About the Report
Base Year 2024Global Automated Closed Cell Therapy Processing Systems Market Overview
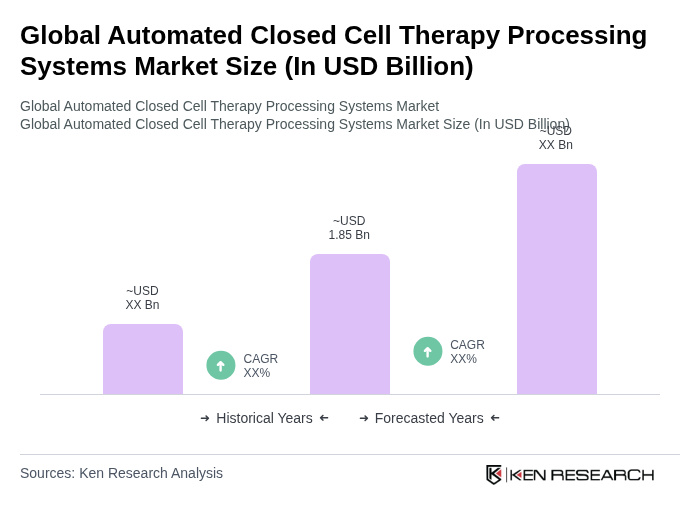
- The Global Automated Closed Cell Therapy Processing Systems Market is valued at USD 1.85 billion, based on recent analysis. Growth is driven by rising demand for cell and gene therapies, especially CAR-T and other immunotherapies, with automation offering enhanced reproducibility, sterility, and operational efficiency compared to manual methods. Furthermore, the expanding pipeline of advanced therapies and technological advances in single-use systems and AI-enabled monitoring further propel market expansion.
- Key players in this market include Thermo Fisher Scientific, Miltenyi Biotec, Lonza, Sartorius, and Cellares. These companies play a significant role in driving growth and maintaining high standards in cell therapy processing through innovation and strategic partnerships. North America leads the market due to its mature biotechnology ecosystem, accessible funding, and a high volume of cell therapy clinical activities, while Asia-Pacific is emerging quickly owing to increased investments in cell therapy infrastructure and favorable government backing.
- In 2024, the U.S. government announced a strategic plan to enhance domestic cell therapy manufacturing capabilities. This plan includes an investment of approximately USD 2.6 billion toward regenerative medicine research, including manufacturing innovation, supporting the deployment of automated processing systems for advanced therapies.
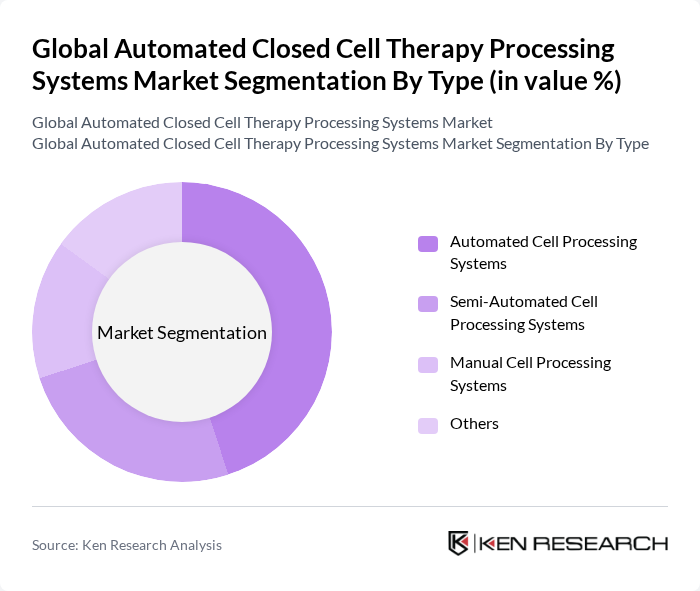
Global Automated Closed Cell Therapy Processing Systems Market Segmentation
By Type:The market is segmented into various types, including Automated Cell Processing Systems, Semi-Automated Cell Processing Systems, Manual Cell Processing Systems, and Others. Among these, Automated Cell Processing Systems dominate the market due to their efficiency and ability to minimize human error, which is critical in sensitive cell therapy applications. The trend towards automation in laboratories and manufacturing facilities is driving the adoption of these systems, as they provide consistent results and enhance throughput.
