About the Report
Base Year 2024Global Cardiac Safety Services Market Overview
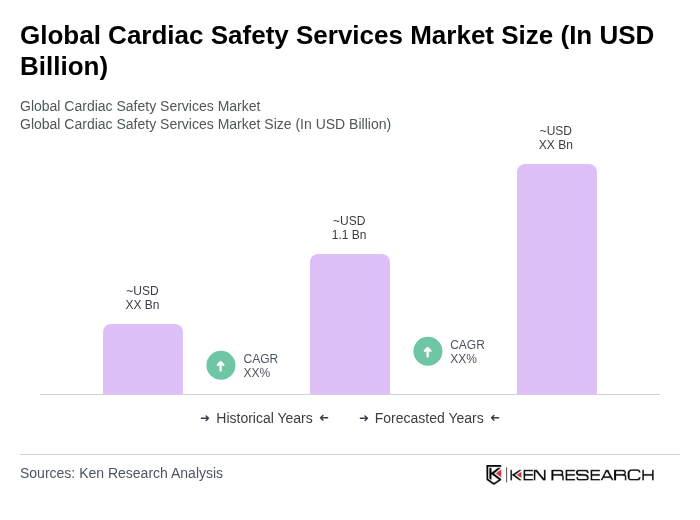
- The Global Cardiac Safety Services Market is valued at USD 1.1 billion, based on a five-year historical analysis. This growth is primarily driven by the increasing prevalence of cardiovascular diseases, the rising demand for cardiac safety monitoring during drug development, and the growing emphasis on regulatory compliance in clinical trials. The market is also supported by advancements in artificial intelligence for ECG analytics, the adoption of digital health solutions, and the expansion of post-marketing surveillance frameworks. The integration of high-density wearable devices and real-time telemetry further enhances the precision and efficiency of cardiac safety assessments .
- Key players in this market are predominantly located in North America and Europe, with the United States and Germany being the most significant contributors. The dominance of these regions can be attributed to their advanced healthcare infrastructure, high investment in research and development, and stringent regulatory frameworks that necessitate comprehensive cardiac safety assessments in clinical trials. North America controls the largest share of market revenue, supported by early adoption of AI-assisted telemetry and harmonized regulatory standards .
- In 2022, the U.S. Food and Drug Administration (FDA) issued the updated "E2D(R1) Post-Approval Safety Data Management: Definitions and Standards for Expedited Reporting" guidance, mandating comprehensive cardiac safety data for all new drug applications. This regulation requires sponsors to include robust cardiac safety assessments, including real-world evidence and digital biomarker integration, to enhance patient safety and ensure that potential cardiac risks are thoroughly evaluated prior to drug approval .
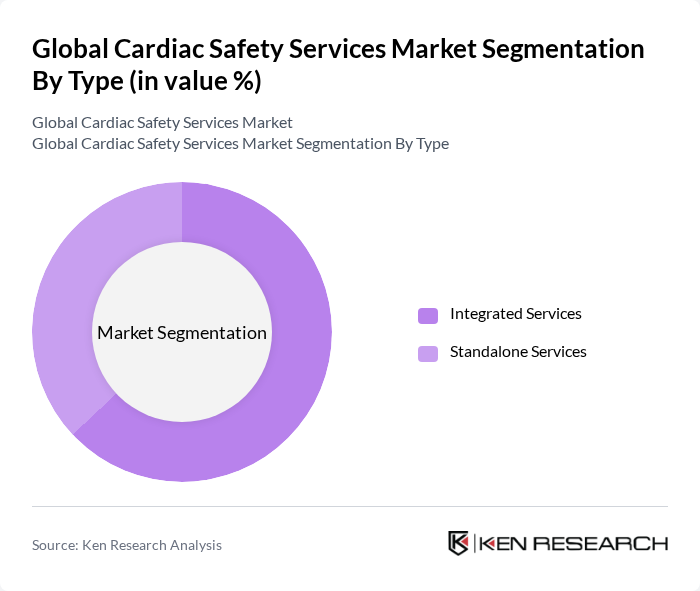
Global Cardiac Safety Services Market Segmentation
By Type:The market is segmented into Integrated Services and Standalone Services. Integrated Services remain the dominant segment, representing the largest market share due to their comprehensive approach, which combines multiple safety assessments into a single package, making them more appealing to pharmaceutical companies. Standalone Services are gaining traction, particularly for specialized or complex studies, as sponsors increasingly seek deep domain expertise for oncology, gene therapy, and high-resolution imaging protocols .
