Region:Middle East
Author(s):Geetanshi
Product Code:KRAC3139
Pages:90
Published On:October 2025
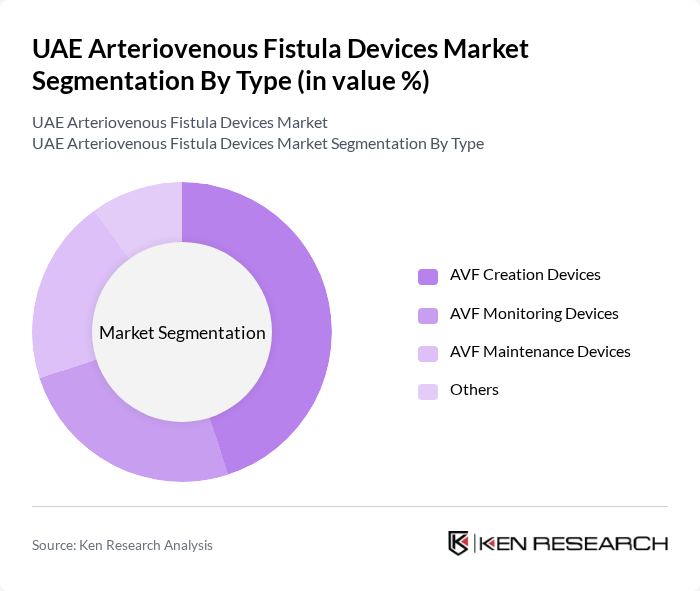
By Type:The market is segmented into various types of arteriovenous fistula devices, including AVF Creation Devices, AVF Monitoring Devices, AVF Maintenance Devices, and Others. Among these, AVF Creation Devices are the most prominent due to their critical role in establishing vascular access for dialysis patients. The increasing number of patients requiring hemodialysis has led to a higher demand for these devices, making them a key focus for manufacturers and healthcare providers. Recent trends highlight the adoption of advanced imaging technologies and AI-assisted monitoring to enhance procedural accuracy and patient safety.
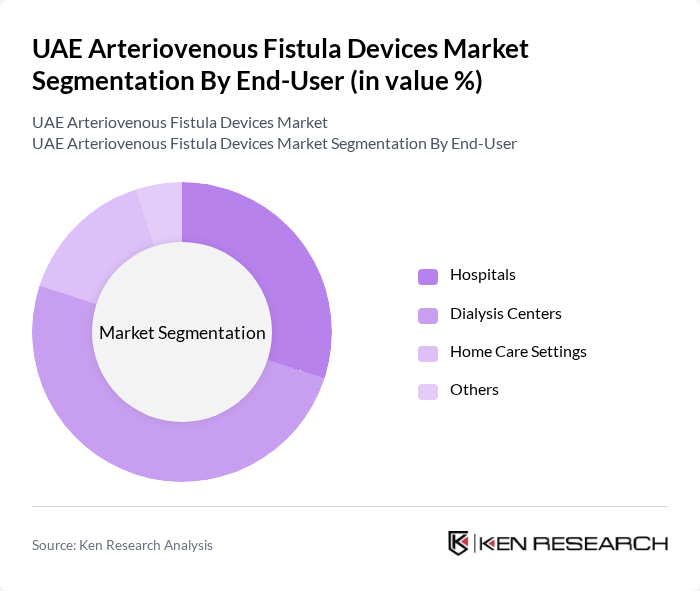
By End-User:The end-user segmentation includes Hospitals, Dialysis Centers, Home Care Settings, and Others. Dialysis Centers are the leading end-users of arteriovenous fistula devices, driven by the increasing number of patients undergoing dialysis treatments. These centers require a consistent supply of high-quality devices to ensure effective and safe procedures, thus significantly influencing market dynamics. The growth of ambulatory surgical centers and the gradual expansion of home dialysis programs also contribute to the diversification of end-user segments.
The UAE Arteriovenous Fistula Devices Market is characterized by a dynamic mix of regional and international players. Leading participants such as Fresenius Medical Care AG & Co. KGaA, Baxter International Inc., B. Braun Melsungen AG, Medtronic plc, Terumo Corporation, Nipro Corporation, Asahi Kasei Corporation, Cook Medical Inc., AngioDynamics, Inc., Teleflex Incorporated, Smiths Medical, Cardinal Health, Inc., 3M Company, Halyard Health, Inc., Vascular Solutions, Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the UAE arteriovenous fistula devices market appears promising, driven by ongoing advancements in medical technology and a growing emphasis on patient-centric care. As healthcare infrastructure expands, particularly in rural areas, access to these devices will improve. Additionally, the integration of digital health technologies is expected to enhance patient monitoring and outcomes, fostering a more efficient healthcare delivery system. These trends indicate a robust growth trajectory for the market in the coming years.
| Segment | Sub-Segments |
|---|---|
| By Type | AVF Creation Devices AVF Monitoring Devices AVF Maintenance Devices Others |
| By End-User | Hospitals Dialysis Centers Home Care Settings Others |
| By Distribution Channel | Direct Sales Distributors Online Sales Others |
| By Region | Abu Dhabi Dubai Sharjah Others |
| By Application | Hemodialysis Therapeutic Apheresis Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Nephrology Clinics | 60 | Nephrologists, Clinic Managers |
| Hospitals with Dialysis Units | 50 | Vascular Surgeons, Procurement Officers |
| Patient Advocacy Groups | 40 | Patient Representatives, Healthcare Advocates |
| Medical Device Distributors | 45 | Sales Managers, Product Specialists |
| Regulatory Bodies | 20 | Regulatory Affairs Officers, Policy Makers |
The UAE Arteriovenous Fistula Devices Market is valued at approximately USD 1 million, driven by the increasing prevalence of chronic kidney diseases and the rising number of patients requiring dialysis treatments.