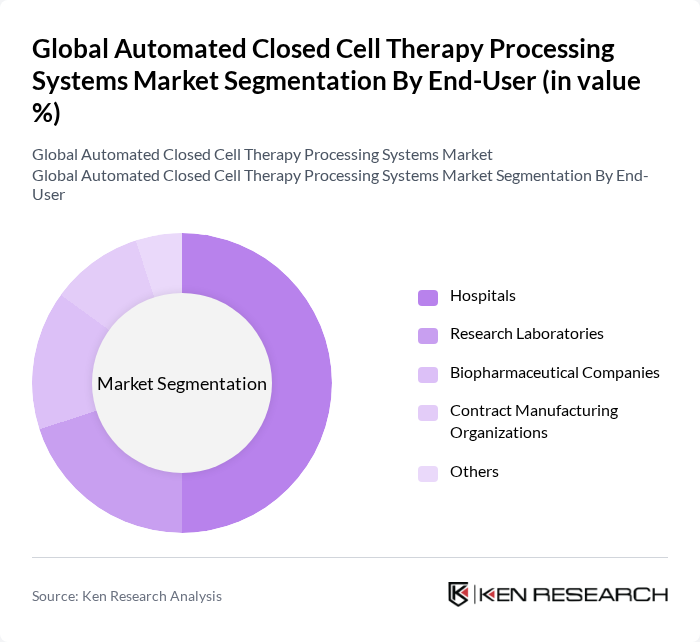
By End-User:The end-user segmentation includes Hospitals, Research Laboratories, Biopharmaceutical Companies, Contract Manufacturing Organizations, and Others. Hospitals are the leading end-users, driven by the increasing number of cell therapy procedures and the need for advanced processing systems to ensure patient safety and treatment efficacy. The growing trend of personalized medicine and the rise in chronic diseases are further propelling the demand for automated systems in hospital settings.
Global Automated Closed Cell Therapy Processing Systems Market Competitive Landscape
The Global Automated Closed Cell Therapy Processing Systems Market is characterized by a dynamic mix of regional and international players. Leading participants such as Thermo Fisher Scientific, Beckman Coulter, Miltenyi Biotec, Merck KGaA, Lonza Group, STEMCELL Technologies, Bio-Rad Laboratories, GE Healthcare, Fujifilm Irvine Scientific, Sartorius AG, Eppendorf AG, Agilent Technologies, BD Biosciences, CellGenix GmbH, Pluristem Therapeutics contribute to innovation, geographic expansion, and service delivery in this space.
Global Automated Closed Cell Therapy Processing Systems Market Industry Analysis
Growth Drivers
- Global Market Revenue:The global market for automated closed cell therapy processing systems is projected to reach USD 1,408.3 million. This significant revenue indicates a robust demand for advanced processing systems, driven by the increasing need for efficient and standardized cell therapies. The growth is supported by rising investments in biotechnology and regenerative medicine, which are crucial for enhancing treatment outcomes. Source: Grand View Research.
- North America Market Share:North America is expected to account for approximately USD 705 million, representing 50.1% of the global market share. This dominance reflects the region's advanced healthcare infrastructure and high adoption rates of innovative therapies. The concentration of leading biotech firms and research institutions further fuels the demand for automated systems, ensuring a steady growth trajectory. Source: Grand View Research.
- Government R&D Funding in the U.S.:In the future, the National Institutes of Health (NIH) allocated USD 2.6 billion for regenerative medicine, while the Biomedical Advanced Research and Development Authority (BARDA) invested over USD 500 million in manufacturing infrastructure. This substantial funding underscores the government's commitment to advancing cell therapy technologies, thereby enhancing the market for automated processing systems. Source: Emergen Research.
Market Challenges
- High Upfront Costs:The initial capital expenditures required for automated closed cell therapy processing systems are substantial, posing a significant barrier to entry for smaller biotech companies and academic institutions. While specific numeric values are not disclosed, the financial constraints limit the ability of these entities to invest in advanced technologies, hindering overall market growth. Source: Market.us report.
