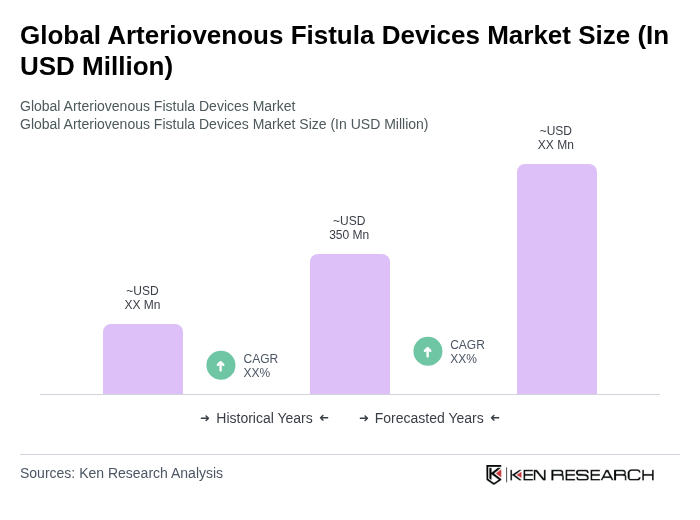
Global Arteriovenous Fistula Devices Market Overview
- The Global Arteriovenous Fistula Devices Market is valued at USD 350 million, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of chronic kidney diseases and the rising number of patients requiring dialysis, supplemented by growing healthcare spending and investments in chronic disease management. The demand for arteriovenous fistula devices is further supported by advancements in medical technology, such as minimally invasive endovascular procedures, and the growing awareness of the benefits of arteriovenous fistulas over other vascular access methods.
- Key players in this market are concentrated in regions such as North America and Europe, where healthcare infrastructure is well-developed, and there is a high prevalence of chronic diseases. Countries like the United States and Germany dominate the market due to their advanced healthcare systems, significant investments in medical research, a high number of dialysis patients, and strong government support for chronic kidney disease management programs, which drives the demand for arteriovenous fistula devices.
- In recent years, regulatory bodies have implemented stringent guidelines to ensure the safety and efficacy of medical devices, including arteriovenous fistula devices. For instance, the Medical Device Regulation (MDR) 2017/745 issued by the European Parliament and the Council in 2017 requires manufacturers to conduct clinical evaluations, implement a quality management system, and obtain CE marking certification for Class IIb and III devices like arteriovenous fistula creation systems, ensuring compliance through post-market surveillance and performance thresholds for vascular access patency.
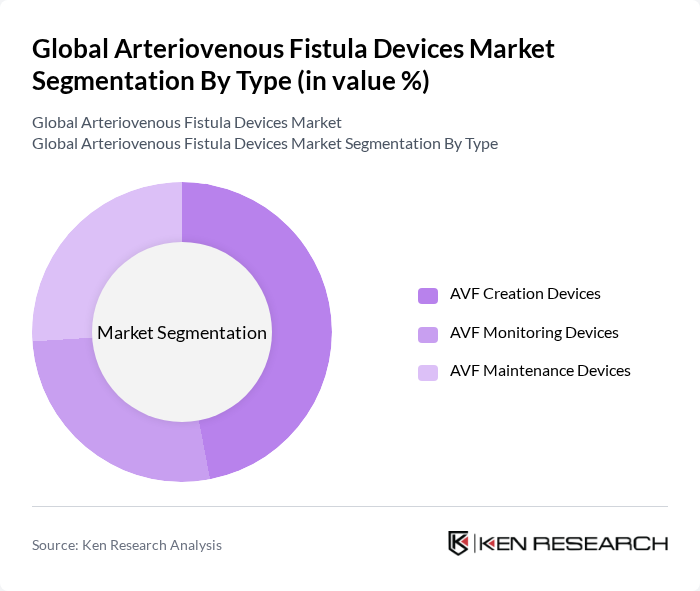
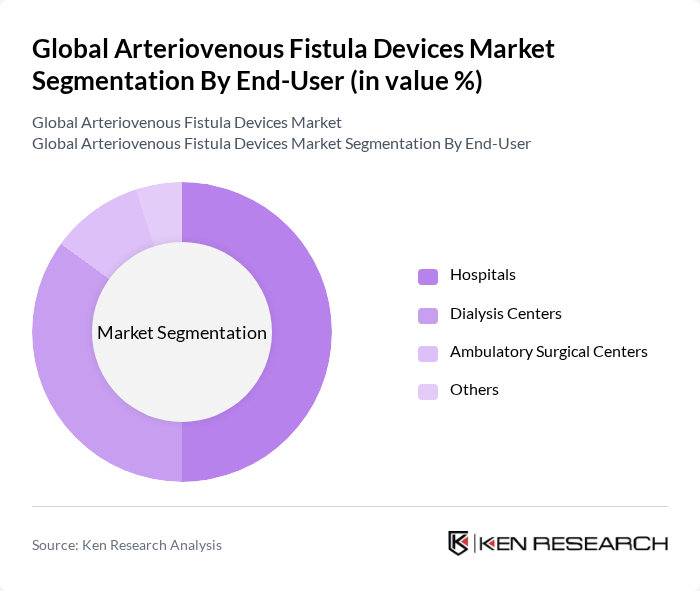
Global Arteriovenous Fistula Devices Market Segmentation
By Type:
The arteriovenous fistula devices market is segmented into three main types: AVF Creation Devices, AVF Monitoring Devices, and AVF Maintenance Devices. Among these, AVF Creation Devices dominate the market due to the increasing number of patients requiring dialysis, the preference for arteriovenous fistulas as the preferred vascular access method, and strong demand for minimally invasive surgery and surgical fistula procedures that provide long-term vascular access with fewer complications compared to synthetic grafts or catheters. The growing awareness of the benefits of AVF, such as lower infection rates and better long-term outcomes, drives the demand for these devices. Additionally, advancements in technology, including radiofrequency or thermal energy systems, have improved the efficiency and effectiveness of AVF creation, further solidifying its market leadership.
