Region:Global
Author(s):Geetanshi
Product Code:KRAD4873
Pages:97
Published On:December 2025
By Type:The market is segmented into various types of sample preparation techniques, including Solid Phase Extraction (SPE), Liquid-Liquid Extraction (LLE), Protein Precipitation, Filtration & Centrifugation, Dilution & Evaporation, QuEChERS and Dispersive SPE, and Others (Derivatization, Microwave-Assisted, etc.). Each of these techniques serves specific applications and industries, contributing to the overall market dynamics, with growing adoption of automated and high-throughput formats (such as 96?well plates, robotic platforms, and cartridge systems) in regulated and research laboratories.
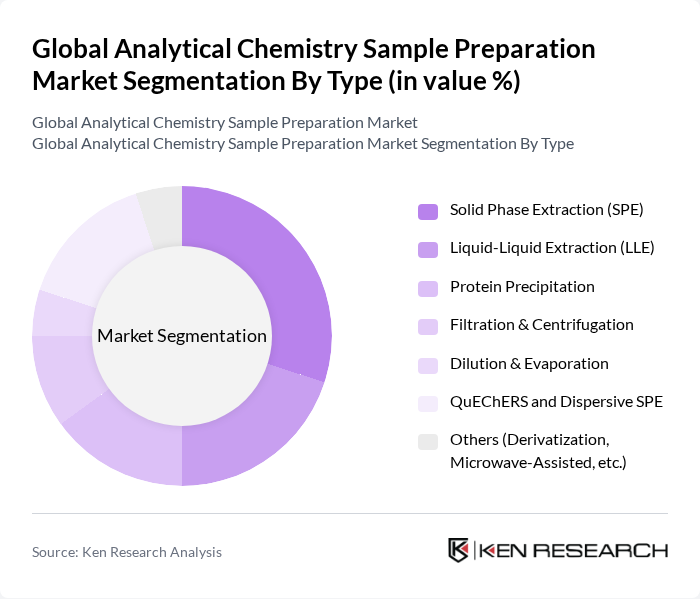
The Solid Phase Extraction (SPE) technique is currently dominating the market due to its efficiency in isolating analytes from complex matrices, which is crucial in pharmaceutical, environmental, and food-testing applications. The increasing focus on high-throughput analysis, automation of LC?MS and GC?MS workflows, and the need for reproducibility in results are driving the adoption of SPE in cartridge, disk, plate, and on?line formats. Additionally, advancements in SPE sorbent chemistries (such as mixed?mode, polymeric, and specialty phases) and method miniaturization are enhancing its applicability across bioanalysis, environmental monitoring, and food residue testing, making it a preferred choice among laboratories.
By End-User:The market is segmented based on end-users, including Pharmaceutical & Biotechnology Companies, Contract Research Organizations (CROs) & Contract Testing Laboratories, Environmental Testing Laboratories, Food & Beverage Testing Laboratories, Clinical & Diagnostic Laboratories, Academic & Research Institutes, and Others. Each end-user segment has unique requirements and drives demand for specific sample preparation techniques, with pharmaceutical, biotechnology, and contract testing organizations showing strong uptake of automated and kit-based solutions to support bioanalysis, stability testing, and quality control.

Pharmaceutical & Biotechnology Companies are the leading end-users in the analytical chemistry sample preparation market, reflecting the intensity of small?molecule and biologics R&D, bioanalytical testing, and regulatory compliance activities in this sector. The increasing focus on complex drug modalities (such as biologics and cell and gene therapies), stringent regulatory requirements for method validation and quality control, and growing volumes of samples in clinical trials are driving the demand for advanced, automated, and kit-based sample preparation techniques. These companies require high precision, accuracy, and data integrity in their analytical processes, which further enhances the adoption of innovative sample preparation methods, including automated platforms, microplates, and integrated software-controlled workflows.
