for Training
10.4 User Readiness for Adoption
10.4.1 Awareness Levels
10.4.2 Training Needs
10.4.3 Technology Acceptance
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of ROI
10.5.2 Expansion Opportunities
10.5.3 User Feedback Mechanisms
11. Global AI Enabled Medical Devices Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Framework
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of market reports from healthcare industry associations and research firms
- Review of published articles in medical journals focusing on AI applications in healthcare
- Examination of regulatory frameworks and guidelines from health authorities regarding AI-enabled devices
Primary Research
- Interviews with product managers at leading medical device manufacturers
- Surveys with healthcare professionals using AI-enabled devices in clinical settings
- Focus groups with patients to understand user experience and acceptance of AI technologies
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and industry reports
- Triangulation of data from primary research and secondary sources to ensure consistency
- Sanity checks through feedback from a panel of healthcare technology experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global healthcare expenditure and technology adoption rates
- Segmentation of the market by device type, application, and geographical region
- Incorporation of trends in telemedicine and remote patient monitoring into market forecasts
Bottom-up Modeling
- Collection of sales data from key manufacturers of AI-enabled medical devices
- Estimation of market penetration rates based on device usage in various healthcare settings
- Calculation of revenue projections based on average selling prices and expected sales volumes
Forecasting & Scenario Analysis
- Multi-variable forecasting using historical growth rates and emerging technology trends
- Scenario analysis based on potential regulatory changes and market disruptions
- Development of baseline, optimistic, and pessimistic market growth scenarios through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| AI-Enabled Diagnostic Devices | 100 | Radiologists, Pathologists, Medical Device Engineers |
| Wearable Health Monitoring Devices | 80 | Cardiologists, General Practitioners, Patients |
| Robotic Surgery Systems | 60 | Surgeons, Surgical Technologists, Hospital Administrators |
| AI in Imaging Technologies | 90 | Imaging Technicians, Healthcare IT Specialists, Clinical Researchers |
| Telehealth Solutions | 70 | Telehealth Coordinators, IT Managers, Healthcare Policy Makers |
Frequently Asked Questions
What is the current value of the Global AI Enabled Medical Devices Market?
The Global AI Enabled Medical Devices Market is valued at approximately USD 13.7 billion, driven by advancements in AI technology, increasing demand for personalized healthcare solutions, and the rising prevalence of chronic diseases.