Region:Global
Author(s):Geetanshi
Product Code:KRAD0064
Pages:86
Published On:August 2025
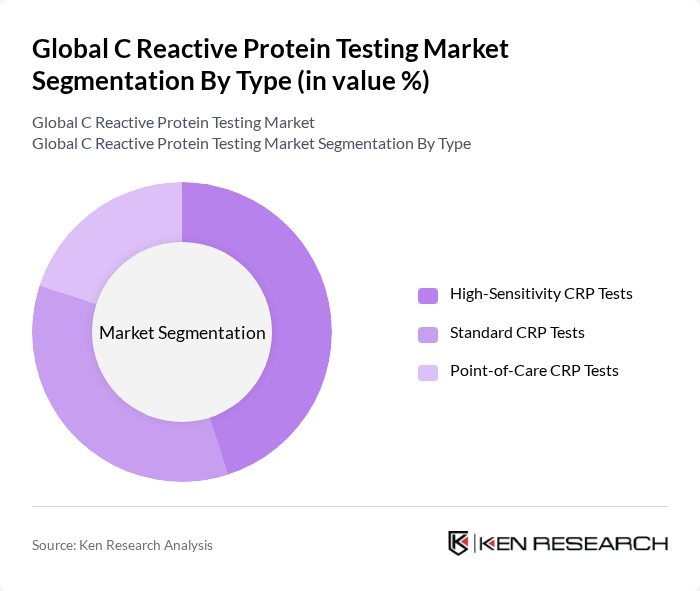
By Type:The market is segmented into High-Sensitivity CRP Tests, Standard CRP Tests, and Point-of-Care CRP Tests. High-Sensitivity CRP Tests are increasingly preferred for their ability to detect low levels of CRP, which is critical for cardiovascular risk assessment and early detection of chronic inflammation. Standard CRP Tests remain widely used in clinical settings for general inflammation detection, while Point-of-Care Tests are gaining popularity for their convenience, rapid turnaround times, and utility in emergency and outpatient settings.
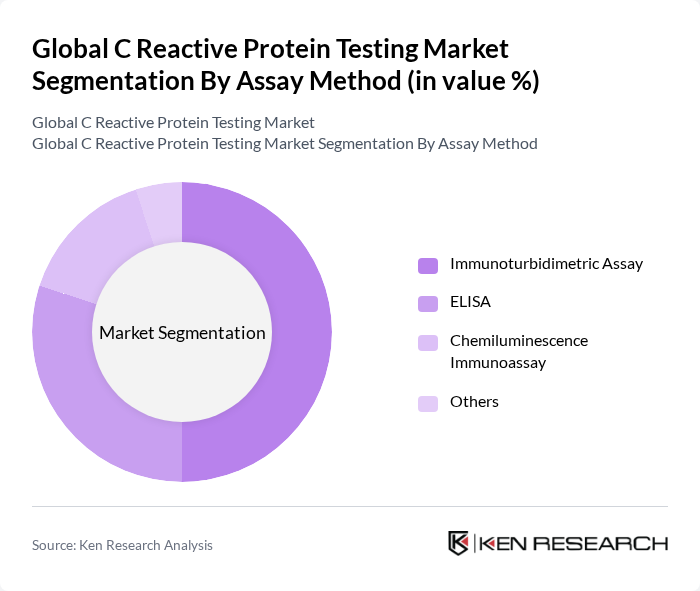
By Assay Method:The market is categorized into Immunoturbidimetric Assay, ELISA, Chemiluminescence Immunoassay, and Others. Immunoturbidimetric Assay is the most widely adopted method due to its high accuracy, reliability, and compatibility with automated laboratory analyzers. ELISA is valued for its sensitivity and specificity, making it suitable for both clinical and research applications. Chemiluminescence Immunoassay is gaining traction for its rapid processing times and enhanced analytical performance. The "Others" category includes emerging technologies such as electrochemical impedance spectroscopy and microneedle-based sensors, which are being developed to further improve CRP detection capabilities.