- Regulatory Complexity:The lack of harmonized regulatory standards across different jurisdictions complicates the approval process for automated systems. Each region has distinct validation requirements, which can delay product launches and increase compliance costs. This regulatory complexity poses a challenge for companies seeking to scale their operations internationally, impacting market accessibility. Source: GlobeNewswire.
Global Automated Closed Cell Therapy Processing Systems Market Future Outlook
The future of the automated closed cell therapy processing systems market appears promising, driven by technological advancements and increasing demand for personalized medicine. The integration of artificial intelligence and machine learning into processing systems is expected to enhance efficiency and accuracy, while the shift towards decentralized manufacturing will facilitate broader access to therapies. As regulatory frameworks evolve, companies will likely find new opportunities for growth and innovation in this dynamic landscape.
Market Opportunities
- Asia-Pacific Market Growth:The Asia-Pacific region is projected to reach a market size of USD 280 million, with a rapid growth rate driven by increasing investments in biotechnology and supportive government policies. This under-penetrated market presents significant opportunities for companies looking to expand their footprint in high-growth areas. Source: Growth Market Reports.
- Modular, Automation-Enabled Platforms:The development of next-generation modular systems that incorporate real-time monitoring and AI/data analytics offers substantial market opportunities. These innovations can enhance operational efficiency and scalability, making them attractive to manufacturers aiming to optimize their production processes. Source: Growth Market Reports.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Automated Cell Processing Systems Semi-Automated Cell Processing Systems Manual Cell Processing Systems Others |
| By End-User | Hospitals Research Laboratories Biopharmaceutical Companies Contract Manufacturing Organizations Others |
| By Application | Cancer Treatment Genetic Disorders Autoimmune Diseases Others |
| By Component | Cell Separation Systems Cell Culture Systems Cell Storage Systems Others |
| By Technology | Magnetic Separation Density Gradient Centrifugation Filtration Technology Others |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Market Type | Research Market Commercial Market Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., Food and Drug Administration, European Medicines Agency)
Manufacturers and Producers
Healthcare Providers and Hospitals
Biotechnology and Pharmaceutical Companies
Technology Providers
Industry Associations
Financial Institutions
Players Mentioned in the Report:
Thermo Fisher Scientific
Beckman Coulter
Miltenyi Biotec
Merck KGaA
Lonza Group
STEMCELL Technologies
Bio-Rad Laboratories
GE Healthcare
Fujifilm Irvine Scientific
Sartorius AG
Eppendorf AG
Agilent Technologies
BD Biosciences
CellGenix GmbH
Pluristem Therapeutics
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Automated Closed Cell Therapy Processing Systems Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Automated Closed Cell Therapy Processing Systems Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Automated Closed Cell Therapy Processing Systems Market Analysis
3.1 Growth Drivers
3.1.1 Increasing demand for personalized medicine
3.1.2 Advancements in cell therapy technologies
3.1.3 Rising prevalence of chronic diseases
3.1.4 Supportive government initiatives and funding
3.2 Market Challenges
3.2.1 High costs of automated systems
3.2.2 Regulatory hurdles and compliance issues
3.2.3 Limited awareness among healthcare providers
3.2.4 Technical complexities in system integration
3.3 Market Opportunities
3.3.1 Expansion into emerging markets
3.3.2 Collaborations with research institutions
3.3.3 Development of cost-effective solutions
3.3.4 Increasing investment in R&D
3.4 Market Trends
3.4.1 Growing adoption of AI and machine learning
3.4.2 Shift towards decentralized manufacturing
3.4.3 Integration of IoT in therapy processing
3.4.4 Focus on sustainability and eco-friendly practices
3.5 Government Regulation
3.5.1 FDA guidelines for cell therapy products
3.5.2 EMA regulations on advanced therapy medicinal products
3.5.3 Compliance with GMP standards
