Region:Middle East
Author(s):Shubham
Product Code:KRAD3535
Pages:85
Published On:November 2025
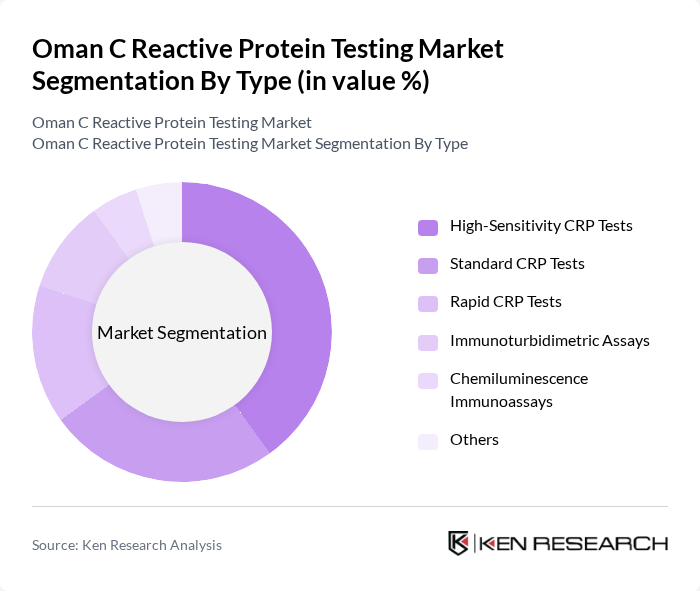
By Type:The market is segmented into various types of CRP tests, including High-Sensitivity CRP Tests, Standard CRP Tests, Rapid CRP Tests, Immunoturbidimetric Assays, Chemiluminescence Immunoassays, and Others. Among these, High-Sensitivity CRP Tests are gaining traction due to their ability to detect low levels of CRP, which is crucial for assessing cardiovascular risks. The increasing focus on preventive healthcare and personalized medicine is driving the demand for these advanced testing methods. Immunoturbidimetric and chemiluminescence immunoassays are also widely adopted due to their accuracy and efficiency in clinical laboratories .
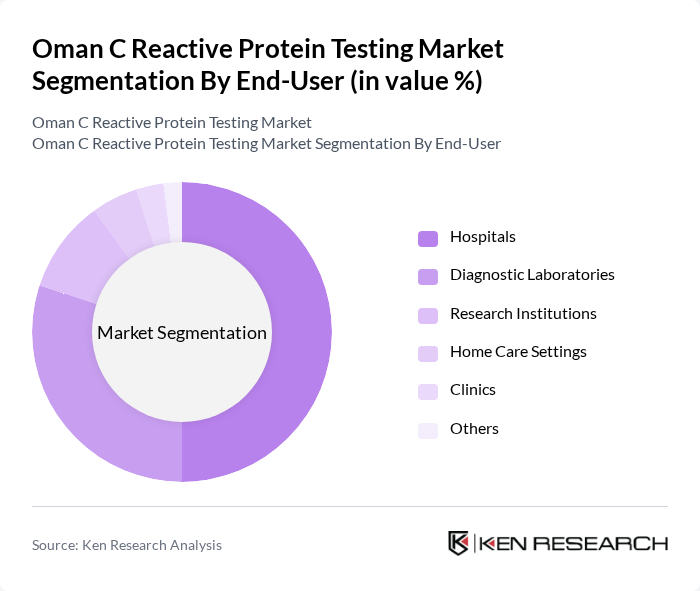
By End-User:The end-user segmentation includes Hospitals, Diagnostic Laboratories, Research Institutions, Home Care Settings, Clinics, and Others. Hospitals are the leading end-users due to their comprehensive healthcare services and the need for accurate diagnostic tools in patient management. The increasing number of hospital admissions for chronic diseases is further propelling the demand for CRP testing in these facilities. Diagnostic laboratories are also significant contributors, supported by the expansion of private healthcare and laboratory networks in Oman .
The Oman C Reactive Protein Testing Market is characterized by a dynamic mix of regional and international players. Leading participants such as Roche Diagnostics, Abbott Laboratories, Siemens Healthineers, Beckman Coulter, Thermo Fisher Scientific, Ortho Clinical Diagnostics, Bio-Rad Laboratories, Sysmex Corporation, QuidelOrtho Corporation, Mindray Medical International, Randox Laboratories, Becton, Dickinson and Company, DiaSorin S.p.A., Fujifilm Holdings Corporation, Grifols S.A. contribute to innovation, geographic expansion, and service delivery in this space.
The future of the CRP testing market in Oman appears promising, driven by ongoing healthcare reforms and increased investment in medical technology. The government is prioritizing healthcare infrastructure improvements, which will enhance access to diagnostic services. Additionally, the growing trend towards personalized medicine and the integration of artificial intelligence in diagnostics are expected to further streamline testing processes, making CRP testing more efficient and accessible to a broader population.
| Segment | Sub-Segments |
|---|---|
| By Type | High-Sensitivity CRP Tests Standard CRP Tests Rapid CRP Tests Immunoturbidimetric Assays Chemiluminescence Immunoassays Others |
| By End-User | Hospitals Diagnostic Laboratories Research Institutions Home Care Settings Clinics Others |
| By Application | Cardiovascular Disease Diagnosis Inflammatory Disease Monitoring Infection Detection Autoimmune Disease Assessment Others |
| By Distribution Channel | Direct Sales Online Sales Distributors Hospital Procurement Others |
| By Region | Muscat Dhofar Al Batinah Al Dakhiliyah Others |
| By Technology | Immunoassays Enzyme-Linked Immunosorbent Assay (ELISA) Lateral Flow Assays Chemiluminescence Immunoassays Immunoturbidimetric Assays Others |
| By Policy Support | Government Subsidies Tax Incentives Research Grants Others |
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Hospital Laboratories | 40 | Laboratory Managers, Pathologists |
| Private Diagnostic Centers | 40 | Clinical Directors, Lab Technicians |
| General Practitioners | 40 | Family Physicians, Internal Medicine Specialists |
| Patient Awareness Surveys | 150 | Patients, Caregivers |
| Healthcare Policy Makers | 40 | Health Administrators, Policy Analysts |
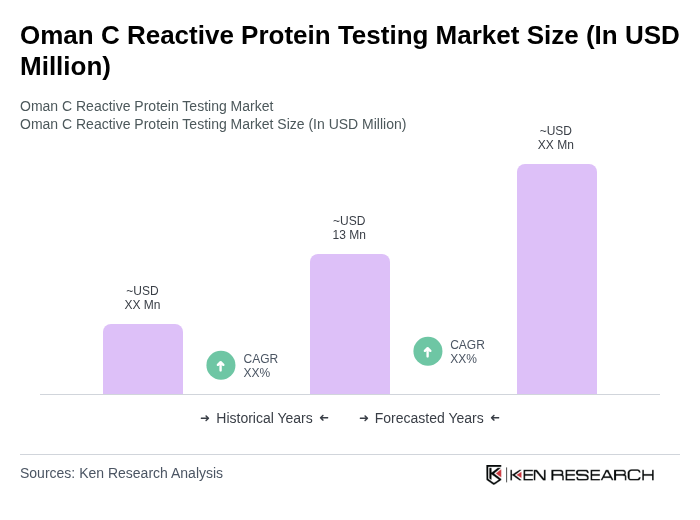
The Oman C Reactive Protein Testing Market is valued at approximately USD 13 million, reflecting a five-year historical analysis and normalization from global and regional market size estimates. This growth is driven by the rising prevalence of chronic diseases and advancements in diagnostic technologies.