The Global C Reactive Protein Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Abbott Laboratories, Roche Diagnostics (F. Hoffmann-La Roche Ltd.), Siemens Healthineers, Beckman Coulter (Danaher Corporation), Thermo Fisher Scientific, Inc., Ortho Clinical Diagnostics, Bio-Rad Laboratories, Inc., QuidelOrtho Corporation, Becton, Dickinson and Company (BD), Sysmex Corporation, Mindray Medical International Limited, Hologic, Inc., DiaSorin S.p.A., Fujirebio, Grifols S.A., Quest Diagnostics Incorporated, Getein Biotech, Inc., Merck KGaA, Zoetis Inc. contribute to innovation, geographic expansion, and service delivery in this space.
The future of CRP testing in the None region appears promising, driven by increasing healthcare investments and a shift towards personalized medicine. As healthcare systems prioritize preventive care, the demand for CRP testing is expected to rise. Additionally, the integration of artificial intelligence in diagnostic processes is likely to enhance testing accuracy and efficiency. These trends indicate a robust growth trajectory for CRP testing, with significant advancements anticipated in technology and patient care strategies.
| Segment | Sub-Segments |
|---|---|
| By Type | High-Sensitivity CRP Tests Standard CRP Tests Point-of-Care CRP Tests |
| By Assay Method | Immunoturbidimetric Assay ELISA Chemiluminescence Immunoassay Others |
| By Detection Range | hs-CRP (High Sensitivity) Conventional CRP cCRP |
| By Disease Area | Cardiovascular Diseases Cancer Rheumatoid Arthritis Inflammatory Bowel Disease Endometriosis Lupus Others |
| By End-User | Hospitals & Clinics Diagnostic Laboratories Home Care Settings Others |
| By Application | Cardiovascular Disease Diagnosis Inflammatory Disease Monitoring Infection Detection Others |
| By Distribution Channel | Direct Sales Online Sales Distributors |
| By Region | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Pricing Model | Premium Pricing Competitive Pricing Value-Based Pricing |
| By Others | Niche Testing Solutions Emerging Technologies |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Clinical Laboratories | 120 | Laboratory Managers, Technicians |
| Healthcare Providers | 100 | Physicians, Nurse Practitioners |
| Diagnostic Equipment Manufacturers | 60 | Product Managers, Sales Directors |
| Health Insurance Companies | 50 | Policy Analysts, Underwriters |
| Patient Advocacy Groups | 40 | Advocacy Coordinators, Community Outreach Managers |
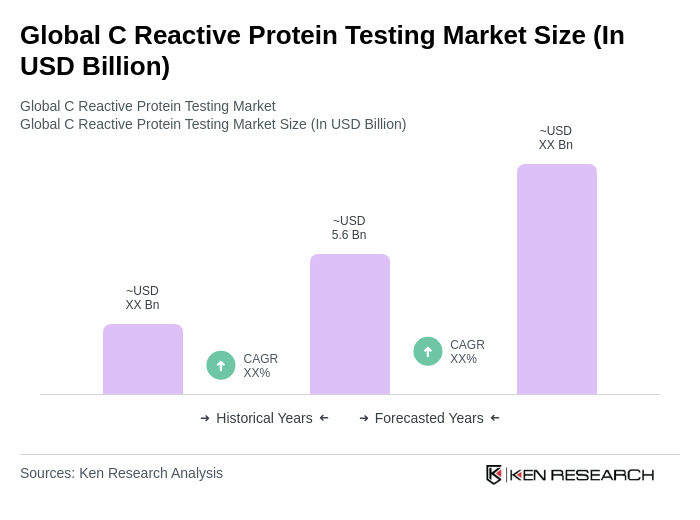
The Global C Reactive Protein Testing Market is valued at approximately USD 5.6 billion, driven by the rising prevalence of chronic diseases and advancements in diagnostic technologies. This market is expected to grow significantly in the coming years.