3.5.4 National health policies promoting cell therapies
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Automated Closed Cell Therapy Processing Systems Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Automated Closed Cell Therapy Processing Systems Market Segmentation
8.1 By Type
8.1.1 Automated Cell Processing Systems
8.1.2 Semi-Automated Cell Processing Systems
8.1.3 Manual Cell Processing Systems
8.1.4 Others
8.2 By End-User
8.2.1 Hospitals
8.2.2 Research Laboratories
8.2.3 Biopharmaceutical Companies
8.2.4 Contract Manufacturing Organizations
8.2.5 Others
8.3 By Application
8.3.1 Cancer Treatment
8.3.2 Genetic Disorders
8.3.3 Autoimmune Diseases
8.3.4 Others
8.4 By Component
8.4.1 Cell Separation Systems
8.4.2 Cell Culture Systems
8.4.3 Cell Storage Systems
8.4.4 Others
8.5 By Technology
8.5.1 Magnetic Separation
8.5.2 Density Gradient Centrifugation
8.5.3 Filtration Technology
8.5.4 Others
8.6 By Region
8.6.1 North America
8.6.2 Europe
8.6.3 Asia-Pacific
8.6.4 Latin America
8.6.5 Middle East & Africa
8.7 By Market Type
8.7.1 Research Market
8.7.2 Commercial Market
8.7.3 Others
9. Global Automated Closed Cell Therapy Processing Systems Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Penetration Rate
9.2.5 Customer Retention Rate
9.2.6 Pricing Strategy
9.2.7 Product Innovation Rate
9.2.8 Operational Efficiency
9.2.9 Sales Conversion Rate
9.2.10 Customer Satisfaction Score
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Thermo Fisher Scientific
9.5.2 Beckman Coulter
9.5.3 Miltenyi Biotec
9.5.4 Merck KGaA
9.5.5 Lonza Group
9.5.6 STEMCELL Technologies
9.5.7 Bio-Rad Laboratories
9.5.8 GE Healthcare
9.5.9 Fujifilm Irvine Scientific
9.5.10 Sartorius AG
9.5.11 Eppendorf AG
9.5.12 Agilent Technologies
9.5.13 BD Biosciences
9.5.14 CellGenix GmbH
9.5.15 Pluristem Therapeutics
10. Global Automated Closed Cell Therapy Processing Systems Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Preferred Procurement Channels
10.1.4 Evaluation Criteria for Suppliers
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends in Healthcare Infrastructure
10.2.2 Spending on Technology Upgrades
10.2.3 Budgeting for Research and Development
10.2.4 Energy Efficiency Initiatives
10.3 Pain Point Analysis by End-User Category
10.3.1 Challenges in Implementation
10.3.2 Cost-Related Issues
10.3.3 Technical Support Needs
10.3.4 Regulatory Compliance Concerns
10.4 User Readiness for Adoption
10.4.1 Training and Support Requirements
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Feedback Mechanisms
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Measurement of Success Metrics
10.5.2 Case Studies of Successful Implementations
10.5.3 Opportunities for Upscaling
10.5.4 Long-Term Value Realization
11. Global Automated Closed Cell Therapy Processing Systems Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Value Proposition Development
1.3 Revenue Streams Analysis
1.4 Cost Structure Evaluation
1.5 Key Partnerships Exploration
1.6 Customer Segmentation
1.7 Channels of Distribution
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
2.3 Target Audience Identification
2.4 Communication Strategies
2.5 Digital Marketing Approaches
2.6 Event Marketing Plans
2.7 Partnership Marketing
3. Distribution Plan
3.1 Urban Retail Strategies
3.2 Rural NGO Tie-Ups
3.3 Online Distribution Channels
3.4 Direct Sales Approaches
3.5 Distributor Partnerships
3.6 Logistics and Supply Chain Management
3.7 Inventory Management Solutions
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands Analysis
4.3 Competitor Pricing Strategies
4.4 Customer Willingness to Pay
4.5 Discounting Strategies
4.6 Bundling Opportunities
4.7 Value-Based Pricing Models
5. Unmet Demand & Latent Needs
5.1 Category Gaps Identification
5.2 Consumer Segments Analysis
5.3 Emerging Trends Exploration
5.4 Feedback from Current Users
5.5 Future Needs Assessment
5.6 Innovation Opportunities
5.7 Market Entry Barriers
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-Sales Service
6.3 Customer Feedback Mechanisms
6.4 Engagement Strategies
6.5 Retention Strategies
6.6 Customer Education Initiatives
6.7 Community Building Efforts
7. Value Proposition
7.1 Sustainability Initiatives
7.2 Integrated Supply Chains
7.3 Cost-Effectiveness
7.4 Quality Assurance
7.5 Customer-Centric Innovations
7.6 Competitive Differentiation
7.7 Long-Term Partnerships