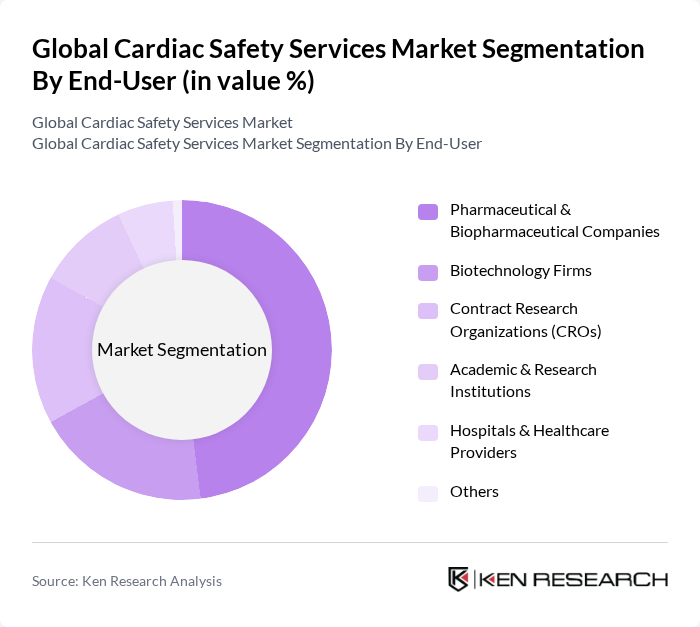
By End-User:The end-user segmentation includes Pharmaceutical & Biopharmaceutical Companies, Biotechnology Firms, Contract Research Organizations (CROs), Academic & Research Institutions, Hospitals & Healthcare Providers, and Others. Pharmaceutical & Biopharmaceutical Companies continue to dominate this segment due to their extensive need for cardiac safety services during drug development and clinical trials. However, hospitals and healthcare providers are experiencing the fastest growth, driven by the adoption of advanced cardiac monitoring technologies and the increasing complexity of cardiovascular patient management .
Global Cardiac Safety Services Market Competitive Landscape
The Global Cardiac Safety Services Market is characterized by a dynamic mix of regional and international players. Leading participants such as Clario (formerly ERT), BioTelemetry, Inc. (a Philips company), Banook Group, Medpace, Inc., ICON plc, IQVIA, Parexel International Corporation, Covance (LabCorp Drug Development), Charles River Laboratories, SGS SA, Celerion, Inc., Richmond Pharmacology, Bioclinica (now part of Clario), Certara, Inc., Eurofins Scientific contribute to innovation, geographic expansion, and service delivery in this space.
Global Cardiac Safety Services Market Industry Analysis
Growth Drivers
- Increasing Prevalence of Cardiovascular Diseases:The World Health Organization reported that cardiovascular diseases (CVDs) are responsible for approximately 17.9 million deaths annually, accounting for 32% of all global deaths. In future, the prevalence of CVDs is projected to rise, with an estimated 1.4 billion adults affected worldwide. This alarming trend drives the demand for cardiac safety services, as pharmaceutical companies and clinical research organizations seek to ensure drug safety and efficacy in this vulnerable population.
- Rising Demand for Clinical Trials:The global clinical trials market is expected to reach $65 billion in future, driven by the increasing need for innovative therapies and drug development. With over 300,000 clinical trials registered globally, the demand for cardiac safety assessments is surging. This growth is fueled by the need for comprehensive safety evaluations, particularly in cardiovascular drug development, where patient safety is paramount, necessitating robust cardiac safety services.
- Advancements in Cardiac Safety Technologies:The cardiac safety services sector is witnessing significant technological advancements, including the integration of wearable devices and remote monitoring systems. In future, the global market for wearable health technology is projected to exceed $60 billion. These innovations enhance real-time monitoring of cardiac parameters, improving patient outcomes and driving the adoption of cardiac safety services as pharmaceutical companies leverage these technologies for more effective drug safety assessments.
Market Challenges
- High Costs Associated with Cardiac Safety Services:The financial burden of cardiac safety services can be substantial, with costs ranging from $100,000 to over $1 million per clinical trial, depending on the complexity and duration. This high expenditure poses a significant challenge for smaller pharmaceutical companies and research organizations, potentially limiting their ability to conduct comprehensive cardiac safety assessments and hindering overall market growth.
- Limited Awareness Among Healthcare Providers:A survey conducted by the American Heart Association revealed that nearly 40% of healthcare providers lack adequate knowledge regarding cardiac safety assessments. This gap in awareness can lead to underutilization of cardiac safety services, as providers may not fully understand the importance of these assessments in drug development, ultimately impacting patient safety and the effectiveness of new therapies.