By End-User:
The market is also segmented by end-user, which includes Hospitals, Ambulatory Surgical Centers, and Dialysis Centers. Hospitals are the leading end-user segment, primarily due to their advanced surgical infrastructure, trained vascular specialists, and high dialysis patient volumes that implement long-term care programs promoting AVF device adoption. These centers are specifically designed to cater to the needs of dialysis patients, making them the primary consumers of arteriovenous fistula devices. The increasing number of dialysis centers and the growing patient population further enhance the demand for these devices in this segment, with ambulatory surgical centers showing fastest growth from preference for outpatient dialysis procedures and minimally invasive AVF creation.
Global Arteriovenous Fistula Devices Market Competitive Landscape
The Global Arteriovenous Fistula Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Fresenius Medical Care, Baxter International Inc., B. Braun Melsungen AG, Medtronic plc, BD (Becton Dickinson), Terumo Corporation, Nipro Corporation, Cook Medical, AngioDynamics, Inc., Asahi Intecc Co., Ltd., Merit Medical Systems, Inc., Teleflex Incorporated, Cardinal Health, Inc., Smiths Medical, Boston Scientific Corporation contribute to innovation, geographic expansion, and service delivery in this space.
Global Arteriovenous Fistula Devices Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Chronic Kidney Disease:The World Health Organization reported that approximately 850 million people worldwide suffer from chronic kidney disease (CKD), with a significant portion requiring dialysis. In future, the prevalence of CKD is projected to rise by 5% annually, driven by factors such as diabetes and hypertension. This growing patient population necessitates effective vascular access solutions, including arteriovenous fistulas, thereby propelling market growth and increasing demand for related devices.
- Rising Demand for Dialysis Treatments:According to the International Society of Nephrology, the number of patients requiring dialysis is expected to reach 3 million in future. This surge is primarily due to the aging population and the increasing incidence of lifestyle-related diseases. As dialysis becomes more prevalent, the need for reliable and efficient arteriovenous fistula devices will escalate, driving market expansion and innovation in device technology to meet patient needs.
- Technological Advancements in Device Design:The arteriovenous fistula device market is witnessing rapid technological advancements, with innovations such as bioengineered grafts and smart monitoring systems. For instance, the introduction of biocompatible materials has improved patient outcomes, reducing complications by 30%. In future, these advancements are expected to enhance the efficacy and safety of AV fistulas, thereby increasing their adoption among healthcare providers and patients alike, further stimulating market growth.
Market Challenges
- High Cost of Surgical Procedures:The average cost of creating an arteriovenous fistula in future can exceed $15,000, which poses a significant barrier for many patients and healthcare systems. This high cost is often compounded by additional expenses related to post-operative care and potential complications. As a result, many patients may opt for less expensive alternatives, limiting the market potential for AV fistula devices and creating challenges for manufacturers in this sector.
- Complications Associated with AV Fistula Creation:Complications such as thrombosis and infection occur in approximately 20% of AV fistula procedures, leading to increased healthcare costs and patient dissatisfaction. In future, these complications can result in extended hospital stays, with an average additional cost of $8,000 per patient. Such challenges not only affect patient outcomes but also deter healthcare providers from recommending AV fistulas, impacting market growth negatively.
Global Arteriovenous Fistula Devices Market Future Outlook
The future of the arteriovenous fistula devices market in future appears promising, driven by increasing healthcare investments and a focus on improving patient outcomes. The integration of digital health technologies is expected to enhance monitoring and management of dialysis patients, while the shift towards home dialysis solutions will create new avenues for device innovation. As healthcare providers prioritize patient-centric care, the demand for advanced AV fistula devices will likely grow, fostering a competitive landscape for manufacturers.
Market Opportunities
- Expansion into Emerging Markets:Emerging markets in future present significant growth opportunities for arteriovenous fistula devices. With rising healthcare expenditures projected to reach $200 billion in future, manufacturers can capitalize on the increasing demand for dialysis solutions, particularly in regions with high CKD prevalence, thereby enhancing their market presence and revenue potential.
- Development of Minimally Invasive Techniques:The trend towards minimally invasive surgical techniques is gaining traction in future, with a projected increase in demand for such procedures by 25% in future. This shift offers manufacturers the opportunity to innovate and develop advanced AV fistula devices that reduce recovery times and complications, ultimately improving patient satisfaction and expanding market share.