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding Initiatives
8.3 Distribution Setup
8.4 Training and Development
8.5 Market Research Activities
8.6 Stakeholder Engagement
8.7 Performance Monitoring
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging Considerations
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 Joint Ventures
10.2 Greenfield Investments
10.3 Mergers & Acquisitions
10.4 Distributor Model
10.5 Risk Assessment
10.6 Strategic Alliances
10.7 Market Adaptation Strategies
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines for Implementation
11.3 Funding Sources
11.4 Financial Projections
11.5 Cost-Benefit Analysis
11.6 Investment Return Expectations
11.7 Risk Mitigation Strategies
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
12.2 Risk Management Framework
12.3 Control Mechanisms
12.4 Partnership Evaluation Criteria
12.5 Long-Term Strategic Goals
12.6 Flexibility in Operations
12.7 Exit Strategies
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-Term Sustainability
13.3 Profit Margin Projections
13.4 Revenue Growth Strategies
13.5 Cost Management Techniques
13.6 Financial Health Indicators
13.7 Market Positioning for Profitability
14. Potential Partner List
14.1 Distributors
14.2 Joint Ventures
14.3 Acquisition Targets
14.4 Strategic Alliances
14.5 Research Collaborations
14.6 Technology Partners
14.7 Supply Chain Partners
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Timeline for Key Activities
15.2.2 Milestone Tracking
15.2.3 Performance Metrics
15.2.4 Resource Allocation
Research Methodology
Phase 1: Approach1
Desk Research
- Industry reports from leading market research firms focusing on automated closed cell therapy systems
- Published articles and white papers from academic journals on advancements in cell therapy technologies
- Regulatory guidelines and updates from health authorities such as the FDA and EMA regarding closed cell therapy
Primary Research
- Interviews with key opinion leaders in the field of regenerative medicine and cell therapy
- Surveys conducted with clinical researchers and practitioners involved in cell therapy applications
- Field interviews with technology developers and manufacturers of automated closed cell therapy systems
Validation & Triangulation
- Cross-validation of findings through multiple data sources including market reports and expert interviews
- Triangulation of quantitative data from sales figures and qualitative insights from expert opinions
- Sanity checks through peer reviews and feedback from industry panels
Phase 2: Market Size Estimation1
Top-down Assessment
- Analysis of global healthcare expenditure trends and their impact on cell therapy investments
- Segmentation of the market by application areas such as oncology, orthopedics, and neurology
- Incorporation of growth rates from emerging markets and technological advancements in automation
Bottom-up Modeling
- Estimation of market size based on sales data from leading manufacturers of closed cell therapy systems
- Operational cost analysis derived from production and distribution expenses of automated systems
- Volume projections based on clinical trial data and adoption rates in healthcare facilities
Forecasting & Scenario Analysis
- Multi-variable forecasting using factors such as technological innovation and regulatory changes
- Scenario modeling based on potential market disruptions and shifts in healthcare policies
- Development of baseline, optimistic, and pessimistic market growth projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Oncology Cell Therapy Applications | 100 | Clinical Oncologists, Research Scientists |
| Orthopedic Regenerative Medicine | 80 | Orthopedic Surgeons, Rehabilitation Specialists |
| Neurology and Cell Therapy | 70 | Neurologists, Clinical Researchers |
| Manufacturers of Automated Systems | 60 | Product Managers, R&D Directors |
| Healthcare Facility Administrators | 90 | Hospital Administrators, Procurement Officers |
Frequently Asked Questions
What is the current value of the Global Automated Closed Cell Therapy Processing Systems Market?
The Global Automated Closed Cell Therapy Processing Systems Market is valued at approximately USD 1.85 billion, driven by the increasing demand for cell and gene therapies, particularly CAR-T and immunotherapies, which benefit from automation's enhanced efficiency and reproducibility.