Global Cardiac Safety Services Market Future Outlook
The future of cardiac safety services is poised for growth, driven by technological advancements and an increasing focus on patient-centric care. As healthcare systems evolve, the integration of artificial intelligence and machine learning will enhance the efficiency of cardiac safety assessments. Additionally, the growing emphasis on real-world evidence will further support the development of innovative therapies, ensuring that cardiac safety remains a priority in drug development and regulatory compliance.
Market Opportunities
- Expansion of Telemedicine and Remote Monitoring:The telemedicine market is projected to reach $459 billion in future, creating opportunities for cardiac safety services to integrate remote monitoring solutions. This expansion allows for continuous patient monitoring, improving data collection and enhancing the safety assessment process, ultimately leading to better patient outcomes and increased service adoption.
- Collaborations Between Pharmaceutical Companies and Service Providers:Strategic partnerships are on the rise, with over 200 collaborations reported in future alone. These alliances enable pharmaceutical companies to leverage specialized cardiac safety services, enhancing their drug development processes. Such collaborations can lead to improved safety profiles for new therapies, fostering innovation and driving market growth in the cardiac safety sector.
Scope of the Report
| Segment | Sub-Segments |
|---|---|
| By Type | Integrated Services Standalone Services |
| By End-User | Pharmaceutical & Biopharmaceutical Companies Biotechnology Firms Contract Research Organizations (CROs) Academic & Research Institutions Hospitals & Healthcare Providers Others |
| By Service Type | Cardiac Safety Monitoring (e.g., ECG, Holter, Telemetry) Data Management & Analysis Services Risk Assessment & Regulatory Consulting Cardiovascular Imaging Services Others |
| By Geography | North America Europe Asia-Pacific Latin America Middle East & Africa |
| By Technology | Electrocardiogram (ECG) Monitoring Holter Monitoring Event Monitoring Ambulatory Blood Pressure Monitoring Cardiac Imaging (e.g., Echocardiography, MRI) Others |
| By Application | Drug Development Clinical Trials (Phase I-IV) Regulatory Compliance & Risk Management Chronic Disease Management Others |
| By Policy Support | Government Grants Tax Incentives Research Funding Others |
Key Target Audience
Investors and Venture Capitalist Firms
Government and Regulatory Bodies (e.g., U.S. Food and Drug Administration, European Medicines Agency)
Pharmaceutical Companies
Biotechnology Firms
Contract Research Organizations (CROs)
Medical Device Manufacturers
Health Insurance Providers
Clinical Research Institutions
Players Mentioned in the Report:
Clario (formerly ERT)
BioTelemetry, Inc. (a Philips company)
Banook Group
Medpace, Inc.
ICON plc
IQVIA
Parexel International Corporation
Covance (LabCorp Drug Development)
Charles River Laboratories
SGS SA
Celerion, Inc.
Richmond Pharmacology
Bioclinica (now part of Clario)
Certara, Inc.