The Global Analytical Chemistry Sample Preparation Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific Inc., Agilent Technologies Inc., Waters Corporation, Revvity Inc. (formerly PerkinElmer Inc.), Merck KGaA (MilliporeSigma), Shimadzu Corporation, Sartorius AG, Bio-Rad Laboratories Inc., Phenomenex Inc. (a Danaher company), Eppendorf SE, Avantor Inc. (including VWR), Restek Corporation, Tecan Group Ltd., Hamilton Company, Gerstel GmbH & Co. KG contribute to innovation, geographic expansion, and service delivery in this space.
The future of the analytical chemistry sample preparation market appears promising, driven by technological advancements and increasing regulatory demands. As laboratories increasingly adopt automated systems, efficiency and accuracy in sample preparation will improve significantly. Furthermore, the integration of artificial intelligence and machine learning into analytical processes is expected to enhance data analysis capabilities, leading to more precise results. These trends will likely foster innovation and collaboration among industry players, positioning the market for sustained growth in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | Solid Phase Extraction (SPE) Liquid-Liquid Extraction (LLE) Protein Precipitation Filtration & Centrifugation Dilution & Evaporation QuEChERS and Dispersive SPE Others (Derivatization, Microwave-Assisted, etc.) |
| By End-User | Pharmaceutical & Biotechnology Companies Contract Research Organizations (CROs) & Contract Testing Laboratories Environmental Testing Laboratories Food & Beverage Testing Laboratories Clinical & Diagnostic Laboratories Academic & Research Institutes Others |
| By Application | Chromatography Sample Preparation Mass Spectrometry Sample Preparation Spectroscopy Sample Preparation Genomics & Proteomics Sample Preparation Drug Discovery & Development Quality Control & Regulatory Testing Others |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology / Workflow | Manual Sample Preparation Semi-automated Sample Preparation Fully Automated & Robotic Sample Preparation Systems High-throughput Sample Preparation |
| By Product Type | Instruments & Systems Consumables (Cartridges, Columns, Plates, Tips, Vials, etc.) Reagents & Kits Software & Informatics Accessories & Others |
| By Service Type | Method Development & Validation Services Sample Preparation Outsourcing Services Training & Application Support Services Instrument Installation, Maintenance & Calibration Services Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Pharmaceutical Sample Preparation | 100 | Laboratory Managers, Quality Control Analysts |
| Environmental Testing Laboratories | 80 | Environmental Scientists, Lab Technicians |
| Food Safety Testing | 70 | Food Safety Inspectors, Quality Assurance Managers |
| Academic Research Institutions | 60 | Research Professors, Graduate Students |
| Industrial Chemical Analysis | 90 | Process Engineers, R&D Managers |
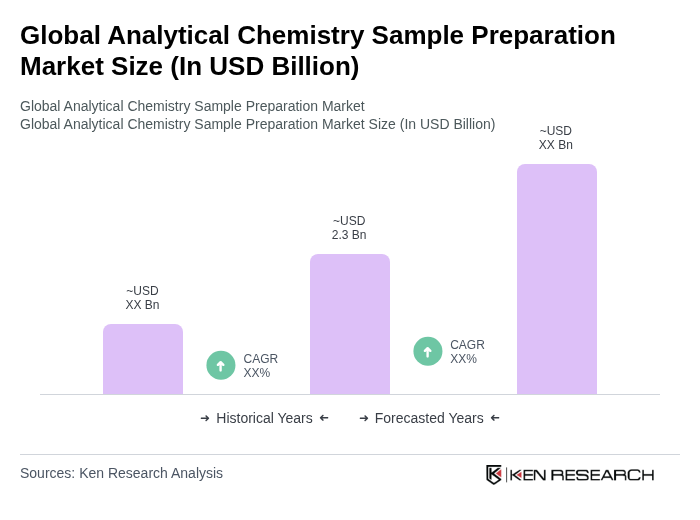
The Global Analytical Chemistry Sample Preparation Market is valued at approximately USD 2.3 billion, driven by the increasing demand for high-quality analytical results across various sectors, including pharmaceuticals, biotechnology, environmental testing, and food safety.