Eurofins Scientific
Table of Contents
Market Assessment Phase
1. Executive Summary and Approach
2. Global Cardiac Safety Services Market Overview
2.1 Key Insights and Strategic Recommendations
2.2 Global Cardiac Safety Services Market Overview
2.3 Definition and Scope
2.4 Evolution of Market Ecosystem
2.5 Timeline of Key Regulatory Milestones
2.6 Value Chain & Stakeholder Mapping
2.7 Business Cycle Analysis
2.8 Policy & Incentive Landscape
3. Global Cardiac Safety Services Market Analysis
3.1 Growth Drivers
3.1.1 Increasing prevalence of cardiovascular diseases
3.1.2 Rising demand for clinical trials
3.1.3 Advancements in cardiac safety technologies
3.1.4 Growing regulatory requirements for drug safety
3.2 Market Challenges
3.2.1 High costs associated with cardiac safety services
3.2.2 Limited awareness among healthcare providers
3.2.3 Stringent regulatory frameworks
3.2.4 Competition from alternative safety assessment methods
3.3 Market Opportunities
3.3.1 Expansion of telemedicine and remote monitoring
3.3.2 Increasing investment in healthcare R&D
3.3.3 Collaborations between pharmaceutical companies and service providers
3.3.4 Development of personalized medicine approaches
3.4 Market Trends
3.4.1 Integration of AI and machine learning in cardiac safety
3.4.2 Shift towards patient-centric care models
3.4.3 Growing emphasis on real-world evidence
3.4.4 Rise in outsourcing of cardiac safety services
3.5 Government Regulation
3.5.1 FDA guidelines on cardiac safety assessments
3.5.2 EMA regulations for drug safety monitoring
3.5.3 ICH E14 guidelines for cardiac safety studies
3.5.4 National health policies promoting cardiovascular health
4. SWOT Analysis
5. Stakeholder Analysis
6. Porter's Five Forces Analysis
7. Global Cardiac Safety Services Market Market Size, 2019-2024
7.1 By Value
7.2 By Volume
7.3 By Average Selling Price
8. Global Cardiac Safety Services Market Segmentation
8.1 By Type
8.1.1 Integrated Services
8.1.2 Standalone Services
8.2 By End-User
8.2.1 Pharmaceutical & Biopharmaceutical Companies
8.2.2 Biotechnology Firms
8.2.3 Contract Research Organizations (CROs)
8.2.4 Academic & Research Institutions
8.2.5 Hospitals & Healthcare Providers
8.2.6 Others
8.3 By Service Type
8.3.1 Cardiac Safety Monitoring (e.g., ECG, Holter, Telemetry)
8.3.2 Data Management & Analysis Services
8.3.3 Risk Assessment & Regulatory Consulting
8.3.4 Cardiovascular Imaging Services
8.3.5 Others
8.4 By Geography
8.4.1 North America
8.4.2 Europe
8.4.3 Asia-Pacific
8.4.4 Latin America
8.4.5 Middle East & Africa
8.5 By Technology
8.5.1 Electrocardiogram (ECG) Monitoring
8.5.2 Holter Monitoring
8.5.3 Event Monitoring
8.5.4 Ambulatory Blood Pressure Monitoring
8.5.5 Cardiac Imaging (e.g., Echocardiography, MRI)
8.5.6 Others
8.6 By Application
8.6.1 Drug Development
8.6.2 Clinical Trials (Phase I-IV)
8.6.3 Regulatory Compliance & Risk Management
8.6.4 Chronic Disease Management
8.6.5 Others
8.7 By Policy Support
8.7.1 Government Grants
8.7.2 Tax Incentives
8.7.3 Research Funding
8.7.4 Others
9. Global Cardiac Safety Services Market Competitive Analysis
9.1 Market Share of Key Players
9.2 Cross Comparison of Key Players
9.2.1 Company Name
9.2.2 Group Size (Large, Medium, or Small as per industry convention)
9.2.3 Revenue Growth Rate
9.2.4 Market Share in Cardiac Safety Services
9.2.5 Number of Cardiac Safety Studies Conducted Annually
9.2.6 Global Geographic Coverage (Number of Countries/Regions Served)
9.2.7 Service Portfolio Breadth (Number of Distinct Cardiac Safety Services)
9.2.8 Regulatory Compliance Rate (e.g., FDA/EMA/ICH adherence)
9.2.9 Client Retention Rate
9.2.10 Innovation Index (e.g., AI/ML Integration, Digital Platforms)
9.2.11 Customer Satisfaction Score (NPS or Equivalent)
9.2.12 Strategic Partnerships & Collaborations
9.3 SWOT Analysis of Top Players
9.4 Pricing Analysis
9.5 Detailed Profile of Major Companies
9.5.1 Clario (formerly ERT)
9.5.2 BioTelemetry, Inc. (a Philips company)
9.5.3 Banook Group
9.5.4 Medpace, Inc.
9.5.5 ICON plc
9.5.6 IQVIA
9.5.7 Parexel International Corporation
9.5.8 Covance (LabCorp Drug Development)
9.5.9 Charles River Laboratories
9.5.10 SGS SA
9.5.11 Celerion, Inc.
9.5.12 Richmond Pharmacology
9.5.13 Bioclinica (now part of Clario)
9.5.14 Certara, Inc.
9.5.15 Eurofins Scientific
10. Global Cardiac Safety Services Market End-User Analysis
10.1 Procurement Behavior of Key Ministries
10.1.1 Budget Allocation Trends
10.1.2 Decision-Making Processes
10.1.3 Contracting Preferences
10.1.4 Others
10.2 Corporate Spend on Infrastructure & Energy
10.2.1 Investment Trends
10.2.2 Spending Priorities
10.2.3 Budgeting Cycles
10.2.4 Others
10.3 Pain Point Analysis by End-User Category
10.3.1 Common Challenges Faced
10.3.2 Service Gaps
10.3.3 Regulatory Hurdles
10.3.4 Others
10.4 User Readiness for Adoption
10.4.1 Training Needs
10.4.2 Technology Acceptance Levels
10.4.3 Infrastructure Readiness
10.4.4 Others
10.5 Post-Deployment ROI and Use Case Expansion
10.5.1 Performance Metrics
10.5.2 Case Studies
10.5.3 Future Use Cases
10.5.4 Others
11. Global Cardiac Safety Services Market Future Size, 2025-2030
11.1 By Value
11.2 By Volume
11.3 By Average Selling Price
Go-To-Market Strategy Phase
1. Whitespace Analysis + Business Model Canvas
1.1 Market Gaps Identification
1.2 Business Model Development
2. Marketing and Positioning Recommendations
2.1 Branding Strategies
2.2 Product USPs
3. Distribution Plan
3.1 Urban Retail vs Rural NGO Tie-ups
4. Channel & Pricing Gaps
4.1 Underserved Routes
4.2 Pricing Bands
5. Unmet Demand & Latent Needs
5.1 Category Gaps
5.2 Consumer Segments
6. Customer Relationship
6.1 Loyalty Programs
6.2 After-sales Service
7. Value Proposition
7.1 Sustainability
7.2 Integrated Supply Chains
8. Key Activities
8.1 Regulatory Compliance
8.2 Branding
8.3 Distribution Setup
9. Entry Strategy Evaluation
9.1 Domestic Market Entry Strategy
9.1.1 Product Mix
9.1.2 Pricing Band
9.1.3 Packaging
9.2 Export Entry Strategy
9.2.1 Target Countries
9.2.2 Compliance Roadmap
10. Entry Mode Assessment
10.1 JV
10.2 Greenfield
10.3 M&A
10.4 Distributor Model
11. Capital and Timeline Estimation
11.1 Capital Requirements
11.2 Timelines
12. Control vs Risk Trade-Off
12.1 Ownership vs Partnerships
13. Profitability Outlook
13.1 Breakeven Analysis
13.2 Long-term Sustainability
14. Potential Partner List
14.1 Distributors
14.2 JVs
14.3 Acquisition Targets
15. Execution Roadmap
15.1 Phased Plan for Market Entry
15.1.1 Market Setup
15.1.2 Market Entry
15.1.3 Growth Acceleration
15.1.4 Scale & Stabilize
15.2 Key Activities and Milestones
15.2.1 Milestone Planning
15.2.2 Activity Tracking
Research Methodology
Phase 1: Approach1
Desk Research
- Analysis of industry reports from healthcare organizations and market research firms
- Review of published articles in medical journals focusing on cardiac safety services
- Examination of regulatory guidelines and safety protocols from health authorities
Primary Research
- Interviews with cardiologists and clinical researchers specializing in cardiac safety
- Surveys conducted with healthcare administrators managing cardiac safety programs
- Focus groups with patients who have undergone cardiac safety assessments
Validation & Triangulation
- Cross-validation of findings through multiple expert interviews and literature reviews
- Triangulation of data from clinical trials, patient outcomes, and market trends
- Sanity checks through feedback from a panel of industry experts
Phase 2: Market Size Estimation1
Top-down Assessment
- Estimation of market size based on global healthcare expenditure on cardiac services
- Segmentation by geographic regions and types of cardiac safety services offered
- Incorporation of trends in telemedicine and remote patient monitoring
Bottom-up Modeling
- Data collection from leading cardiac safety service providers on service volumes
- Cost analysis based on pricing models for various cardiac safety assessments
- Volume x cost calculations for different service types and patient demographics
Forecasting & Scenario Analysis
- Multi-variable regression analysis incorporating factors like aging population and chronic disease prevalence
- Scenario modeling based on advancements in cardiac technology and regulatory changes
- Development of baseline, optimistic, and pessimistic market projections through 2030
Phase 3: CATI Sample Composition1
| Scope Item/Segment | Sample Size | Target Respondent Profiles |
|---|---|---|
| Cardiac Safety Assessments in Hospitals | 100 | Cardiologists, Hospital Administrators |
| Telecardiology Services | 60 | Telehealth Coordinators, IT Managers |
| Clinical Trials for Cardiac Drugs | 50 | Clinical Research Coordinators, Pharmacologists |
| Patient Monitoring Solutions | 40 | Healthcare IT Specialists, Cardiac Nurses |
| Regulatory Compliance in Cardiac Safety | 40 | Regulatory Affairs Managers, Compliance Officers |
Frequently Asked Questions
What is the current value of the Global Cardiac Safety Services Market?
The Global Cardiac Safety Services Market is valued at approximately USD 1.1 billion, reflecting a significant growth driven by the increasing prevalence of cardiovascular diseases and the demand for cardiac safety monitoring during drug development.